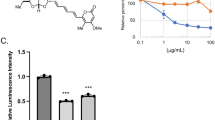
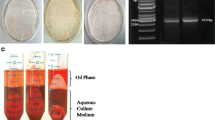
Abstract
Key message
Our paper describes the potential roles of lipid droplets of Taxus media cell suspension in the biosynthesis and secretion of paclitaxel and, therefore, highlights their involvement in improving its production.
Abstract
Paclitaxel (PTX) is a highly potent anticancer drug that is mainly produced using Taxus sp. cell suspension cultures. The main purpose of the current study is to characterize cellular LDs from T. media cell suspension with a particular focus on the biological connection of their associated proteins, the caleosins (CLOs), with the biosynthesis and secretion of PTX. A pure LD fraction obtained from T. media cells and characterized in terms of their proteome. Interestingly, the cellular LD in T. media sequester the PTX. This was confirmed in vitro, where about 96% of PTX (C0PTX,aq [mg L−1]) in the aqueous solution was partitioned into the isolated LDs. Furthermore, silencing of CLO-encoding genes in the T. media cells led to a net decrease in the number and size of LDs. This coincided with a significant reduction in expression levels of TXS, DBAT and DBTNBT, key genes in the PTX biosynthesis pathway. Subsequently, the biosynthesis of PTX was declined in cell culture. In contrast, treatment of cells with 13-hydroperoxide C18:3, a substrate of the peroxygenase activity, induced the expression of CLOs, and, therefore, the accumulation of cellular LDs in the T. media cells cultures, thus increasing the PTX secretion. The accumulation of stable LDs is critically important for effective secretion of PTX. This is modulated by the expression of caleosins, a class of LD-associated proteins with a dual role conferring the structural stability of LDs as well as regulating lipidic bioactive metabolites via their enzymatic activity, thus enhancing the biosynthesis of PTX.








Similar content being viewed by others
Abbreviations
- PTX:
-
Paclitaxel
- LDs:
-
Lipid droplets
- CLO:
-
Caleosin
- OLEO:
-
Oleosin
- TCDD:
-
2,3,7,8-Polychlorinateddibenzo-p-dioxins
- TAG:
-
Triacylglycerol
- LOX:
-
Lipoxygenase
- PXG:
-
Peroxygenase
- JA:
-
Jasmonic acid
- MeJA:
-
Methyl jasmonic acid
- 13-HPOT:
-
13-Hydroperoxide linolenic acid
- 13-LOX:
-
13-Lipoxygenase
- AOS:
-
Allene oxide synthase
- PXG:
-
Peroxygenase
- DES:
-
Divinyl ether synthase
- HPL:
-
Hydroperoxide lyase
References
Almagro L, Belchi-Navarro S, Martinez-Marquez A, Bru R, Pedreno MA (2015) Enhanced extracellular production of trans-resveratrol in Vitis vinifera suspension cultured cells by using cyclodextrins and coronatine. Plant Physiol Biochem 97:361–367
Bachmann A, Hause B, Maucher H, Garbe E, Voros K, Weichert H, Wasternack C, Feussner I (2002) Jasmonate-induced lipid peroxidation in barley leaves initiated by distinct 13-LOX forms of chloroplasts. Biol Chem 383:1645–1657
Bate NJ, Rothstein SJ (1998) C6-volatiles derived from the lipoxygenase pathway induce a subset of defense-related genes. Plant J 16:561–569
Belchi-Navarro S, Almagro L, Lijavetzky D, Bru R, Pedreno MA (2012) Enhanced extracellular production of trans-resveratrol in Vitis vinifera suspension cultured cells by using cyclodextrins and methyljasmonate. Plant Cell Rep 31:81–89
Besagni C, Kessler F (2013) A mechanism implicating plastoglobules in thylakoid disassembly during senescence and nitrogen starvation. Planta 237:463–470
Bissery MC, Guenard D, Gueritte-Voegelein F, Lavelle F (1991) Experimental antitumor activity of taxotere (RP 56976, NSC 628503), a taxol analogue. Cancer Res 51:4845–4852
Bissett D, Kaye SB (1993) Taxol and taxotere—current status and future prospects. Eur J Cancer 29A:1228–1231
Blee E (2002) Impact of phyto-oxylipins in plant defense. Trends Plant Sci 7:315–322
Blee E, Durst F (1987) Hydroperoxide-dependent sulfoxidation catalyzed by soybean microsomes. Arch Biochem Biophys 254:43–52
Blee E, Boachon B, Burcklen M, Le Guedard M, Hanano A, Heintz D, Ehlting J, Herrfurth C, Feussner I, Bessoule JJ (2014) The reductase activity of the Arabidopsis caleosin RESPONSIVE TO DESSICATION20 mediates gibberellin-dependent flowering time, abscisic acid sensitivity, and tolerance to oxidative stress. Plant Physiol 166:109–124
Bonfill M, Palazón J, Cusidó RM, Joly S, Morales C, Piñol MT (2003) Influence of elicitors on taxane production and 3-hydroxy-3-methylglutaryl coenzymeA reductase activity in Taxus media cells. Plant Physiol Biochem 41:91–96
Boucher J, Cengelli F, Trumbic D, Marison IW (2008) Sorption of hydrophobic organic compounds (HOC) in rapeseed oil bodies. Chemosphere 70:1452–1458
Boven E, Venema-Gaberscek E, Erkelens CA, Bissery MC, Pinedo HM (1993) Antitumor activity of taxotere (RP 56976, NSC 628503), a new taxol analog, in experimental ovarian cancer. Ann Oncol 4:321–324
Braakhuis BJ, Hill BT, Dietel M, Kelland LR, Aapro MS, Zoli W, Lelieveld P (1994) In vitro antiproliferative activity of docetaxel (Taxotere), paclitaxel (Taxol) and cisplatin against human tumour and normal bone marrow cells. Anticancer Res 14:205–208
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cai Z, Kastell A, Knorr D, Smetanska I (2012) Exudation: an expanding technique for continuous production and release of secondary metabolites from plant cell suspension and hairy root cultures. Plant Cell Rep 31:461–477
Chang W, Zhan GM, Zheng S, Li Y, Li X, Li W, Li G, Lin Z, Xie Z, Zhao Z, Lou H (2015) Trapping toxins within lipid droplets is a resistance mechanism in fungi. Nat Sci Rep 5:15133
Chapman KD, Dyer JM, Mullen RT (2012) Biogenesis and functions of lipid droplets in plants: thematic review series: lipid droplet synthesis and metabolism: from yeast to man. J Lipid Res 53:215–226
Chapman KD, Aziz M, Dyer JM, Mullen RT (2019) Mechanisms of lipid droplet biogenesis. Biochem J 476:1929–1942
Charuchinda P, Waditee-Sirisattha R, Kageyama H, Yamada D, Sirisattha S, Tanaka Y, Mahakhant A, Takabe T (2015) Caleosin from Chlorella vulgaris TISTR 8580 is salt-induced and heme-containing protein. Biosci Biotechnol Biochem 79:1119–1124
Chen MC, Chyan CL, Lee TT, Huang SH, Tzen JT (2004) Constitution of stable artificial oil bodies with triacylglycerol, phospholipid, and caleosin. J Agric Food Chem 52:3982–3987
Chiang CJ, Lin LJ, Chen CJ (2011) Caleosin-based nanoscale oil bodies for targeted delivery of hydrophobic anticancer drugs. J Nanopart Res 13:7127–7137
Chiang CJ, Lin SC, Lin LJ, Chen CJ, Chao YP (2012) Caleosin-assembled oil bodies as a potential delivery nanocarrier. Appl Microbiol Biotechnol 93:1905–1915
Croteau R, Ketchum RE, Long RM, Kaspera R, Wildung MR (2006) Taxol biosynthesis and molecular genetics. Phytochem Rev 5:75–97
Crown J, O’Leary M, Ooi WS (2004) Docetaxel and paclitaxel in the treatment of breast cancer: a review of clinical experience. Oncologist 9(Suppl 2):24–32
Cusido RM, Palazon J, Bonfill M, Navia-Osorio A, Morales C, Pinol MT (2002) Improved paclitaxel and baccatin III production in suspension cultures of Taxus media. Biotechnol Prog 18:418–423
Cusido RM, Onrubia M, Sabater-Jara AB, Moyano E, Bonfill M, Goossens A, Angeles Pedreno M, Palazon J (2014) A rational approach to improving the biotechnological production of taxanes in plant cell cultures of Taxus spp. Biotechnol Adv 32:1157–1167
De Vries J, Ischebeck T (2020) Ties between stress and lipid droplets pre-date seeds. Trends Plant Sci 25:1203–1214
Deboever E, Deleu M, Mongrand S, Lins L, Fauconnier ML (2020) Plant-pathogen interactions: underestimated roles of phyto-oxylipins. Trends Plant Sci 25:22–34
Esnay N, Dyer MJ, Mullen RT, Chapman KD (2020) Lipid droplet-peroxisome connections in plants. Contact 3:1–14
Exposito O, Syklowska-Baranek K, Moyano E, Onrubia M, Bonfill M, Palazon J, Cusido RM (2010) Metabolic responses of Taxus media transformed cell cultures to the addition of methyl jasmonate. Biotechnol Prog 26:1145–1153
Farag MA, Westphal H, Eissa TF, Wessjohann LA, Meyer A (2017) Effect of oxylipins, terpenoid precursors and wounding on soft corals’ secondary metabolism as analyzed via UPLC/MS and chemometrics. Molecules. https://doi.org/10.3390/molecules22122195
Fernandez-Santos R, Izquierdo Y, Lopez A, Muniz L, Martinez M, Cascon T, Hamberg M, Castresana C (2020a) Protein profiles of lipid droplets during the hypersensitive defense response of Arabidopsis against Pseudomonas infection. Plant Cell Physiol 61:1144–1157
Fett-Neto AG, Melanson SJ, Nicholson SA, Pennington JJ, Dicosmo F (1994a) Improved taxol yield by aromatic carboxylic acid and amino acid feeding to cell cultures of Taxus cuspidata. Biotechnol Bioeng 44:967–971
Fett-Neto AG, Zhang WY, Dicosmo F (1994b) Kinetics of taxol production, growth, and nutrient uptake in cell suspensions of Taxus cuspidata. Biotechnol Bioeng 44:205–210
Feussner I, Wasternack C (2002) The lipoxygenase pathway. Annu Rev Plant Biol 53:275–297
Froissard M, D’Andrea S, Boulard C, Chardot T (2009) Heterologous expression of AtClo1, a plant oil body protein, induces lipid accumulation in yeast. FEMS Yeast Res 9:428–438
Gao Q, Goodman JM (2015) The lipid droplet-a well-connected organelle. Front Cell Dev Biol 3:49
Geem KR, Kim DH, Lee DW, Kwon Y, Lee J, Kim JH, Hwang I (2019) Jasmonic acid-inducible TSA1 facilitates ER body formation. Plant J 97:267–280
Goossens A, Farmer EE (2019) Regulatory Oxylipins Anno 2019: Jasmonates Galore in the Plant Oxylipin Research Community. Plant Cell Physiol 60:2609–2612
Grechkin AN (2002) Hydroperoxide lyase and divinyl ether synthase. Prostaglandins Other Lipid Mediat 68–69:457–470
Griffiths G (2015) Biosynthesis and analysis of plant oxylipins. Free Radic Res 49:565–582
Guenard D, Gueritte-Voegelein F, Dubois J, Potier P (1993) Structure-activity relationships of taxol and taxotere analogues. J Natl Cancer Inst Monogr 15:79–82
Gueritte-Voegelein F, Guenard D, Potier P (1992) Anticancer substances of vegetable origin. spindle poisons: vincaleukoblastine, leurocristine and navelbine; taxol and taxotere. C R Seances Soc Biol Fil 186:433–440
Gueritte-Voegelein F, Guenard D, Dubois J, Wahl A, Potier P (1994) Chemical and biological studies on taxol (paclitaxel) and taxotere (docetaxel), new antineoplastic agents. J Pharm Belg 49:193–205
Gwak Y, Hwang YS, Wang B, Kim M, Jeong J, Lee CG, Hu Q, Han D, Jin E (2014) Comparative analyses of lipidomes and transcriptomes reveal a concerted action of multiple defensive systems against photooxidative stress in Haematococcus pluvialis. J Exp Bot 65:4317–4334
Hanano A, Burcklen M, Flenet M, Ivancich A, Louwagie M, Garin J, Blee E (2006) Plant seed peroxygenase is an original heme-oxygenase with an EF-hand calcium binding motif. J Biol Chem 281:33140–33151
Hanano A, Almousally I, Shaban M, Blee E (2015a) A caleosin-like protein with peroxygenase activity mediates Aspergillus flavus development, aflatoxin accumulation, and seed infection. Appl Environ Microbiol 81:6129–6144
Hanano A, Almousally I, Shaban M, Moursel N, Shahadeh A, Alhajji E (2015b) Differential tissue accumulation of 2,3,7,8-tetrachlorinated dibenzo-p-dioxin in Arabidopsis thaliana affects plant chronology, lipid metabolism and seed yield. BMC Plant Biol 15:193
Hanano A, Bessoule JJ, Heitz T, Blee E (2015c) Involvement of the caleosin/peroxygenase RD20 in the control of cell death during Arabidopsis responses to pathogens. Plant Signal Behav 10:e991574
Hanano A, Almousally I, Shaban M, Rahman F, Blee E, Murphy DJ (2016a) Biochemical, transcriptional, and bioinformatic analysis of lipid droplets from seeds of date palm (Phoenix dactylifera L.) and their use as potent sequestration agents against the toxic pollutant, 2,3,7,8-tetrachlorinated dibenzo-p-dioxin. Front Plant Sci 7:836
Hanano A, Almousally I, Shaban M, Rahman F, Hassan M, Murphy DJ (2016b) Specific caleosin/peroxygenase and lipoxygenase activities are tissue-differentially expressed in date palm (Phoenix dactylifera L.) seedlings and are further induced following exposure to the toxin 2,3,7,8-tetrachlorodibenzo-p-dioxin. Front Plant Sci 7:2025
Hanano A, Alkara M, Almousally I, Shaban M, Rahman F, Hassan M, Murphy DJ (2018a) The peroxygenase activity of the Aspergillus flavus caleosin, AfPXG, modulates the biosynthesis of aflatoxins and their trafficking and extracellular secretion via lipid droplets. Front Microbiol 9:158
Hanano A, Almousally I, Shaban M, Murphy DJ (2018b) Arabidopsis plants exposed to dioxin result in a WRINKLED seed phenotype due to 20S proteasomal degradation of WRI1. J Exp Bot 69:1781–1794
Hanano A, Shaban M, Almousally I, Murphy DJ (2018c) Identification of a dioxin-responsive oxylipin signature in roots of date palm: involvement of a 9-hydroperoxide fatty acid reductase, caleosin/peroxygenase PdPXG2. Sci Rep 8:13181
Hanano A, Almousally I, Shaban M (2019) Exposure of Aspergillus flavus NRRL 3357 to the environmental toxin, 2,3,7,8-tetrachlorinated dibenzo-p-dioxin, results in a hyper aflatoxicogenic phenotype: a possible role for caleosin/peroxygenase (AfPXG). Front Microbiol 10:2338
Hanano A, Shaban M, Murphy DJ (2021) Functional involvement of caleosin/peroxygenase PdPXG4 in the accumulation of date palm leaf lipid droplets after exposure to dioxins. Environ Pollut 281:116966
Hezari M, Ketchum RE, Gibson DM, Croteau R (1997) Taxol production and taxadiene synthase activity in Taxus canadensis cell suspension cultures. Arch Biochem Biophys 337:185–190
Huang AHC (2018) Plant lipid droplets and their associated proteins: potential for rapid advances. Plant Physiol 176:1894–1918
Hudak KA, Thompson JE (1996) Flotation of lipid-protein particles containing triacylglycerol and phospholipid from the cytosol of carnation petals. Physiol Plant 98:810–818
Hudak KA, Thompson JE (1997) Subcellular localization of secondary lipid metabolites including fragrance volatiles in carnation petals. Plant Physiol 114:705–713
Jamme F, Vindigni JD, Mechin V, Cherifi T, Chardot T, Froissard M (2013) Single cell synchrotron FT-IR microspectroscopy reveals a link between neutral lipid and storage carbohydrate fluxes in S. cerevisiae. PLoS ONE 8:e74421
Jang G, Yoon Y, Choi YD (2020b) Crosstalk with jasmonic acid integrates multiple responses in plant development. Int J Mol Sci. https://doi.org/10.3390/ijms21010305
Jennewein S, Croteau R (2001) Taxol: biosynthesis, molecular genetics, and biotechnological applications. Appl Microbiol Biotechnol 57:13–19
Jolivet P, Roux E, D’Andrea S, Davanture M, Negroni L, Zivy M, Chardot T (2004) Protein composition of oil bodies in Arabidopsis thaliana ecotype WS. Plant Physiol Biochem 42:501–509
Kawahara M, Furuse K (1992) Recent results of anticancer drugs acting on microtubules—navelbine, taxol and taxotere for the treatment of lung cancer. Gan to Kagaku Ryoho 19:2150–2156
Kelland LR, Abel G (1992) Comparative in vitro cytotoxicity of taxol and taxotere against cisplatin-sensitive and -resistant human ovarian carcinoma cell lines. Cancer Chemother Pharmacol 30:444–450
Koda Y (1992) The role of jasmonic acid and related compounds in the regulation of plant development. Int Rev Cytol 135:155–199
Kory N, Farese RV Jr, Walther TC (2016) Targeting fat: mechanisms of protein localization to lipid droplets. Trends Cell Biol 26:535–546
Laudert D, Weiler EW (1998) Allene oxide synthase: a major control point in Arabidopsis thaliana octadecanoid signalling. Plant J 15:675–684
Lomova MV, Sukhorukov GB, Antipina MN (2010) Antioxidant coating of micronsize droplets for prevention of lipid peroxidation in oil-in-water emulsion. ACS Appl Mater Interfaces 2:3669–3676
Luo J, Xia W, Cao P, Xiao Z, Zhang Y, Liu M, Zhan C, Wang N (2019) Integrated transcriptome analysis reveals plant hormones jasmonic acid and salicylic acid coordinate growth and defense responses upon fungal infection in poplar. Biomolecules 9:12
Ma P, Mumper JR (2013) Paclitaxel nano-delivery systems: a comprehensive review. J Nanomed Nanotechnol 4:1000164
Matsuno R, Adachi S (1993) Lipid encapsulation technology—techniques and applications to food. Trends Food Sci Technol 4:256–261
Murphy DJ (1993) Structure, function and biogenesis of storage lipid bodies and oleosins in plants. Prog Lipid Res 32:247–280
Murphy DJ (2001) The biogenesis and functions of lipid bodies in animals, plants and microorganisms. Prog Lipid Res 40:325–438
Murphy DJ (2012) The dynamic roles of intracellular lipid droplets: from archaea to mammals. Protoplasma 249:541–585
Murphy DJ, Hernendez-Pinzon I, Patel K, Hope RG, McLauchlan J (2000) New insights into the mechanisms of lipid-body biogenesis in plants and other organisms. Front Microbiol 28:710–711
Naested H, Frandsen GI, Jauh GY, Hernandez-Pinzon I, Nielsen HB, Murphy DJ, Rogers JC, Mundy J (2000) Caleosins: Ca2+-binding proteins associated with lipid bodies. Plant Mol Biol 44:463–476
Navia-Osorio A, Garden H, Cusido RM, Palazon J, Alfermann AW, Pinol MT (2002) Production of paclitaxel and baccatin III in a 20-L airlift bioreactor by a cell suspension of Taxus wallichiana. Planta Med 68:336–340
Nguyen LN, Nosanchuk JD (2011) Lipid droplet formation protects against gluco/lipotoxicity in Candida parapsilosis: an essential role of fatty acid desaturase Ole1. Cell Cycle 10:3159–3167
Nixon M, Chan SHP (1997) A simple and sensitive colorimetric method for the determination of long-chain free fatty acids in subcellular organelles. Anal Biochem 97:403–409
Onrubia M, Cusido RM, Ramirez K, Hernandez-Vazquez L, Moyano E, Bonfill M, Palazon J (2013a) Bioprocessing of plant in vitro systems for the mass production of pharmaceutically important metabolites: paclitaxel and its derivatives. Curr Med Chem 20:880–891
Onrubia M, Moyano E, Bonfill M, Cusido RM, Goossens A, Palazon J (2013b) Coronatine, a more powerful elicitor for inducing taxane biosynthesis in Taxus media cell cultures than methyl jasmonate. J Plant Physiol 170:211–219
Palazon J, Cusido RM, Bonfill M, Morales C, Pinol MT (2003) Inhibition of paclitaxel and baccatin III accumulation by mevinolin and fosmidomycin in suspension cultures of Taxus baccata. J Biotechnol 101:157–163
Partridge M, Murphy DJ (2009) Roles of a membrane-bound caleosin and putative peroxygenase in biotic and abiotic stress responses in Arabidopsis. Plant Physiol Biochem 47:796–806
Pasaribu B, Chen CS, Liao YK, Jiang PL, Tzen JTC (2017) Identification of caleosin and oleosin in oil bodies of pine pollen. Plant Physiol Biochem 111:20–29
Pateraki I, Andersen-Ranberg J, Hamberger B, Maree Heskes A, Martens HJ, Zerbe P, Bach SS, Møller BL, Bohlmann J, Hamberger B (2014) Manoyl oxide (13R), the biosynthetic precursor of forskolin, is synthesized in specialized root cork cells in Coleus forskohlii. Plant Physiolgy 164:1222–1236
Porta H, Rocha-Sosa M (2002) Plant lipoxygenases. Physiological and molecular features. Plant Physiol 130:15–21
Pyc M, Gidda SK, Seay D, Esnay N, Kretzschmar FK, Cai Y, Doner NM, Greer MS, Hull JJ, Coulon D, Brehelin C, Yurchenko O, de Vries J, Valerius O, Braus G, Ischebeck T, Chapman KD, Dyer JM, Mullen RT (2021) LDIP cooperates with SEIPIN and LDAP to facilitate lipid droplet biogenesis in Arabidopsis. Plant Cell 33:3076–3103
Rahman F, Hassan M, Hanano A, Fitzpatrick DA, McCarthy CGP, Murphy DJ (2018a) Evolutionary, structural and functional analysis of the caleosin/peroxygenase gene family in the Fungi. BMC Genomics 19:976
Rahman F, Hassan M, Rosli R, Almousally I, Hanano A, Murphy DJ (2018b) Evolutionary and genomic analysis of the caleosin/peroxygenase (CLO/PXG) gene/protein families in the Viridiplantae. PLoS ONE 13:e0196669
Ramirez-Estrada K, Altabella T, Onrubia M, Moyano E, Notredame C, Osuna L, Vanden Bossche R, Goossens A, Cusido RM, Palazon J (2016) Transcript profiling of jasmonate-elicited Taxus cells reveals a beta-phenylalanine-CoA ligase. Plant Biotechnol J 14:85–96
Rangel C, Niell H, Miller A, Cox C (1994) Taxol and taxotere in bladder cancer: in vitro activity and urine stability. Cancer Chemother Pharmacol 33:460–464
Reinbothe C, Springer A, Samol I, Reinbothe S (2009) Plant oxylipins: role of jasmonic acid during programmed cell death, defence and leaf senescence. FEBS J 276:4666–4681
Ringel I, Horwitz SB (1991) Studies with RP 56976 (taxotere): a semisynthetic analogue of taxol. J Natl Cancer Inst 83:288–291
Romani F, Banić E, Florent NS, Kanazawa T, Goodger QDJ, Mentink AR, Dierschke T, Zachgo S, Ueda T, Bowman LJ, Tsiantis M, Moreno JE (2020) Oil body formation in Marchantia polymorpha is controlled by MpC1HDZ and serves as a defense against arthropod herbivores. Curr Biol 30:2815–2828
Sabater-Jara AB, Onrubia M, Moyano E, Bonfill M, Palazon J, Pedreno MA, Cusido RM (2014) Synergistic effect of cyclodextrins and methyl jasmonate on taxane production in Taxus x media cell cultures. Plant Biotechnol J 12:1075–1084
Sarmiento C, Ross JHE, Herman E, Murphy DJ (1997) Expression and subcellular targeting of a soybean oleosin in transgenic rapeseed. Implications for the mechanism of oil-body formation in seeds. Plant J 11:783–796
Savchenko TV, Zastrijnaja OM, Klimov VV (2014) Oxylipins and plant abiotic stress resistance. Biochem Mosc 79:362–375
Shao F, Wilson IW, Qiu D (2021) The research progress of taxol in Taxus. Curr Pharm Biotechnol 22:360–366
Sharma VK, Monostori T, Gobel C, Hansch R, Bittner F, Wasternack C, Feussner I, Mendel RR, Hause B, Schulze J (2006) Transgenic barley plants overexpressing a 13-lipoxygenase to modify oxylipin signature. Phytochemistry 67:264–276
Shimada TL, Hara-Nishimura I (2015) Leaf oil bodies are subcellular factories producing antifungal oxylipins. Curr Opin Plant Biol 25:145–150
Srinivasan V, Pestchanker L, Moser S, Hirasuna TJ, Taticek RA, Shuler ML (1995) Taxol production in bioreactors: kinetics of biomass accumulation, nutrient uptake, and taxol production by cell suspensions of Taxus baccata. Biotechnol Bioeng 47:666–676
Stage TB, Bergmann TK, Kroetz DL (2018) Clinical pharmacokinetics of paclitaxel monotherapy: an updated literature review. Clin Pharmacokinet 57:7–19
Stintzi A, Weber H, Reymond P, Browse J, Farmer EE (2001) Plant defense in the absence of jasmonic acid: the role of cyclopentenones. Proc Natl Acad Sci USA 98:12837–12842
Talebi M, Ghassempour A, Talebpour Z, Rassouli A, Dolatyari L (2004) Optimization of the extraction of paclitaxel from Taxus baccata L. by the use of microwave energy. J Sep Sci 27:1130–1136
Tanchak MA, Fowke LC (1987) The morphology of multivesicular bodies in soybean protoplasts and their role in endocytosis. Protoplasma 138:173–182
Tse YC, Mo B, Hillmer S, Zhao M, Lo SW, Robinson DG, Jiang L (2004) Identification of multivesicular bodies as prevacuolar compartments in Nicotiana tabacum BY-2 cells. Plant Cell 16:672–693
Tzen JT, Huang AH (1992) Surface structure and properties of plant seed oil bodies. J Cell Biol 117:327–335
Tzen JTC, Lie GC, Huang AHC (1992) Characterization of the charged components and their topology on the surface of plant seed oil bodies. J Biol Chem 267:15626–15634
Tzen J, Cao Y, Laurent P, Ratnayake C, Huang A (1993) Lipids, proteins, and structure of seed oil bodies from diverse species. Plant Physiol 101:267–276
van Herpen NA, Schrauwen-Hinderling VB (2008) Lipid accumulation in non-adipose tissue and lipotoxicity. Physiol Behav 94:231–241
Vongpaseuth K, Roberts SR (2007) Advancements in the understanding of paclitaxel metabolism in tissue culture. Curr Pharm Biotechnol 8:219–236
Wani MC, Horwitz SB (2014) Nature as a remarkable chemist: a personal story of the discovery and development of taxol. Anticancer Drugs 25:482–487
Whisson SC, Avrova AO, Van West P, Jones JT (2005) A method for double-stranded RNA-mediated transient gene silencing in Phytophthora infestans. Mol Plant Pathol 6:153–163
White DA, Fisk ID, Gray DA (2006) Characterisation of oat (Avena sativa L.) oil bodies and intrinsically associated E-vitamers. J Cereal Sci 43:244–249
Willmann MR (2002) Plant defense in the absence of jasmonic acid. Trends Plant Sci 7:8–9
Wu J, Lin L (2003) Enhancement of taxol production and release in Taxus chinensis cell cultures by ultrasound, methyl jasmonate and in situ solvent extraction. Appl Microbiol Biotechnol 62:151–155
Zhang C, Qu Y, Lian Y, Chapman M, Chapman N, Xin J, Xin H, Liu L (2020) A new insight into the mechanism for cytosolic lipid droplet degradation in senescent leaves. Physiol Plant 168:835–844
Zhi Y, Taylor MC, Campbell PM, Warden AC, Shrestha P, El Tahchy A, Rolland V, Vanhercke T, Petrie JR, White RG, Chen W, Singh SP, Liu Q (2017) Comparative lipidomics and proteomics of lipid droplets in the mesocarp and seed tissues of Chinese tallow (Triadica sebifera). Front Plant Sci 8:1339
Zienkiewicz K, Zienkiewicz A (2020c) Degradation of lipid droplets in plants and algae—right time, many paths, one goal. Front Plant Sci 11:579019
Acknowledgements
We thank Prof. Dr. Ibrahim OTHMAN, Director General of the Atomic Energy Commission of Syria and Dr. Nizar MIRALI, Head of the Department of Molecular Biology and Biotechnology for their crucial support. Our co-authors, Rosa M. Cusido and Edgar Perez-Matas, also thank the Spanish MEIC (BIO2017-82374-R) (AEI/FEDER, UE) and the Generalitat de Catalunya (2017SGR242).
Funding
No funding to be declared.
Author information
Authors and Affiliations
Contributions
AH conceived and designed the experimental work and wrote the first draft of manuscript. EPM contributed to knowledge transfer of tissues culture protocols. MS performed the experiments. RMC supervised the work, read and commented the manuscript. DJM revised and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Availability of data and materials
Not applicable.
Code availability
Not applicable.
Ethics approval
Not applicable.
Consent to participate and consent for publication
All of the authors contributed substantially to the manuscript and approved the final submission in Plant Cell Reports.
Additional information
Communicated by Kunpeng Jia.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hanano, A., Perez-Matas, E., Shaban, M. et al. Characterization of lipid droplets from a Taxus media cell suspension and their potential involvement in trafficking and secretion of paclitaxel. Plant Cell Rep 41, 853–871 (2022). https://doi.org/10.1007/s00299-021-02823-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-021-02823-0