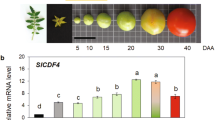
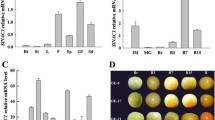
Abstract
Key message
SlPRE2 is gibberellin inducible and mediates plant response to gibberellin. Silencing of SlPRE2 decreases tomato fruit size, pericarp thickness, placenta size and seed size by regulating cell expansion.
Abstract
Gibberellin is one of the crucial hormones essential for plant growth and developmental processes, including seed germination, stem elongation, and sex expression. Previous studies indicated gibberellin could control fruit development by regulation of genes downstream gibberellin pathway. In the present study, we found that the SlPRE2, a bHLH family transcription factor gene, is highly expressed in immature green fruit. Silencing of SlPRE2 caused reduction of fruits size, pericarp thickness, and placenta size. Meanwhile, smaller seeds were observed in SlPRE2 silenced lines. In addition, the SlPRE2-silenced fruit mesocarp had reduced cell size and expression of SlXTH2 and SlXTH5 which are involved in cell enlargement. Further research showed that SlPRE2 is gibberellic acid-inducible and the expression of gibberellin metabolism-related genes in immature green fruit was affected by the downregulation of SlPRE2. Moreover, the SlPRE2-silenced plants had changed responses to application of exogenous gibberellic acid and paclobutrazol, an inhibitor of gibberellin biosynthesis. These findings indicated that SlPRE2 is a regulator of fruit development and affects plant response to gibberellic acid via the gibberellin pathway.








Similar content being viewed by others
Abbreviations
- B:
-
Breaker
- BR:
-
Brassinosteroid
- DPA:
-
Days post-anthesis
- GA:
-
Gibberellin
- GA3 :
-
Gibberellic acid
- IMG:
-
Immature green fruit
- MG:
-
Mature green fruit
- PAC:
-
Paclobutrazol
References
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1
Bai MY, Fan M, Oh E, Wang ZY (2012a) A triple helix–loop–helix/basic helix–loop–helix cascade controls cell elongation downstream of multiple hormonal and environmental signaling pathways in Arabidopsis. Plant Cell 24:4917–4929
Bai MY, Shang JX, Oh E, Fan M, Bai Y, Zentella R, Sun TP, Wang ZY (2012b) Brassinosteroid, gibberellin and phytochrome impinge on a common transcription module in Arabidopsis. Nat Cell Biol 14:810–817
Bergervoet JHW, Verhoeven HA, Gilissen LJW, Bino RJ (1996) High amounts of nuclear DNA in tomato (Lycopersicon esculentum Mill) pericarp. Plant Sci 116:141–145
Bisbis B, Delmas F, Joubès J, Sicard A, Hernould M, Inzé D, Mouras A, Chevalier C (2006) Cyclin-dependent kinase (CDK) inhibitors regulate the CDK–cyclin complex activities in endoreduplicating cells of developing tomato fruit. J Bio Chem 281:7374–7383
Carrera E, Ruiz-Rivero O, Peres LE, Atares A, Garcia-Martinez JL (2012) Characterization of the procera tomato mutant shows novel functions of the SlDELLA protein in the control of flower morphology, cell division and expansion, and the auxin-signaling pathway during fruit-set and development. Plant Physiol 160:1581–1596
Catalá C, Rose JK, York WS, Albersheim P, Darvill AG, Bennett AB (2001) Characterization of a tomato xyloglucan endotransglycosylase gene that is down-regulated by auxin in etiolated hypocotyls. Plant Physiol 127:1180–1192
Chen GP, Hackett R, Walker D, Taylor A, Lin ZF, Grierson D (2004) Identification of a specific isoform of tomato lipoxygenase (TomloxC) involved in the generation of fatty acid-derived flavor compounds. Plant Physiol 136:2641–2651
Chen KY, Cong B, Wing R, Vrebalov J, Tanksley SD (2007) Changes in regulation of a transcription factor lead to autogamy in cultivated tomatoes. Science 318:643–645
Cheniclet C, Rong WY, Causse M, Frangne N, Bolling L, Carde JP, Renaudin JP (2005) Cell expansion and endoreduplication show a large genetic variability in pericarp and contribute strongly to tomato fruit growth. Plant Physiol 139:1984–1994
Colebrook EH, Thomas SG, Phillips AL, Hedden P (2014) The role of gibberellin signalling in plant responses to abiotic stress. J Exp Biol 217:67–75
Cong B, Liu JP, Tanksley SD (2002) Natural alleles at a tomato fruit size quantitative trait locus differ by heterochronic regulatory mutations. Proc Natl Acad Sci 99:13606–13611
De Jong M, Wolters-Arts M, Schimmel BC, Stultiens CL, de Groot PF, Powers SJ, Tikunov YM, Bovy AG, Mariani C, Vriezen WH (2015) Solanum lycopersicum AUXIN RESPONSE FACTOR 9 regulates cell division activity during early tomato fruit development. J Exp Bot 66:3405–3416
de Lucas M, Daviere J-M, Rodriguez-Falcon M, Pontin M, Iglesias-Pedraz JM, Lorrain S, Fankhauser C, Blazquez MA, Titarenko E, Prat S (2008) A molecular framework for light and gibberellin control of cell elongation. Nature 451:480
Depuydt S, Hardtke CS (2011) Hormone signalling crosstalk in plant growth regulation. Curr Biol 21:R365–R373
Dill A, Sun T-P (2001) Synergistic derepression of gibberellin signaling by removing RGA and GAI function in Arabidopsis thaliana. Genetics 159:777–785
Dostal H, Leopold A (1967) Gibberellin delays ripening of tomatoes. Science 158:1579–1580
Endo T, Fujii H, Sugiyama A, Nakano M, Nakajima N, Ikoma Y, Omura M, Shimada T (2016) Overexpression of a citrus basic helix–loop–helix transcription factor (CubHLH1), which is homologous to Arabidopsis activation-tagged bri1 suppressor 1 interacting factor genes, modulates carotenoid metabolism in transgenic tomato. Plant Sci 243:35–48
Exposito-Rodriguez M, Borges AA, Borges-Perez A, Perez JA (2008) Selection of internal control genes for quantitative real-time RT-PCR studies during tomato development process. Bmc Plant Biol 8:131
Fos M, Proaño K, Nuez F, García-Martínez JL (2001) Role of gibberellins in parthenocarpic fruit development induced by the genetic system pat-3/pat-4 in tomato. Physiol Plantarum 111:545–550
Gillaspy G, Ben-David H, Gruissem W (1993) Fruits: a developmental perspective. Plant Cell 5:1439
Giovannoni JJ (2004) Genetic regulation of fruit development and ripening. Plant Cell 16:S170–S180
Groot SP, Bruinsma J, Karssen CM (1987) The role of endogenous gibberellin in seed and fruit development of tomato: studies with a gibberellin-deficient mutant. Physiol Plantarum 71:184–190
Hao Y, Oh E, Choi G, Liang Z, Wang ZY (2012) Interactions between HLH and bHLH factors modulate light-regulated plant development. Mol Plant 5:688–697
Ikeda M, Mitsuda N, Ohme-Takagi M (2013) ATBS1 INTERACTING FACTORs negatively regulate Arabidopsis cell elongation in the triantagonistic bHLH system. Plant Signal Behav 8:e23448
Joubès J, Walsh D, Raymond P, Chevalier C (2000) Molecular characterization of the expression of distinct classes of cyclins during the early development of tomato fruit. Planta 211:430–439
Kvarnheden A, Yao JL, Zhan X, O’Brien I, Morris BA (2000) Isolation of three distinct CycD3 genes expressed during fruit development in tomato. J Exp Bot 51:1789–1797
Lee S, Yang KY, Kim YM, Park SY, Kim SY, Soh MS (2006) Overexpression of PRE1 and its homologous genes activates Gibberellin-dependent responses in Arabidopsis thaliana. Plant Cell Physiol 47:591–600
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Middleton AM, Úbeda-Tomás S, Griffiths J, Holman T, Hedden P, Thomas SG, Phillips AL, Holdsworth MJ, Bennett MJ, King JR (2012) Mathematical modeling elucidates the role of transcriptional feedback in gibberellin signaling. Proc Natl Acad Sci 109:7571–7576
Murase K, Hirano Y, T-p Sun, Hakoshima T (2008) Gibberellin-induced DELLA recognition by the gibberellin receptor GID1. Nature 456:459–463
Peng J, Harberd NP (1997) Gibberellin deficiency and response mutations suppress the stem elongation phenotype of phytochrome-deficient mutants of Arabidopsis. Plant Physiol 113:1051–1058
Picken A (1984) A review of pollination and fruit set in the tomato (Lycopersicon esculentum Mill.). J Hortic Sci 59:1–13
Powell ALT, Nguyen CV, Hill T, Cheng KL, Figueroa-Balderas R, Aktas H, Ashrafi H, Pons C, Fernandez-Munoz R, Vicente A, Lopez-Baltazar J, Barry CS, Liu Y, Chetelat R, Granell A, Van Deynze A, Giovannoni JJ, Bennett AB (2012) Uniform ripening encodes a golden 2-like transcription factor regulating tomato fruit chloroplast development. Science 336:1711–1715
Primack RB (1987) Relationships among flowers, fruits, and seeds. Annu Rev Ecol Syst 18:409–430
Saladié M, Rose JK, Cosgrove DJ, Catalá C (2006) Characterization of a new xyloglucan endotransglucosylase/hydrolase (XTH) from ripening tomato fruit and implications for the diverse modes of enzymic action. Plant J 47:282–295
Saville R, Gosman N, Burt C, Makepeace J, Steed A, Corbitt M, Chandler E, Brown J, Boulton M, Nicholson P (2012) The ‘Green Revolution’ dwarfing genes play a role in disease resistance in Triticum aestivum and Hordeum vulgare. J Exp Bot 63:1271–1283
Seo PJ, Hong SY, Kim SG, Park CM (2011) Competitive inhibition of transcription factors by small interfering peptides. Trends Plant Sci 16:541–549
Serrani JC, Sanjuán R, Ruiz-Rivero O, Fos M, García-Martínez JL (2007) Gibberellin regulation of fruit set and growth in tomato. Plant Physiol 145:246–257
Shi LF, Olszewski NE (1998) Gibberellin and abscisic acid regulate GAST1 expression at the level of transcription. Plant Mol Biol 38:1053–1060
Shimada A, Ueguchi-Tanaka M, Nakatsu T, Nakajima M, Naoe Y, Ohmiya H, Kato H, Matsuoka M (2008) Structural basis for gibberellin recognition by its receptor GID1. Nature 456:520–523
Sotelo-Silveira M, Marsch-Martinez N, de Folter S (2014) Unraveling the signal scenario of fruit set. Planta 239:1147–1158
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Tanksley SD (2004) The genetic, developmental, and molecular bases of fruit size and shape variation in tomato. Plant Cell 16:S181–S189
Tyler L, Thomas SG, Hu J, Dill A, Alonso JM, Ecker JR, T-p Sun (2004) DELLA proteins and gibberellin-regulated seed germination and floral development in Arabidopsis. Plant Physiol 135:1008–1019
Ubeda-Tomás S, Swarup R, Coates J, Swarup K, Laplaze L, Beemster GT, Hedden P, Bhalerao R, Bennett MJ (2008) Root growth in Arabidopsis requires gibberellin/DELLA signalling in the endodermis. Nat Cell Biol 10:625–628
Vivian-Smith A, Koltunow AM (1999) Genetic analysis of growth-regulator-induced parthenocarpy in arabidopsis. Plant Physiol 121:437–451
Wang H, Zhu Y, Fujioka S, Asami T, Li J (2009) Regulation of Arabidopsis brassinosteroid signaling by atypical basic helix–loop–helix proteins. Plant Cell 21:3781–3791
Zhou P, Song M, Yang Q, Su L, Hou P, Guo L, Zheng X, Xi Y, Meng F, Xiao Y (2014) Both PHYTOCHROME RAPIDLY REGULATED1 (PAR1) and PAR2 promote seedling photomorphogenesis in multiple light signaling pathways. Plant Physiol 164:841–852
Zhu Z, Chen G, Guo X, Yin W, Yu X, Hu J, Hu Z (2017) Overexpression of SlPRE2, an atypical bHLH transcription factor, affects plant morphology and fruit pigment accumulation in tomato. Sci Rep 7:5786
Acknowledgements
This work was supported by National Natural Science Foundation of China (nos. 30600044, 31572129).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Kan Wang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhu, Z., Liang, H., Chen, G. et al. The bHLH transcription factor SlPRE2 regulates tomato fruit development and modulates plant response to gibberellin. Plant Cell Rep 38, 1053–1064 (2019). https://doi.org/10.1007/s00299-019-02425-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-019-02425-x