Abstract
Key message
Elevated antioxidant status and positive abiotic stress response in dehydration enhance cell resistance to cryoinjury, and controlling oxidative damage via reactive oxygen species homeostasis maintenance leads to high survival.
Abstract
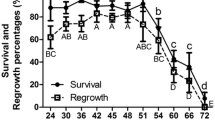
Cryoprotectants are important for cell survival in cryopreservation, but high concentrations can also cause oxidative stress. Adding vitamin C to the cryoprotectant doubled the survival ratio in Arabidopsis thaliana 60-h seedlings (seedlings after 60-h germination) cryopreservation. In this study, the metabolites and transcriptional profiling of 60-h seedlings were analyzed in both the control cryopreservation procedure (CCP) and an improved cryopreservation procedure (ICP) to reveal the mechanism of plant cell response to oxidative stress from cryopreservation. Reactive oxygen species (ROS) and peroxidation levels reached a peak after rapid cooling–warming in CCP, which were higher than that in ICP. In addition, gene regulation was significantly increased in CCP and decreased in ICP during rapid cooling–warming. Before cryogenic treatment, the number of specifically regulated genes was nearly 10 times higher in ICP dehydration than CCP dehydration. Among these genes, DREBs/CBFs were beneficial to cope with cryoinjury, and calcium-binding protein, OXI1, WRKY and MYB family members as key factors in ROS signal transduction activated the ROS-producing and ROS-scavenging networks including AsA-GSH and GPX cycles involved in scavenging H2O2. Finally, elevated antioxidant status and oxidative stress response in the improved dehydration enhanced seedling resistance to cryogenic treatment, maintained ROS homeostasis and improved cell recovery after cryopreservation.









Similar content being viewed by others
References
Abe H, Urao T, Ito T, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2003) Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling. Plant Cell 15:63–78
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Baust JM (2002) Molecular mechanisms of cellular demise associated with cryopreservation failure. Cell Preserv Technol 1:17–31
Benson EE (2008a) Cryopreservation theory. In: Reed BM (ed) Plant Cryopreservation: a practical guide. Springer-Verlag, USA, pp 15–32
Benson EE (2008b) Cryopreservation of phytodiversity: a critical appraisal of theory & practice. Crit Rev Plant Sci 27:141–219
Benson EE, Bremner D (2004) Oxidative stress in the frozen plant: a free radical point of view. In: Fuller BJ, Lane N, Benson EE (eds) Life in the frozen state. CRC Press, USA, pp 205–241
Benson EE, Noronha-Dutra AA (1988) Chemiluminescence in cryopreserved plant tissue cultures: the possible role of singlet oxygen in cryoinjury. Cryo Lett 9:120–131
Benson EE, Lynch PT, Jones J (1992) The detection of lipid peroxidation products in cryoprotected and frozen rice cells—consequences for post thaw survival. Plant Sci 85:107–114
Bowler C, Fluhr R (2000) The role of calcium and activated oxygens as signals for controlling cross-tolerance. Trends Plant Sci 5:241–246
Burbridge E, Diamond M, Dix PJ, McCabe PF (2006) Use of cell morphology to evaluate the effect of a peroxidase gene on cell death induction thresholds in tobacco. Plant Sci 171:139–146
Chen MX, Lung SC, Du ZY, Chye ML (2014) Engineering plants to tolerate abiotic stresses. Biocatal Agric Biotechnol 3:81–87
Chen GQ, Ren L, Zhang J, Reed BM, Zhang D, Shen XH (2015) Cryopreservation affects ROS-induced oxidative stress and antioxidant response in Arabidopsis seedlings. Cryobiology 70:38–47
Chinnusamy V, Zhu J, Zhu JK (2007) Cold stress regulation of gene expression in plants. Trends Plant Sci 12:444–451
Coelho SM, Taylor AR, Ryan KP, Sousa-Pinto I, Brown MT, Brownlee C (2002) Spatiotemporal patterning of reactive oxygen production and Ca2+ wave propagation in Fucus rhizoid cells. Plant Cell 14:2369–2381
Coll NS, Epple P, Dangl JL (2011) Programmed cell death in the plant immune system. Cell Death Differ 18:1247–1256
Dat JF, Vandenabeele S, Vranová E, Van Montagu M, Inzé D, Van Breusegem F (2000) Dual action of the active oxygen species during plant stress responses. Cell Mol Life Sci 57:779–795
Dat JF, Pellinen R, Beeckman T, Van De Cotte B, Langebartels C, Kangasjärvi J, Inzé D, Van Breusegem F (2003) Changes in hydrogen peroxide homeostasis trigger an active cell death process in tobacco. Plant J 33:621–632
Du Z, Zhou X, Ling Y, Zhang Z, Su Z (2010) agriGO: a GO analysis toolkit for the agricultural community. Nucl Acids Res 38:W64–W70
Eisen MB, Spellman PT, Brown PO, Botstein D (1998) Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 95:14863–14868
Engelmann F (2004) Cryopreservation: progress and prospects. In Vitro Cell Dev-Pl 40:427–433
Epple P, Mack AA, Morris VR, Dangl JL (2003) Antagonistic control of oxidative stress-induced cell death in Arabidopsis by two related, plant-specific zinc finger proteins. Proc Natl Acad Sci USA 100:6831–6836
Fang JY, Wetten A, Johnston J (2008) Headspace volatile markers for sensitivity of cocoa (Theobroma cacao L.) somatic embryos to cryopreservation. Plant Cell Rep 27:453–461
Fowler S, Thomashow MF (2002) Arabidopsis transcriptome profiling indicates that multiple regulatory pathways are activated during cold acclimation in addition to the CBF cold response pathway. Plant Cell 14:1675–1690
Fumitaka A, Koji S, Kiyoyuki M, Kinya T (2002) A single nucleotide polymorphism in the alternative oxidase gene among rice varieties differing in low temperature tolerance. FEBS Lett 527:181–185
Gadjev I, Vanderauwera S, Gechev TS, Laloi C, Minkov IN, Shulaev V, Apel K, Inzé D, Mittler R, Van Breusegem F (2006) Transcriptomic footprints disclose specificity of reactive oxygen species signaling in Arabidopsis. Plant Physiol 141:434–445
Gechev TS, van Breusegem F, Stone JM, Denev I, Laloi C (2006) Reactive oxygen species as signals that modulate plant stress responses and programmed cell death. Bio Essays 28:1091–1101
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Halliwell B (2006) Reactive species and antioxidants: redox biology is a fundamental theme of aerobic life. Plant Physiol 141:312–322
Harding K, Johnston JW, Benson EE (2009) Exploring the physiological basis of cryopreservation success and failure in clonally propagated in vitro crop plant germplasm. Agric Food Sci 18:103–116
Hsieh TH, Lee JT, Yang PT, Chiu LH, Charng YY, Wang YC, Chan MT (2002) Heterology expression of the Arabidopsis C-repeat/dehydration response element binding factor 1 gene confers elevated tolerance to chilling and oxidative stresses in transgenic tomato. Plant Physiol 129:1086–1094
Hu D, Ma G, Wang Q, Yao JH, Wang Y, Pritchard HW, Wang XF (2012) Spatial and temporal nature of reactive oxygen species production and programmed cell death in elm (Ulmus pumila L.) seeds during controlled deterioration. Plant Cell Environ 35:2045–2059
Johnston JW, Harding K, Benson EE (2007) Antioxidant status and genotypic tolerance of Ribes in vitro cultures to cryopreservation. Plant Sci 172:524–534
Knight H, Knight MR (2001) Abiotic stress signalling pathways: specificity and cross-talk. Trends Plant Sci 6:262–267
Kovtun Y, Chiu WL, Tena G, Sheen J (2000) Functional analysis of oxidative stress activated mitogen-activated protein kinase cascade in plants. Proc Natl Acad Sci USA 97:2940–2945
Kulus D, Zalewska M (2014) Cryopreservation as a tool used in long-term storage of ornamental species—a review. Sci Hortic 168:88–107
Lippold F, Sanchez DH, Musialak M, Schlereth A, Scheible WR, Hincha DK, Udvardi MK (2009) AtMyb41 regulates transcriptional and metabolic responses to osmotic stress in Arabidopsis. Plant Physiol 149:1761–1772
Liu J, Li Z, Wang Y, Xing D (2014) Overexpression of ALTERNATIVE OXIDASE1a alleviates mitochondria-dependent programmed cell death induced by aluminium phytotoxicity in Arabidopsis. J Exp Bot 65:4465–4478
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lynch PT, Siddika A, Johnston JW, Trigwell SM, Mehra A, Benelli C, Lambardi M, Benson EE (2011) Effects of osmotic pretreatments on oxidative stress, antioxidant profiles and cryopreservation of olive somatic embryos. Plant Sci 181:47–56
Maxwell DP, Wang Y, McIntosh L (1999) The alternative oxidase lowers mitochondrial reactive oxygen production in plant cells. Proc Natl Acad Sci USA 96:8271–8276
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakashima K, Ito Y, Yamaguchi-Shinozaki K (2009) Transcriptional regulatory networks in response to abiotic stresses in Arabidopsis and grasses. Plant Physiol 149:88–95
Osakabe Y, Yamaguchi-Shinozaki K, Shinozaki K, Tran LP (2013) Sensing the environment: key roles of membrane-localized kinases in plant perception and response to abiotic stress. J Exp Bot 64:445–458
Reed BM (2001) Implementing cryogenic storage of clonally propagated plants. Cryo Lett 22:97–104
Reed BM (2008) Cryopreservation—practical considerations. In: Reed BM (ed) Plant cryopreservation: a practical guide. Springer-Verlag, USA, pp 3–13
Ren X, Chen Z, Liu Y, Zhang H, Zhang M, Liu Q, Hong X, Zhu J, Gong Z (2010) ABO3, a WRKY transcription factor, mediates plant responses to abscisic acid and drought tolerance in Arabidopsis. Plant J 63:417–429
Ren L, Zhang D, Jiang XN, Gai Y, Wang WM, Reed BM, Shen XH (2013) Peroxidation due to cryoprotectant treatment is a vital factor for cell survival in Arabidopsis cryopreservation. Plant Sci 212:37–47
Ren L, Zhang D, Reed BM, Shen XH (2014) Antioxidants and anti-stress compounds improve the survival of cryopreserved Arabidopsis seedlings. Acta Hortic 1039:57–62
Rentel MC, Lecourieux D, Ouaked F, Usher SL, Petersen L, Okamoto H, Knight H, Peck SC, Grierson CS, Hirt H, Knight MR (2004) OXI1 kinase is necessary for oxidative burst-mediated signalling in Arabidopsis. Nature 427:858–861
Rizhsky L, Davletova S, Liang HJ, Mittler R (2004) The zinc finger protein Zat12 is required for cytosolic ascorbate peroxidase 1 expression during oxidative stress in Arabidopsis. J Biol Chem 279:11736–11743
Rodriguez MC, Petersen M, Mundy J (2010) Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol 61:621–649
Rushton DL, Tripathi P, Rabara RC, Lin J, Ringler P, Boken AK, Langum TJ, Smidt L, Boomsma DD, Emme NJ, Chen X, Finer JJ, Shen QJ, Rushton PJ (2012) WRKY transcription factors: key components in abscisic acid signalling. Plant Biotechnol J 10:2–11
Sakai A, Hirai D, Niino T (2008) Development of PVS-based vitrification and encapsulation-vitrification protocols. In: Reed BM (ed) Plant cryopreservation: a practical guide. Springer-Verlag, USA, pp 33–57
Seo PJ, Lee SB, Suh MC, Park MJ, Go YS, Park CM (2011) The MYB96 transcription factor regulates cuticular wax biosynthesis under drought conditions in Arabidopsis. Plant Cell 23:1138–1152
Skyba M, Petijová L, Košuth J, Koleva DP, Ganeva TG, Kapchina-Toteva VM, Čellárová E (2012) Oxidative stress and antioxidant response in Hypericum perforatum L. plants subjected to low temperature treatment. J Plant Physiol 169:955–964
Sreenivasulu N, Sopory SK, Kavi Kishor PB (2007) Deciphering the regulatory mechanisms of abiotic stress tolerance in plants by genomic approaches. Gene 388:1–13
Taylor MJ, Song YC, Brockbank KGM (2004) Vitrification in tissue preservation: New developments. In: Fuller B, Lane N, Benson EE (eds) Life in the Frozen State. CRC Press, London, pp 603–644
Uchendu EE, Leonard SW, Traber MG, Reed BM (2010a) Vitamins C and E improve regrowth and reduce lipid peroxidation of blackberry shoot tips following cryopreservation. Plant Cell Rep 29:25–35
Uchendu EE, Muminova M, Gupta S, Reed BM (2010b) Antioxidant and anti-stress compounds improve regrowth of cryopreserved Rubus shoot tips. In Vitro Cell Dev-Pl 46:386–393
Vacca RA, de Pinto MC, Valenti D, Passarella S, Marra E, De Gara L (2004) Production of reactive oxygen species, alteration of cytosolic ascorbate peroxidase, and impairment of mitochondrial metabolism are early events in heat shock-induced programmed cell death in tobacco bright-yellow 2 cells. Plant Physiol 134:1100–1112
Varghese B, Naithani SC (2008) Oxidative metabolism-related changes in cryogenically stored neem (Azadirachta indica A. Juss) seeds. J Plant Physiol 165:755–765
Volk GM (2010) Application of functional genomics and proteomics to plant cryopreservation. Curr Genomics 11:24–29
Vranová E, Atichartpongkul S, Villarroel R, Van Montagu M, Inzé D, Van Camp W (2002) Comprehensive analysis of gene expression in Nicotiana tabacum leaves acclimated to oxidative stress. Proc Natl Acad Sci USA 99:10870–10875
Wang ZC, Deng XX (2004) Cryopreservation of shoot-tips of citrus using vitrification: effect of reduced form of glutathione. Cryo Lett 25:43–50
Wen B, Wang RL, Cheng HY, Song SQ (2010) Cytological and physiological changes in orthodox maize embryos during cryopreservation. Protoplasma 239:57–67
Whitaker C, Beckett RP, Minibayeva FV, Kranner I (2010) Production of reactive oxygen species in excised, desiccated and cryopreserved explants of Trichilia dregeana Sond. S Afr J Bot 76:112–118
Wu YL (2011) Cryopreservation of dendrobium wardianum by vitrification. Dissertation, Shanghai Jiao Tong University
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 31170655 and No. 31300580) and Shanghai Jiao Tong University ‘Agri-X’ Interdisciplinary Research Foundation (No. 2015003). The authors thank Prof. Hong-Quan Yang (Fudan University, Shanghai, China) for experimental materials.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by S. A. Merkle.
Electronic supplementary material
Below is the link to the electronic supplementary material.
299_2015_1859_MOESM1_ESM.tif
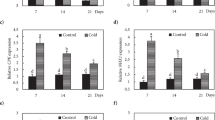
Fig. S1 qRT-PCR analysis of partial differentially expressed genes in microarrayCK, untreated; OM, after osmoprotection; DH, after dehydration; RW, after rapid warming; DL, after dilution; RC, after 24-h recovery. (TIFF 4286 kb)
299_2015_1859_MOESM2_ESM.tif
Fig. S2 Expression patterns comparison of microarray and qRT-PCR in partial differentially expressed genesCK, untreated; OM, after osmoprotection; DH, after dehydration; RW, after rapid warming; DL, after dilution; RC, after 24-h recovery. (TIFF 1558 kb)
299_2015_1859_MOESM3_ESM.tif
Fig. S3 Cluster and GO classification analyses of differential expression genes in the control cryopreservation procedure (TIFF 4570 kb)
299_2015_1859_MOESM4_ESM.tif
Fig. S4 Cluster and GO classification analyses of differential expression genes in the improved cryopreservation procedure (TIFF 3900 kb)
299_2015_1859_MOESM5_ESM.tif
Fig. S5 Gene annotation and expression of ROS signal transduction network in the CCP and ICPOsmo, osmoprotection; Dehy, dehydration; Warm, rapid cooling and rapid warming; Dilu, dilution; Reco: recovery for 24 h. The up-regulated genes are indicated in red and down-regulated genes in green. The intensity of the colors increases as the expression differences increase, as shown in the bar at the bottom. (TIFF 5855 kb)
Rights and permissions
About this article
Cite this article
Ren, L., Zhang, D., Chen, Gq. et al. Transcriptomic profiling revealed the regulatory mechanism of Arabidopsis seedlings response to oxidative stress from cryopreservation. Plant Cell Rep 34, 2161–2178 (2015). https://doi.org/10.1007/s00299-015-1859-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-015-1859-9