Abstract
Key message
Botrytis cinerea induced expression of 15 LrWRKY genes; overexpression of LrWRKY39 and LrWRKY41a increased resistance and susceptibility, respectively, to B. cinerea in a manner related to SA and JA signaling.
Abstract
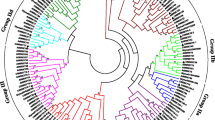
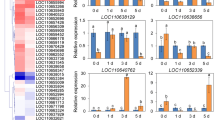
WRKY transcription factors (TFs), a large family, play important roles in coping with biotic stresses. Lilium regale Wilson is a lily species with strong resistance to fungi and viruses; however, functional characterization of LrWRKY TFs remains very limited. Here, a total of 25 LrWRKY members were identified from the L. regale transcriptome, and 15 LrWRKY genes were significantly induced by Botrytis cinerea. Based on their structural features, B. cinerea-responsive LrWRKY genes could be classified into six subgroups (Groups I, IIa–d, and III), and sequence alignment showed that 12 LrWRKY proteins have a well-conserved WRKYGQK domain, while 3 LrWRKYs have a variant sequence (WRKYGKK or WRMYEQK). Quantitative RT-PCR analysis revealed tissue-specific expression of B. cinerea-responsive LrWRKY genes and their expression profiles in response to defense-related hormones salicylic acid (SA), methyl jasmonate (MeJA) and hydrogen peroxide. LrWRKY39 and LrWRKY41a, which encode two LrWRKY TFs with different three-dimensional (3D) models of the WRKY domain, were cloned, and both proteins were targeted to the nucleus. Overexpression of LrWRKY39 and LrWRKY41a in Arabidopsis thaliana increased the resistance and susceptibility to B. cinerea, respectively, compared to the wild type. Similar results were also observed in tobacco and lily (L. longiflorum ‘Snow Queen’) by transient transformation analyses. Their distinct roles may be related to changes in the transcript levels of SA-/JA-responsive genes. Our results provide new insights into B. cinerea-responsive LrWRKY members and the biological functions of two different 3D models of LrWRKY TFs in defense responses to B. cinerea infection.









Similar content being viewed by others
Data availability
All the supporting data can be found as additional file along with this manuscript. The raw sequence data and materials during the current study are available upon reasonable request from corresponding author.
References
Babu MM, Iyer LM, Balaji S, Aravind L (2006) The natural history of the WRKY-GCM1 zinc fingers and the relationship between transcription factors and transposons. Nucleic Acids Res 34(22):6505–6520. https://doi.org/10.1093/nar/gkl888
Birkenbihl RP, Diezel C, Somssich IE (2012) Arabidopsis WRKY33 is a key transcriptional regulator of hormone and metabolic responses towards Botrytis cinerea infection. Plant Physiol 159:266–285
Chen L, Song Y, Li S, Zhang L, Zou C, Yu D (2012) The role of WRKY transcription factors in plant abiotic stresses. BBA-Gene Regal Mech 1819(2):120–128. https://doi.org/10.1016/j.bbagrm.2011.09.002
Chen X, Liu J, Lin G, Wang A, Wang Z, Lu G (2013) Overexpression of AtWRKY28 and AtWRKY75 in Arabidopsis enhances resistance to oxalic acid and Sclerotinia sclerotiorum. Plant Cell Rep 32:1589–1599
Cheng XK, Zhao YX, Jiang QS, Yang J, Zhao WS, Taylor IA, Peng YL, Wang DL, Liu JF (2019) Structural basis of dimerization and dual w-box DNA recognition by rice WRKY domain. Nucleic Acids Res 47(8):4308–4318
Chi Y, Yang Y, Zhou Y, Zhou J, Fan B, Yu JQ, Chen Z (2013) Protein-protein interactions in the regulation of WRKY transcription factors. Mol Plant 6:287–300
Choquer M, Fournier E, Kunz C, Levis C, Pradier JM, Simon A, Viaud M (2007) Botrytis cinerea virulence factors: new insights into a necro-trophic and polyphageous pathogen. FEMS Microbiol Lett 277:1–10
Chujo T, Miyamoto K, Shimogawa T, Shimizu T, Otake Y, Yokotani N, Nishizawa Y, Shibuya N, Nojiri H, Yamane H, Minami E, Okada K (2013) OsWRKY28, a PAMP-responsive transrepressor, negatively regulates innate immune responses in rice against rice blast fungus. Plant Mol Biol 82(1–2):23–37. https://doi.org/10.1007/s11103-013-0032-5
Cui Q, Yan X, Gao X, Zhang DM, He HB, Jia GX (2018) Analysis of WRKY transcription factors and characterization of two Botrytis cinerea-responsive LrWRKY genes from Lilium regale. Plant Physiol Bioch 127:525–536. https://doi.org/10.1016/j.plaphy.2018.04.027
Dean R, Van KJA, Pretorius ZA, Hammond-Kosack KE, Di Pietro A, Spanu PD, Rudd JJ, Dickman M, Kahmann R, Ellis J, Foster GD (2012) The Top 10 fungal pathogens in molecular plant pathology. Mol Plant Pathol 13:414–430
Deng B, Wang WJ, Ruan CQ, Deng LL, Yao SX, Zeng KF (2020) Involvement of CsWRKY70 in salicylic acid-induced citrus fruit resistance against Penicillium digitatum. Hortic Res 7:157. https://doi.org/10.1038/s41438-020-00377-y
Dong JX, Chen CH, Chen ZX (2003) Expression profiles of the Arabidopsis WRKY gene superfamily during plant defense response. Plant Mol Biol 51:21–37
Duan MR, Nan J, Liang YH, Mao P, Lu L, Li L, Wei C, Lai L, Li Y, Su XD (2007) DNA binding mechanism revealed by high resolution crystal structure of Arabidopsis thaliana WRKY1 protein. Nucleic Acids Res 35:1145–1154
Eulgem T, Rushton PJ, Schmelzer E, Hahlbrock K, Somssich IE (1999) Early nuclear events in plant defence signalling: rapid gene activation by WRKY transcription factors. EMBO J 18:4689–4699
Eulgem T, Rushton PJ, Robatzek S, Somssich IE (2000) The WRKY superfamily of plant transcription factors. Trends Plant Sci 5(5):199–206
Ferrari S, Plotnikova JM, Lorenzo GD, Ausubel FM (2003) Arabidopsis local resistance to Botrytis cinerea involves salicylic acid and camelexin and requires EDS4 and PAD2 but not SID2, EDS5 or PAD4. Plant J 35:193–205
Fu YY, Zhu YY, Yang W, Xu WJ, Li Q, Chen M, Yang LP (2020) Isolation and functional identification of a Botrytis cinerea-responsive caffeoyl-CoA O-methyltransferase gene from Lilium regale Wilson. Plant Physiol Bioch 157:379–389
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227. https://doi.org/10.1146/annurev.phyto.43.040204.135923
Han Q, Chen R, Yang Y, Cui XM, Ge F, Chen CY, Liu DQ (2016) A glutathione S -transferase gene from Lilium regale Wilson confers transgenic tobacco resistance to Fusarium oxysporum. Sci Hortic 198:370–378
He H, Liu DQ, Zhang NN, Zheng W, Han Q, Ji B, Ge F, Chen CY (2014) The PR10 gene family is highly expressed in Lilium regale Wilson during Fusarium oxysporum f. sp. lilii infection. Genes Genom 36:497–507
Higashi K, Ishiga Y, Inagaki Y, Toyoda K, Shiraishi T, Ichinose Y (2008) Modulation of defense signal transduction by flagellin-induced WRKY41 transcription factor in Arabidopsis thaliana. Mol Genet Genomics 279(3):303–312
Hou PF, Chen CY (2003) Early stages of infection of lily leaves by Botrytis elliptica and B. cinerea. Plant Pathol Bull 12:103–108
Hu J, Barlet X, Deslandes L, Hirsch J, Feng DX, Somssich I, Marco Y (2008) Transcriptional responses of Arabidopsis thaliana during wilt disease caused by the soil-borne phytopathogenic bacterium. Ralstonia solanacearum. PLoS One 3:e2589
Huang S, Gao Y, Liu J, Peng X, Niu X, Fei Z, Cao S, Liu Y (2012) Genome-wide analysis of WRKY transcription factors in Solanum lycopersicum. Mol Genet Genomics 287:495–513
Ishiguro S, Nakamura K (1994) Characterization of a cDNA encoding a novel DNA-binding protein, SPF1, that recognizes SP8 sequences in the 5’upstream regions of genes coding for sporamin and beta-amylase from sweet potato. Mol Gen Genet 244(6):563–571. https://doi.org/10.1007/BF00282746
Jiang Y, Duan Y, Jia Y, Ye S, Zhu J, Zhang F, Lu W, Di F, Luo K (2014) Genome wide identification and characterization of the Populus WRKY transcription factor family and analysis of their expression in response to biotic and abiotic stresses. J Exp Bot 65(22):6629–6644
Jirage D, Tootle TL, Reuber TL, Frost LN, Feys BJ, Parker JE, Ausubel FM, Glazebrook J (1999) Arabidopsis thaliana PAD4 encodes a lipase-like gene that is important for salicylic acid signaling. Proc Natl Acad Sci USA 96:13583–13588
Kalde M, Barth M, Somssich IE, Lippok B (2003) Members of the Arabidopsis WRKY group III transcription factors are part of different plant defense signaling pathways. Mol Plant Microbe Interact 16(4):295–305
Kim KC, Fan B, Chen Z (2006) Pathogen-induced Arabidopsis WRKY7 is a transcriptional repressor and enhances plant susceptibility to Pseudomonas syringae. Plant Physiol 142:1180–1192
Lai Z, Vinod KM, Zheng Z, Fan B, Chen Z (2008) Roles of Arabidopsis WRKY3 and WRKY4 transcription factors in plant responses to pathogens. BMC Plant Biol 8:68
Lai Z, Li Y, Wang F, Cheng Y, Fan B, Yu JQ, Chen Z (2011) Arabidopsis sigma factor binding proteins are activators of the WRKY33 transcription factor in plant defense. Plant Cell 23:3824–3841
Levée V, Major I, Levasseur C, Tremblay L, Mackay J, Séguin A (2009) Expression profiling and functional analysis of Populus WRKY23 reveals a regulatory role in defense. New Phytol 184(1):48–70
Li J, Brader G, Palva ET (2004) The WRKY70 transcription factor: a node of convergence for jasmonate-mediated and salicylate-mediated signals in plant defense. Plant Cell 16:319–331
Li J, Brader G, Kariola T, Palva ET (2006) WRKY70 modulates the selection of signaling pathways in plant defense. Plant J 46:477–491
Li S, Zhou X, Chen L, Huang W, Yu D (2010) Functional characterization of Arabidopsis thaliana WRKY39 in heat stress. Mol Cells 29(5):475–483
Li HL, Liu DQ, He H, Zhang NN, Ge F, Chen CY (2014) Molecular cloning of a 14-3-3 protein gene from Lilium regale Wilson and overexpression of this gene in tobacco increased resistance to pathognic fungi. Sci Hortic 168:9–16
Li P, Song A, Gao C, Jiang J, Chen S, Fang W, Zhang F, Chen F (2015) The over-expression of a chrysanthemum WRKY transcription factor enhances aphid resistance. Plant Physiol Bioch 95:26–34
Li J, Xiong YC, Li Y, Ye SQ, Yin Q, Gao SQ, Yang D, Yang M, Palva T, Deng XB (2019) Comprehensive analysis and functional studies of WRKY transcription factors in Nelumbo nucifera. Int J Mol Sci 20(5006):1–19
Lim JH, Rhee HK, Kim YJ, Lim KB, van Tuyl JM (2003) Resistance to Fusarium Oxysporum F.Sp Lilii in Lilium. Acta. Horticul. Sinica. 620:311–318
Liu Q, Wei C, Zhang MF, Jia GX (2016) Evaluation of putative reference genes for quantitative real-time PCR normalization in Lilium regale during development and under stress. Peer J 4:e1837
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Malhat FM, Haggag MN, Loutfy NM, Osman MA, Ahmed MT (2015) Residues of organochlorine and synthetic pyrethroid pesticides in honey, an indicator of ambient environment, a pilot study. Chemosphere 120:457–461
Mangelsen E, Kilian J, Berendzen KW, Kolukisaoglu UH, Harter K, Jansson C, Wanke D (2008) Phylogenetic and comparative gene expression analysis of barley (Hordeum vulgare) WRKY transcription factor family reveals putatively retained functions between monocots and dicots. BMC Genom 9:194
Mao P, Duan M, Wei C, Li Y (2007) WRKY62 transcription factor acts downstream of cytosolic NPR1 and negatively regulates jasmonate-responsive gene expression. Plant Cell Physiol 48:833–842
Melchers LS, Stuiver MH (2000) Novel genes for disease-resistance breeding. Curr Opin Plant Biol 3:147–152
Murray SL, Ingle RA, Petersen LN, Denby KJ (2007) Basal resistance against Pseudomonas syringae in Arabidopsis involves WRKY53 and a protein with homology to a nematode resistance protein. Mol Plant Microbe Interact 20:1431–1438
Mzid R, Marchive C, Blancard D, Deluc L, Barrieu F, Corio-Costet MF, Drira N, Hamdi S, Lauvergeat V (2007) Overexpression of VvWRKY2 in tobacco enhances broad resistance to necrotrophic fungal pathogens. Physiol Plantarum 131:434–447
Nandi A, Moeder W, Kachroo P, Klessig DF, Shah J (2005) Arabidopsis ssi2-conferred susceptibility to Botrytis cinerea is dependent on EDS5 and PAD4. MPMI 18(4):363–370
Oliveira BR, Penetra A, Cardoso VV, Benoliel MJ, Barreto Crespo MT, Samson RA, Pereira VJ (2015) Biodegradation of pesticides using fungi species found in the aquatic environment. Environ Sci Pollut R 22:11781–11791
Pandey SP, Somssich IE (2009) The role of WRKY transcription factors in plant immunity. Plant Physiol 150:1648–1655
Park CY, Lee JH, Yoo JH, Moon BC, Choi MS, Kang YH, Lee SM, Kim HS, Kang KY, Chung WS, Lim CO, Cho MJ (2005) WRKY group IId transcription factors interact with calmodulin. FEBS Lett 579(6):1545–1550
Phukan UJ, Jeena GS, Shukla RK (2016) WRKY transcription factors: molecular regulation and stress responses in plants. Front Plant Sci 7:760. https://doi.org/10.3389/fpls.2016.00760
Riechmann JL, Ratcliffe OJ (2000) A genomic perspective on plant transcription factors. Curr Opin Plant Biol 3(5):423–434
Ross CA, Liu Y, Shen QJ (2007) The WRKY gene family in rice (Oryza sativa). J Integr Plant Biol 49:827–842
Rushton PJ, Somssich IE, Ringler P, Shen QJ (2010) WRKY transcription factors. Trends Plant Sci 15:247–258
Shen QH, Saijo Y, Mauch S, Biskup C, Bieri S, Keller B, Seki H, Ulker B, Somssich IE, Schulze-Lefert P (2007) Nuclear activity of MLA immune receptors links isolate-specific and basal disease-resistance responses. Science 315:1098–1103
Shi WN, Hao LL, Li J, Liu DD, Guo XQ, Han L (2014) The Gossypium hirsutum WRKY gene GhWRKY39-1 promotes pathogen infection defense responses and mediates salt stress tolerance in transgenic Nicotiana benthamiana. Plant Cell Rep 33:483–498
Shim JS, Jung C, Lee S, Min K, Lee YW, Choi Y, Lee JS, Song JT, Kim JK, Choi YD (2013) AtMYB44 regulates WRKY70 expression and modulates antagonistic interaction between salicylic acid and jasmonic acid signaling. Plant J 73:483–495
Sultan S, Ali MA, Atif RM, Azeem F, Nadeem H, Siddique MH, Filiz E, Hussain K, Abbas A (2016) Genome wide analysis of stress responsive WRKY transcription factors in Arabidopsis thaliana. Turk J Agric Food Sci Technol 4:279–290
Sun D, Zhang X, Li S, Jiang CZ, Zhang Y, Niu L (2016) LrABCF1, a GCN-type ATP-binding cassette transporter from Lilium regale, is involved in defense responses against viral and fungal pathogens. Planta 244:1185–1199
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739. https://doi.org/10.1093/molbev/msr12
Tsuda K, Somssich IE (2015) Transcriptional networks in plant immunity. New Phytol 206:932–947. https://doi.org/10.1111/nph.13286
Tsuda K, Sato M, Stoddard T, Glazebrook J, Katagiri F (2009) Network properties of robust immunity in plants. PLoS Genet 5:e1000772
van Verk MC, Pappaioannou D, Neeleman L, Bol JF, Linthorst HJM (2008) A novel WRKY transcription factor is required for induction of PR-1a gene expression by salicylic scid and bacterial elicitors. Plant Physiol 146:1983–1995
Wang M, Thomas N, Jin H (2017a) Cross-kingdom RNA trafficking and environmental RNAi for powerful innovative pre- and post-harvest plant protection. Curr Opin Plant Biol 38:133–141
Wang X, Guo R, Tu M, Wang D, Guo C, Wan R, Li Z, Wang X (2017b) Ectopic expression of the wild grape WRKY transcription factor VqWRKY52 in Arabidopsis thaliana enhances resistance to the biotrophic pathogen Powdery Mildew but not to the necrotrophic pathogen Botrytis cinerea. Front Plant Sci 8:97. https://doi.org/10.3389/fpls.2017.00097
Wang P, Yue C, Chen D, Zheng Y, Zhang Q, Yang J, Ye N (2019) Genome-wide identification of WRKY family genes and their response to abiotic stresses in tea plant (Camellia sinensis). Genes Genom 41:17–33
Wang T, Huang S, Zhang A, Guo P, Liu Y, Xu C, Cong W, Liu B, Xu Z (2020) JMJ17-WRKY40 and HY5-ABI5 modules regulate the expression of ABA-responsive genes in Arabidopsis. New Phytol 230(2):567–584. https://doi.org/10.1111/nph.17177
Xu X, Chen C, Fan B, Chen Z (2006) Physical and functional interactions between pathogen-induced Arabidopsis WRKY18, WRKY40, and WRKY60 transcription factors. Plant Cell 18:1310–1326
Yamasaki K, Kigawa T, Inoue M, Tateno M, Yamasaki T, Yabuki T, Aoki M, Seki E, Matsuda T, Tomo Y et al (2005) Solution structure of an Arabidopsis WRKY DNA binding domain. Plant Cell 17:944–956
Zhang X, Henriques R, Lin SS, Niu QW, Chua NH (2006) Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat Protoc 1(2):641–646. https://doi.org/10.1038/nprot.2006.97
Zhang NN, Liu DQ, Zheng W, He H, Ji B, Han Q, Ge F, Chen CY (2014) A bZIP transcription factor, LrbZIP1, is involved in Lilium regale Wilson defense responses against Fusarium oxysporum f. sp. lilii. Genes Genom 36:789–798
Zheng Z, Qamar SA, Chen Z, Mengiste T (2006) Arabidopsis WRKY33 transcription factor is required for resistance to necrotrophic fungal pathogens. Plant J 48:592–605
Zhou QY, Tian AG, Zou HF, Xie ZM, Lei G, Huang J, Wang CM, Wang HW, Zhang JS, Chen SY (2008) Soybean WRKY-type transcription factor genes, GmWRKY13, GmWRKY21, and GmWRKY54, confer differential tolerance to abiotic stresses in transgenic Arabidopsis plants. Plant Biotechnol J 64:86–503
Zhou T, Wang J, Wang LL, Wang BK, Hu JH, Lan HY, Yu QH (2020) Cloning and prokaryotic expression analysis of a transcription factor gene SlWRKY16 in tomato. Acta Horticul Sinica 47(7):1312–1322
Acknowledgements
The authors thank Dr. Biao Lai in Yangtze Normal University for supplying the plant binary vector pBI121 and also thank Dr. Jian Gao in Yangtze Normal University for the assistance during bioinformatics analysis.
Funding
This work was financially supported by Natural Science Research Project of Chongqing Science and Technology Bureau (cstc2019jcyj-msxmX0014), Science and Technology Research Program of Chongqing Municipal Education Commission (KJQN202001418), Chongqing key Laboratory for Conservation and Utilization of Characteristic Plant Resources in Wuling Mountain Area (TEZWKFKT202005).
Author information
Authors and Affiliations
Contributions
YF, QL and LY: conceived and designed the experiments. YF, JL, HW, YZ and CL: conducted the experiments. YF, SJ and WX: analyzed the data. YF: wrote the first manuscript, LY and QL: supervised and complemented the writing. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Howard S. Judelson.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fu, Y., Li, J., Wu, H. et al. Analyses of Botrytis cinerea-responsive LrWRKY genes from Lilium regale reveal distinct roles of two LrWRKY transcription factors in mediating responses to B. cinerea. Plant Cell Rep 41, 995–1012 (2022). https://doi.org/10.1007/s00299-022-02833-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-022-02833-6