Abstract
Key message
Calli protoplasts isolated from three soybean cultivars are useful tools to evaluate guide RNAs for clustered regularly interspaced short palindromic repeats (CRISPR)-based precise gene editing.
Abstract
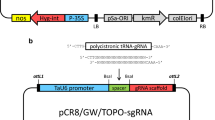
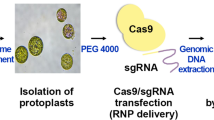
A type V CRISPR effector, LbCpf1(Cas12a) from Lachnospiraceae bacterium ND 2006, has been used for precision editing of the plant genome. We report that callus-derived protoplasts from three soybeans, including Glycine Max var. Williams 82 and two Korean cultivars (Kwangan and Daewon) represent efficient systems for the screening of active crRNA for CRISPR/LbCpf1. CRISPR/LbCpf1 ribonucleoproteins (RNPs) were delivered as complexes of purified endonucleases mixed with designed crRNA to simultaneously edit target genes of GlymaFAD2-1A and GlymaFAD2-1B transfected into three soybean protoplasts including genome-sequenced Williams 82 with cultivars, Kwangan and Daewon. Previously, we reported that nine crRNAs designed for LbCpf1 exhibited varying degrees of editing efficacy for two FAD2 genes. Among the nine crRNAs, the LbCpf1-crRNA3 complexes showed the highest efficiency in soybean cotyledon protoplasts. The new screening systems of callus protoplasts from three soybeans have been successfully used to transfect GFP-tagged markers and CRISPR/LbCpf1 RNPs. The callus protoplasts confirm that the LbCpf1-crRNA3 complex is an active crRNA for LbCpf1 to edit two FAD2 genes similar to cotyledon protoplasts. These results demonstrate that soybean callus protoplast-based CRISPR/crRNA selection is a new and practical tool to screen the efficacy of crRNAs and a prerequisite for progressive regeneration of the edited soybean.





Similar content being viewed by others
References
Cocking EC (1960) A method for the isolation of plant protoplasts and vacuoles. Nature 187:962–963. https://doi.org/10.1038/187962a0
Davey MR, Anthony P, Power JB, Lowe KC (2005) Plant protoplasts: status and biotechnological perspectives. Biotechnol Adv 23:131–171. https://doi.org/10.1016/j.biotechadv.2004.09.008
Deka PC, Sen SK (1976) Differentiation in calli originated from isolated protoplasts of rice (Oryza sativa L.) through plating technique. MGG Mol Gen Genet 145:239–243. https://doi.org/10.1007/BF00325818
Galbraith DW, Harkins KR, Maddox JM et al (1983) Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220:1049–1051. https://doi.org/10.1126/science.220.4601.1049
Haun W, Coffman A, Clasen BM et al (2014) Improved soybean oil quality by targeted mutagenesis of the fatty acid desaturase 2 gene family. Plant Biotechnol J 12:934–940. https://doi.org/10.1111/pbi.12201
Hur JK, Kim K, Been KW et al (2016) Targeted mutagenesis in mice by electroporation of Cpf1 ribonucleoproteins. Nat Biotechnol 34:807–808
Hwang I, Sheen J (2001) Two-component circuitry in Arabidopsis cytokinin signal transduction. Nature 413:383–389. https://doi.org/10.1038/35096500
Jiang WZ, Henry IM, Lynagh PG et al (2017) Significant enhancement of fatty acid composition in seeds of the allohexaploid, Camelina sativa, using CRISPR/Cas9 gene editing. Plant Biotechnol J 15:648–657. https://doi.org/10.1111/pbi.12663
Jie E-Y, Kim S-W, Jang H-R et al (2011) Myo-inositol increases the plating efficiency of protoplast derived from cotyledon of cabbage (Brassica oleracea var. capitata). J Plant Biotechnol 38:69–76. https://doi.org/10.5010/JPB.2011.38.1.069
Kim H, Park M, Kim SJ, Hwang I (2005) Actin filaments play a critical role in vacuolar trafficking at the Golgi complex in plant cells. Plant Cell 17:888–902. https://doi.org/10.1105/tpc.104.028829
Kim H, Kang H, Jang M et al (2010) Homomeric interaction of AtVSR1 is essential for its function as a vacuolar sorting receptor. Plant Physiol 154:134–148. https://doi.org/10.1104/pp.110.159814
Kim D, Kim J, Hur JK et al (2016) Genome-wide analysis reveals specificities of Cpf1 endonucleases in human cells. Nat Biotechnol 34:863–868. https://doi.org/10.1038/nbt.3609
Kim H, Kim ST, Ryu J et al (2017) CRISPR/Cpf1-mediated DNA-free plant genome editing. Nat Commun 8:1–7. https://doi.org/10.1038/ncomms14406
Kleinstiver BP, Tsai SQ, Prew MS et al (2016) Genome-wide specificities of CRISPR-Cas Cpf1 nucleases in human cells. Nat Biotechnol 34:869. https://doi.org/10.1038/nbt.3620
Lee K-J, Seo J-K, Lee H-Y et al (2006) Optimization of genetic transformation conditions for korean soybean cultivars. J Life Sci 16:289–296. https://doi.org/10.5352/JLS.2006.16.2.289
Miao Y, Yan PK, Kim H et al (2006) Localization of green fluorescent protein fusions with the seven Arabidopsis vacuolar sorting receptors to prevacuolar compartments in tobacco BY-2 cells. Plant Physiol 142:945–962. https://doi.org/10.1104/pp.106.083618
Okuley J, Lightner J, Feldmann K et al (1994) Arabidopsis FAD2 gene encodes the enzyme that is essential for polyunsaturated lipid synthesis. Plant Cell 6:147–158. https://doi.org/10.1105/tpc.6.1.147
Park J, Bae S, Kim JS (2015) Cas-designer: a web-based tool for choice of CRISPR-cas9 target sites. Bioinformatics 31:4014–4016. https://doi.org/10.1093/bioinformatics/btv537
Park J, Lim K, Kim J-S, Bae S (2017) Cas-analyzer: an online tool for assessing genome editing results using NGS data. Bioinformatics 33:286–288. https://doi.org/10.1093/bioinformatics/btw561
Schlueter JA, Vasylenko-Sanders IF, Deshpande S et al (2007) The FAD2 gene family of soybean: insights into the structural and functional divergence of a paleopolyploid genome. Crop Sci 47:S-14–S-26. https://doi.org/10.2135/cropsci2006.06.0382tpg
Schmutz J, Cannon SB, Schlueter J et al (2010) Genome sequence of the palaeopolyploid soybean. Nature 463:178–183. https://doi.org/10.1038/nature08670
Sheen J, Hwang S, Niwa Y et al (1995) Green-fluorescent protein as a new vital marker in plant cells. Plant J 8:777–784. https://doi.org/10.1046/j.1365-313X.1995.08050777.x
Sohn EJ, Kim ES, Zhao M et al (2003) Rha1, an Arabidopsis Rab5 homolog, plays a critical role in the vacuolar trafficking of soluble cargo proteins. Plant Cell 15:1057–1070. https://doi.org/10.1105/tpc.009779
Spychalla JP, Kinney AJ, Browse J (1997) Identification of an animal omega-3 fatty acid desaturase by heterologous expression in Arabidopsis. Proc Natl Acad Sci USA 94:1142–1147. https://doi.org/10.1073/pnas.94.4.1142
Takebe I, Otsuki Y, Aoki S (1968) Isolation of tobacco mesophyll cells in intact and active state. Plant Cell Physiol 9:115–124. https://doi.org/10.1093/oxfordjournals.pcp.a079318
Tang X, Lowder LG, Zhang T et al (2017) A CRISPR–Cpf1 system for efficient genome editing and transcriptional repression in plants. Nat Plants 3:1–5. https://doi.org/10.1038/nplants.2017.18
Woo JW, Kim J, Kwon SI et al (2015) DNA-free genome editing in plants with preassembled CRISPR-Cas9 ribonucleoproteins. Nat Biotechnol 33:1162–1164. https://doi.org/10.1038/nbt.3389
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572. https://doi.org/10.1038/nprot.2007.199
Zetsche B, Gootenberg JS, Abudayyeh OO et al (2015) Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 163:759–771. https://doi.org/10.1016/j.cell.2015.09.038
Acknowledgements
We thank Prof. Ik-Young Choi, Prof. Jeong Dong Lee, and Dr. Jung-Kyung Moon for providing initial soybean materials, and Ms. Hyunah Lee in the Central Laboratory of Kangwon National University for confocal microscopy (LSM880 with Airyscan). We deeply appreciate the technical contributions provided by Ms. Min Kyung Choi and Dr. Jiyeon Kweon. Both authors appreciate PCGE lab members.
Funding
This work was supported by the Basic Science Research Program of National Research Foundation of Korea, funded by the Ministry of Education, Science and Technology [Grant No.2018R1A2B6006233], and the New Breeding Technologies Development Program, Rural Development Administration (RDA), Republic of Korea [Grant No. PJ01477602] to HK and The National Research Foundation of Korea and the Center for Women in Science, Engineering, and Technology [Grant No. WISET-2019-674ho] to J.C.
Author information
Authors and Affiliations
Contributions
HK designed and performed the project. JC and HK performed and assisted experiments. HK wrote the manuscript. HK supervised the project. Both authors approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Neal Stewart.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, H., Choi, J. A robust and practical CRISPR/crRNA screening system for soybean cultivar editing using LbCpf1 ribonucleoproteins. Plant Cell Rep 40, 1059–1070 (2021). https://doi.org/10.1007/s00299-020-02597-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-020-02597-x