Abstract
Key message
PAP-FcK and PSA-FcK prostate cancer antigenic proteins transiently co-expressed in plant induce their specific humoral immune responses in mice.
Abstract
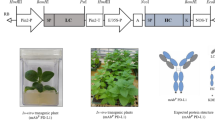
Prostate-specific antigen (PSA) and prostatic acid phosphatase (PAP) have been considered as immunotherapeutic antigens for prostate cancer. The use of a single antigenic agent is unlikely to be effective in eliciting immunotherapeutic responses due to the heterogeneous and multifocal nature of prostate cancer. Thus, multiple antigens have been combined to enhance their anti-cancer effects. In the current study, PSA and PAP were fused to the crystallizable region (Fc region) of immunoglobulin G1 and tagged with KDEL, the endoplasmic reticulum (ER) retention signal motif, to generate PSA-FcK and PAP-FcK, respectively, and were transiently co-expressed in Nicotiana benthamiana. Western blot analysis confirmed the co-expression of PSA-FcK and PAP-FcK (PSA-FcK + PAP-FcK) with a 1:3 ratios in the co-infiltrated plants. PSA-FcK, PAP-FcK, and PSA-FcK + PAP-FcK proteins were successfully purified from N. benthamiana by protein A affinity chromatography. ELISA showed that anti-PAP and anti-PSA antibodies successfully detected PAP-FcK and PSA-FcK, respectively, and both detected PSA-FcK + PAP-FcK. Surface plasmon resonance (SPR) analysis confirmed the binding affinity of the plant-derived Fc fusion proteins to FcγRI/CD64. Furthermore, we also confirmed that mice injected with PSA-FcK + PAP-FcK produced both PSA- and PAP-specific IgGs, demonstrating their immunogenicity. This study suggested that the transient plant expression system can be applied to produce the dual-antigen Fc fusion protein (PSA-FcK + PAP-FcK) for prostate cancer immunotherapy.









Similar content being viewed by others
Data availability
Non-applicable.
References
Arcalis E, Stadlmann J, Rademacher T, Marcel S, Sack M, Altmann F, Stoger E (2013) Plant species and organ influence the structure and subcellular localization of recombinant glycoproteins. Plant Mol Biol 83(1–2):105–117
Balk SP, Ko Y-J, Bubley GJ (2003) Biology of prostate-specific antigen. J Clin Oncol 21(2):383–391
Beck A, Reichert JM (2011) Therapeutic Fc-fusion proteins and peptides as successful alternatives to antibodies. Mabs 3(5):415–416. https://doi.org/10.4161/mabs.3.5.17334
Bicknell AA, Ricci EP (2017) When mRNA translation meets decay. Biochem Soc Trans 45(2):339–351
Correale P, Nieroda C, Zaremba S, Zhu M, Schlom J, Tsang KY, Konstantin W (1997) In vitro generation of human cytotoxic T lymphocytes specific for peptides derived from prostate-specific antigen. J Natl Cancer Inst 89(4):293–300
Daniell H, Singh ND, Mason H, Streatfield SJ (2009) Plant-made vaccine antigens and biopharmaceuticals. Trends Plant Sci 14(12):669–679
De Meyer T, Depicker A (2014) Trafficking of endoplasmic reticulum-retained recombinant proteins is unpredictable in Arabidopsis thaliana. Front Plant Sci 5:473
Fang J, Hu B, Li S, Zhang C, Liu Y, Wang P (2016) A multi-antigen vaccine in combination with an immunotoxin targeting tumor-associated fibroblast for treating murine melanoma. Mol Ther-Oncolytics 3:16007
Farsiani H, Mosavat A, Soleimanpour S, Sadeghian H, Eydgahi MRA, Ghazvini K, Rezaee SA (2016) Fc-based delivery system enhances immunogenicity of a tuberculosis subunit vaccine candidate consisting of the ESAT-6: CFP-10 complex. Mol BioSyst 12(7):2189–2201
Fay EK, Graff JN (2020) Immunotherapy in prostate cancer. Cancers 12(7):1752
Guilliams M, Bruhns P, Saeys Y, Hammad H, Lambrecht BN (2014) The function of Fcγ receptors in dendritic cells and macrophages. Nat Rev Immunol 14(2):94–108
Gulley J, Chen AP, Dahut W, Arlen PM, Bastian A, Steinberg SM, Schlom J (2002) Phase I study of a vaccine using recombinant vaccinia virus expressing PSA (rV-PSA) in patients with metastatic androgen-independent prostate cancer. Prostate 53(2):109–117
Gupta S, Carballido E, Fishman M (2011) Sipuleucel-T for therapy of asymptomatic or minimally symptomatic, castrate-refractory prostate cancer: an update and perspective among other treatments. Onco Targets Ther 4:79
Ha JH, Kim J-E, Kim YS (2016) Immunoglobulin Fc heterodimer platform technology: from design to applications in therapeutic antibodies and proteins. Front Immunol 6:394
Holtrop T, Budding K, Brandsma AM, Leusen JHW (2022) Targeting the high affinity receptor, FcγRI, in autoimmune disease, neuropathy, and cancer. Immunotherapy Advances 2:1–11
Karan D (2013) Prostate immunotherapy: should all guns be aimed at the prostate-specific antigen? Immunotherapy 5(9):907–910
Karan D (2017) Formulation of the bivalent prostate cancer vaccine with surgifoam elicits antigen-specific effector T cells in PSA-transgenic mice. Vaccine 35(43):5794–5798
Karan D, Dubey S, Van Veldhuizen P, Holzbeierlein JM, Tawfik O, Thrasher JB (2011) Dual antigen target-based immunotherapy for prostate cancer eliminates the growth of established tumors in mice. Immunotherapy 3(6):735–746
Kim K, Kang YJ, Park SR, Kim DS, Leee SW, Ko K, Ponndord D, Ko K (2021) Effect of leaf position and days post-infiltration on transient expression of colorectal cancer vaccine candidate proteins GA733-Fc and GA733-FcK in Nicotiana benthamiana plant. PeerJ 9:e10851
Ko K, Koprowski H (2005) Plant biopharming of monoclonal antibodies. Virus Res 111(1):93–100
Kong Z, Wang Y, Ma W (2018) Vaccination in the immunotherapy of glioblastoma. Hum Vaccin Immunother 14(2):255–268
Kristen Davis M, Sarah Wood R, Emily Dill R, Fesko Y, Bitting RL, Harrison MR, George DJ (2015) Optimizing the efficiency and quality of sipuleucel-T delivery in an academic institution. Clin J Oncol Nurs 19(3):297
Levin D, Golding B, Strome SE, Sauna ZE (2015) Fc fusion as a platform technology: potential for modulating immunogenicity. Trends Biotechnol 33(1):27–34
Lim CY, Kim DS, Kang Y, Lee YR, Kim K, Kim MS, Ko K (2022) Immune responses to plant-derived recombinant colorectal cancer glycoprotein EpCAM-FcK fusion protein in mice. Biomol Ther 30(6):546–552
Liu J, Fu M, Wang M, Wan D, Wei Y, Wei X (2022) Cancer vaccines as promising immuno-therapeutics: platforms and current progress. J Hematol Oncol 15:1–28
Lu Z, Lee K-J, Shao Y, Lee J-H, So Y, Choo Y-K, Han YS (2012) Expression of GA733-Fc fusion protein as a vaccine candidate for colorectal cancer in transgenic plants. J Biomed Biotechnol. https://doi.org/10.1155/2012/364240
Ma L, Lukasik E, Gawehns F, Takken FL (2012) The use of agroinfiltration for transient expression of plant resistance and fungal effector proteins in Nicotiana benthamiana leaves. Plant fungal pathogens. Springer, pp 61–74
Madan RA, Gulley JL, Kantoff PW (2013) Demystifying immunotherapy in prostate cancer: understanding current and future treatment strategies. Cancer J (sudbury, Mass) 19(1):50
Naalden D, Verbeek R, Gheysen G (2018) Nicotiana benthamiana as model plant for Meloidogyne graminicola infection. Nematology 20(5):491–499
Narula A, Ellis J, Taliaferro JM, Rissland OS (2019) Coding regions affect mRNA stability in human cells. RNA 25(12):1751–1764
Norkunas K, Harding R, Dale J, Dugdale B (2018) Improving agroinfiltration-based transient gene expression in Nicotiana benthamiana. Plant Methods 14(1):71
Prado GS, Bamogo PKA, de Abreu JAC, Gillet F-X, dos Santos VO, Silva MCM, Brugidou C (2019) Nicotiana benthamiana is a suitable transient system for high-level expression of an active inhibitor of cotton boll weevil α-amylase. BMC Biotechnol 19(1):1–13
Rath T, Baker K, Dumont JA, Peters RT, Jiang H, Qiao S-W, Blumberg RS (2015) Fc-fusion proteins and FcRn: structural insights for longer-lasting and more effective therapeutics. Crit Rev Biotechnol 35(2):235–254
Roos AK, Pavlenko M, Charo J, Egevad L, Pisa P (2005) Induction of PSA-specific CTLs and anti-tumor immunity by a genetic prostate cancer vaccine. Prostate 62(3):217–223
Sahin U, Oehm P, Derhovanessian E et al (2020) An RNA vaccine drives immunity in checkpoint-inhibitor-treated melanoma. Nature 585:107–112
Saif JM, Vadakekolathu J, Rane SS, McDonald D, Ahmad M, Mathieu M, Rees RC (2014) Novel prostate acid phosphatase-based peptide vaccination strategy induces antigen-specific T-cell responses and limits tumour growth in mice. Eur J Immunol 44(4):994–1004
Sainbury F, Thuenemann EC, Lomonossoff GP (2009) pEAQ: versatile expression vectors for easy and quick transient expression of heterologous proteins in plants. Plant Biotechnol J 7:682–693
Schatz A, Mian BM (2019) Current and emerging trends in prostate cancer immunotherapy. Asian J Androl 21(1):6
Shanmugaraj B, Bulaon CJI, Phoolcharoen W (2020) Plant molecular farming: a viable platform for recombinant biopharmaceutical production. Plants 9(7):842
Shin CH, Kang YJ, Soo KH, Kyoo SY, Ko K (2019) Immune response of heterologous recombinant antigenic protein of viral hemorrhagic septicemia virus (VHSV) in mice. Animal Cells Syst 23(2):97–105
Shin CH, Kim K, Kang YJ, Kim D-S, Seo Y-J, Park SR, Kim MK, Lee YK, Kim D-S, Ko K (2022) Effect of IgG Fc-fusion and KDEL-ER retention signal on prostate-specific antigen expression in plant and its immune in mice. Plant Biotechnol Rep 16:729–740
Singh BH, Gulley JL (2014) Therapeutic vaccines as a promising treatment modality against prostate cancer: rationale and recent advances. Ther Adv Vaccines 2(5):137–148
Song I, Lee YK, Kim JW et al (2021) Effect of an endoplasmic reticulum retention signal tagged to human anti-rabies mAb SO57 on its expression in Arabidopsis and plant growth. Mol Cells 44(10):770–779
Thorne H, Willems AJ, Niedermayr E, Hoh IM, Li J, Clouston D, Bolton D (2011) Decreased prostate cancer-specific survival of men with BRCA2 mutations from multiple breast cancer families. Cancer Prev Res 4(7):1002–1010
Tian J-Y, Guo F-J, Zheng G-Y, Ahmad A (2018) Prostate cancer: updates on current strategies for screening, diagnosis and clinical implications of treatment modalities. Carcinogenesis 39(3):307–317
Velonas VM, Woo HH, Remedios CGD, Assinder SJ (2013) Current status of biomarkers for prostate cancer. Int J Mol Sci 14(6):11034–11060
Zhao B, Zhang X, Krummenacher C, Song S, Gao L, Zhang H, Zeng M (2018) Immunization with Fc-based recombinant Epstein-Barr virus gp350 elicits potent neutralizing humoral immune response in a BALB/c mice model. Front Immunol 9:932
Funding
This research was supported by the National Research Foundation of Korea grant [2021R1F1A1063869], Bio & Medical Technology Development Program of the National Research Foundation (NRF) and funded by the Korean government (MSIT) (No. 2019K1A3A1A18116087).
Author information
Authors and Affiliations
Contributions
Conceptualization, SYO, and KK; methodology, SYO, KBK, YJK, HJH, YRK, and PH; validation, MKK, YKL, DSK, and SCM.; formal analysis, SYO; investigation, KBK, and KK; resources, SYO, PH, and KK; data curation, KBK and YJK; writing—original draft preparation, SYO and KBK; writing—review and editing, KK; visualization, KBK and KK; supervision, KK; project administration, KBK and Y.J.K.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Communicated by Baochun Li.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oh, S., Kim, K., Kang, Y.J. et al. Co-transient expression of PSA-Fc and PAP-Fc fusion protein in plant as prostate cancer vaccine candidates and immune responses in mice. Plant Cell Rep 42, 1203–1215 (2023). https://doi.org/10.1007/s00299-023-03028-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-023-03028-3