Abstract
Key message
Critical regions within the rice metallothionein OsMT2b gene promoter are identified and the 5′-untranslated region (5′-UTR) is found essential for the high-level promoter activity in germinated transgenic rice embryos.
Abstract
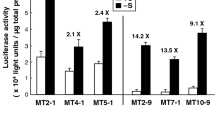
Many metallothionein (MT) genes are highly expressed in plant tissues. A rice subfamily p2 (type 2) MT gene, OsMT2b, has been shown previously to exhibit the most abundant gene expression in young rice seedling. In the present study, transient expression assays and a transgenic approach were employed to characterize the expression of the OsMT2b gene in rice. We found that the OsMT2b gene is strongly and differentially expressed in germinated rice embryos during seed germination and seedling development. Histochemical staining analysis of transgenic rice carrying OsMT2b::GUS chimeric gene showed that high-level GUS activity was detected in germinated embryos and at the meristematic part of other tissues during germination. Deletion analysis of the OsMT2b promoter revealed that the 5′-flanking region of the OsMT2b between nucleotides −351 and −121 relative to the transcriptional initiation site is important for promoter activity in rice embryos, and this region contains the consensus sequences of G box and TA box. Our study demonstrates that the 5′-untranslated region (5′-UTR) of OsMT2b gene is not only necessary for the OsMT2b promoter activity, but also sufficient to augment the activity of a minimal promoter in both transformed cell cultures and germinated transgenic embryos in rice. We also found that addition of the maize Ubi intron 1 significantly enhanced the OsMT2b promoter activity in rice embryos. Our studies reveal that OsMT2b351-ubi(In) promoter can be applied in plant transformation and represents potential for driving high-level production of foreign proteins in transgenic rice.






Similar content being viewed by others
References
Bolle C, Sopory S, Lubberstedt T, Herrmann RG, Oelmuller R (1994) Segments encoding 5′-untranslated leaders of genes for thylakoid proteins contain cis-elements essential for transcription. Plant J 6(4):513–523
Bruce WB, Christensen AH, Klein T, Fromm M, Quail PH (1989) Photoregulation of a phytochrome gene promoter from oat transferred into rice by particle bombardment. Proc Natl Acad Sci USA 86(24):9692–9696
Caspar T, Quail PH (1993) Promoter and leader regions involved in the expression of the Arabidopsis ferredoxin A gene. Plant J 3(1):161–174
Cazzonelli CI, McCallum EJ, Lee R, Botella JR (2005) Characterization of a strong, constitutive mung bean (Vigna radiata L.) promoter with a complex mode of regulation in planta. Transgenic Res 14(6):941–967
Chan MT, Chang HH, Ho SL, Tong WF, Yu SM (1993) Agrobacterium-mediated production of transgenic rice plants expressing a chimeric alpha-amylase promoter/beta-glucuronidase gene. Plant Mol Biol 22(3):491–506
Chan MT, Chao YC, Yu SM (1994) Novel gene expression system for plant cells based on induction of alpha-amylase promoter by carbohydrate starvation. J Biol Chem 269(26):17635–17641
Chen PW, Chiang CM, Tseng TH, Yu SM (2006) Interaction between rice MYBGA and the gibberellin response element controls tissue-specific sugar sensitivity of alpha-amylase genes. Plant Cell 18(9):2326–2340
Chen PW, Fang LW, Lin JW, Tsay HS, Wu HK, Chen LJ (1997) Isolation of cDNA clones for genes that are specifically expressed in the rice embryo. Bot Bull Acad Sin 38:13–20
Chen PW, Lu CA, Yu TS, Tseng TH, Wang CS, Yu SM (2002) Rice alpha-amylase transcriptional enhancers direct multiple mode regulation of promoters in transgenic rice. J Biol Chem 277(16):13641–13649
Chiang CM, Yeh FS, Huang LF, Tseng TH, Chung MC, Wang CS, Lur HS, Shaw JF, Yu SM (2005) Expression of a bi-functional and thermostable amylopullulanase in transgenic rice seeds leads to autohydrolysis and altered composition of starch. Mol Breed 15:125–143
Christensen AH, Quail PH (1996) Ubiquitin promoter-based vectors for high-level expression of selectable and/or screenable marker genes in monocotyledonous plants. Transgenic Res 5(3):213–218
Cobbett C, Goldsbrough P (2002) Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53:159–182
Coyle P, Philcox JC, Carey LC, Rofe AM (2002) Metallothionein: the multipurpose protein. Cell Mol Life Sci 59(4):627–647
Curie C, McCormick S (1997) A strong inhibitor of gene expression in the 5′ untranslated region of the pollen-specific LAT59 gene to tomato. Plant Cell 9(11):2025–2036
Dhadi SR, Deshpande A, Driscoll K, Ramakrishna W (2013) Major cis-regulatory elements for rice bidirectional promoter activity reside in the 5′-untranslated regions. Gene. doi:10.1016/j.gene.2013.05.060
Dickey LF, Gallo-Meagher M, Thompson WF (1992) Light regulatory sequences are located within the 5′ portion of the Fed-1 message sequence. EMBO J 11(6):2311–2317
Dickey LF, Petracek ME, Nguyen TT, Hansen ER, Thompson WF (1998) Light regulation of Fed-1 mRNA requires an element in the 5′ untranslated region and correlates with differential polyribosome association. Plant Cell 10(3):475–484
Duan X, Li X, Xue Q, Abo-el-Saad M, Xu D, Wu R (1996) Transgenic rice plants harboring an introduced potato proteinase inhibitor II gene are insect resistant. Nat Biotechnol 14(4):494–498
Feinberg AP, Vogelstein B (1983) A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132(1):6–13. doi:0003-2697(83)90418-9
Fiume E, Christou P, Giani S, Breviario D (2004) Introns are key regulatory elements of rice tubulin expression. Planta 218(5):693–703. doi:10.1007/s00425-003-1150-0
Freisinger E (2009) Metallothioneins in plants. Metal Ions Life Sci 5:107–153
Freisinger E (2011) Structural features specific to plant metallothioneins. J Biol Inorg Chem 16(7):1035–1045. doi:10.1007/s00775-011-0801-z
Gallie DR, Sleat DE, Watts JW, Turner PC, Wilson TM (1987) The 5′-leader sequence of tobacco mosaic virus RNA enhances the expression of foreign gene transcripts in vitro and in vivo. Nucleic Acids Res 15(8):3257–3273
Gallie DR, Walbot V (1992) Identification of the motifs within the tobacco mosaic virus 5′-leader responsible for enhancing translation. Nucleic Acids Res 20(17):4631–4638
Grill E, Winnacker EL, Zenk MH (1987) Phytochelatins, a class of heavy-metal-binding peptides from plants, are functionally analogous to metallothioneins. Proc Natl Acad Sci USA 84:439–443
Gutierrez RA, MacIntosh GC, Green PJ (1999) Current perspectives on mRNA stability in plants: multiple levels and mechanisms of control. Trends Plant Sci 4(11):429–438. doi:S1360-1385(99)01484-3
Hassinen VH, Tervahauta AI, Schat H, Karenlampi SO (2011) Plant metallothioneins–metal chelators with ROS scavenging activity? Plant Biol (Stuttg) 13(2):225–232. doi:10.1111/j.1438-8677.2010.00398.x
Ho SL, Tong WF, Yu SM (2000) Multiple mode regulation of a cysteine proteinase gene expression in rice. Plant Physiol 122(1):57–66
Hong CY, Cheng KJ, Tseng TH, Wang CS, Liu LF, Yu SM (2004) Production of two highly active bacterial phytases with broad pH optima in germinated transgenic rice seeds. Transgenic Res 13(1):29–39
Hood EE, Helmer GL, Fraley RT, Chilton MD (1986) The hypervirulence of Agrobacterium tumefaciens A281 is encoded in a region of pTiBo542 outside of T-DNA. J Bacteriol 168(3):1291–1301
Hsieh HM, Liu WK, Chang A, Huang PC (1996) RNA expression patterns of a type 2 metallothionein-like gene from rice. Plant Mol Biol 32(3):525–529
Hsieh HM, Liu WK, Huang PC (1995) A novel stress-inducible metallothionein-like gene from rice. Plant Mol Biol 28(3):381–389
Hua XJ, Van de Cotte B, Van Montagu M, Verbruggen N (2001) The 5′ untranslated region of the At-P5R gene is involved in both transcriptional and post-transcriptional regulation. Plant J 26(2):157–169. (tpj1020)
Hulzink RJ, Weerdesteyn H, Croes AF, Gerats T, van Herpen MM, van Helden J (2003) In silico identification of putative regulatory sequence elements in the 5′-untranslated region of genes that are expressed during male gametogenesis. Plant Physiol 132(1):75–83. doi:10.1104/pp.102.014894
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6(13):3901–3907
Kagi JH, Hunziker P (1989) Mammalian metallothionein. Biol Trace Elem Res 21:111–118
Kagi JH, Schaffer A (1988) Biochemistry of metallothionein. Biochemistry 27(23):8509–8515
Kawaguchi R, Bailey-Serres J (2005) mRNA sequence features that contribute to translational regulation in Arabidopsis. Nucleic Acids Res 33(3):955–965
Kumar G, Kushwaha HR, Panjabi-Sabharwal V, Kumari S, Joshi R, Karan R, Mittal S, Singla-Pareek SL, Pareek A (2012) Clustered metallothionein genes are co-regulated in rice and ectopic expression of OsMT1e-P confers multiple abiotic stress tolerance in tobacco via ROS scavenging. BMC Plant Biol 12(1):107. doi:1471-2229-12-107
Lane B, Kajioka R, Kennedy T (1987) The wheat-germ Ec protein is a zinc-containing metallothionein. Biochem Cell Biol 65:1001–1005
LaVaute T, Smith S, Cooperman S, Iwai K, Land W, Meyron-Holtz E, Drake SK, Miller G, Abu-Asab M, Tsokos M, Switzer R 3rd, Grinberg A, Love P, Tresser N, Rouault TA (2001) Targeted deletion of the gene encoding iron regulatory protein-2 causes misregulation of iron metabolism and neurodegenerative disease in mice. Nat Genet 27(2):209–214. doi:10.1038/84859
Lee KW, Chen PW, Lu CA, Chen S, Ho TH, Yu SM (2009) Coordinated responses to oxygen and sugar deficiency allow rice seedlings to tolerate flooding. Sci Signal 2(91):ra61. doi:2/91/ra61
Lin Z, Li WH (2012) Evolution of 5′ untranslated region length and gene expression reprogramming in yeasts. Mol Biol Evol 29(1):81–89. doi:msr143
Liu WX, Liu HL, Chai ZJ, Xu XP, Song YR, le Qu Q (2010) Evaluation of seed storage-protein gene 5′ untranslated regions in enhancing gene expression in transgenic rice seed. TAG Theoretical and applied genetics Theoretische und angewandte Genetik 121(7):1267–1274. doi:10.1007/s00122-010-1386-6
Lu CA, Lim EK, Yu SM (1998) Sugar response sequence in the promoter of a rice alpha-amylase gene serves as a transcriptional enhancer. J Biol Chem 273(17):10120–10131
Margoshes M, Vallee BL (1957) A cadmium protein from equine kidney cortex. J Am Chem Soc 79:4813–4814
Matsumura H, Nirasawa S, Terauchi R (1999) Technical advance: transcript profiling in rice (Oryza sativa L.) seedlings using serial analysis of gene expression (SAGE). Plant J 20(6):719–726
McElroy D, Blowers AD, Jenes B, Wu R (1991) Construction of expression vectors based on the rice actin 1 (Act1) 5′ region for use in monocot transformation. Mol Gen Genet 231(1):150–160
McElroy D, Brettell RIS (1994) Foreign gene expression in transgenic cereals. Trends Biotechnol 12:62–68
McElroy D, Zhang W, Cao J, Wu R (1990) Isolation of an efficient actin promoter for use in rice transformation. Plant Cell 2(2):163–171
Nickelsen J, Fleischmann M, Boudreau E, Rahire M, Rochaix JD (1999) Identification of cis-acting RNA leader elements required for chloroplast psbD gene expression in Chlamydomonas. Plant Cell 11(5):957–970
Palmiter RD (1998) The elusive function of metallothioneins. Proc Natl Acad Sci USA 95(15):8428–8430
Ren Y, Zhao J (2009) Functional analysis of the rice metallothionein gene OsMT2b promoter in transgenic Arabidopsis plants and rice germinated embryos. Plant Sci 176(4):528–538
Resch AM, Ogurtsov AY, Rogozin IB, Shabalina SA, Koonin EV (2009) Evolution of alternative and constitutive regions of mammalian 5′UTRs. BMC Genom 10:162. doi:1471-2164-10-162
Rogers JT, Randall JD, Cahill CM, Eder PS, Huang X, Gunshin H, Leiter L, McPhee J, Sarang SS, Utsuki T, Greig NH, Lahiri DK, Tanzi RE, Bush AI, Giordano T, Gullans SR (2002) An iron-responsive element type II in the 5′-untranslated region of the Alzheimer’s amyloid precursor protein transcript. J Biol Chem 277(47):45518–45528. doi:10.1074/jbc.M207435200
Schledzewski K, Mendel RR (1994) Quantitative transient gene expression: comparison of the promoters for maize polyubiquitin1, rice actin1, maize-derived Emu and CaMV 35S in cells of barley, maize and tobacco. Transgenic Res 3:249–255
Steffens B, Sauter M (2009) Epidermal cell death in rice is confined to cells with a distinct molecular identity and is mediated by ethylene and H2O2 through an autoamplified signal pathway. Plant Cell 21(1):184–196. doi:tpc.108.061887
Sutliff TD, Huang N, Litts JC, Rodriguez RL (1991) Characterization of an alpha-amylase multigene cluster in rice. Plant Mol Biol 16(4):579–591
Thomson AM, Rogers JT, Leedman PJ (2000) Thyrotropin-releasing hormone and epidermal growth factor regulate iron-regulatory protein binding in pituitary cells via protein kinase C-dependent and -independent signaling pathways. J Biol Chem 275(41):31609–31615. doi:10.1074/jbc.M002354200
Thornalley PJ, Vasak M (1985) Possible role for metallothionein protection against reaction induced oxidative stress: kinetics and mechanism of its reaction with superoxide and hydroxyl radicals. Biochem Biophys Acta 827:36–44
Toki S (1997) Rapid and efficient Agrobacterium-mediated transformation in rice. Plant Mol Biol Rep 5:16–21
Turner RL, Glynn M, Taylor SC, Cheung MK, Spurr C, Twell D, Foster GD (1999) Analysis of a translational enhancer present within the 5′-terminal sequence of the genomic RNA of potato virus S. Arch Virol 144(7):1451–1461
Umemura T, Perata P, Futsuhara Y, Yamaguchi J (1998) Sugar sensing and alpha-amylase gene repression in rice embryos. Planta 204(4):420–428
Venti A, Giordano T, Eder P, Bush AI, Lahiri DK, Greig NH, Rogers JT (2004) The integrated role of desferrioxamine and phenserine targeted to an iron-responsive element in the APP-mRNA 5′-untranslated region. Ann NY Acad Sci 1035:34–48. doi:1035/1/34
Wang CS, Vodkin LO (1994) Extraction of RNA from tissue containing high levels of procyannidins that bind RNA. Plant Mol Biol Rep 12(2):132–145
Wever W, McCallum EJ, Chakravorty D, Cazzonelli CI, Botella JR (2010) The 5′ untranslated region of the VR-ACS1 mRNA acts as a strong translational enhancer in plants. Transgenic Res 19(4):667–674. doi:10.1007/s11248-009-9332-6
Wilmink A, van de Ven BC, Dons JJ (1995) Activity of constitutive promoters in various species from the Liliaceae. Plant Mol Biol 28(5):949–955
Wong HL, Sakamoto T, Kawasaki T, Umemura K, Shimamoto K (2004) Down-regulation of metallothionein, a reactive oxygen scavenger, by the small GTPase OsRac1 in rice. Plant Physiol 135(3):1447–1456. doi:10.1104/pp.103.036384
Yu SM, Kuo YH, Sheu G, Sheu YJ, Liu LF (1991) Metabolic derepression of alpha-amylase gene expression in suspension-cultured cells of rice. J Biol Chem 266(31):21131–21137
Yuan J, Chen D, Ren Y, Zhang X, Zhao J (2008) Characteristic and expression analysis of a metallothionein gene, OsMT2b, down-regulated by cytokinin suggests functions in root development and seed embryo germination of rice. Plant Physiol 146(4):1637–1650. doi:pp.107.110304
Zhou G, Xu Y, Li J, Yang L, Liu JY (2006) Molecular analyses of the metallothionein gene family in rice (Oryza sativa L.). J Biochem Mol Biol 39(5):595–606
Zou Z, Eibl C, Koop HU (2003) The stem-loop region of the tobacco psbA 5′UTR is an important determinant of mRNA stability and translation efficiency. Mol Genet Genomics 269(3):340–349. doi:10.1007/s00438-003-0842-2
Zumbrennen KB, Wallander ML, Romney SJ, Leibold EA (2009) Cysteine oxidation regulates the RNA-binding activity of iron regulatory protein 2. Mol Cell Biol 29(8):2219–2229. doi:MCB.00004-09
Acknowledgments
We thank Dr. Pei-Yin Wu for critical review of the manuscript. We also thank Dr. Maurice SB Ku for scientific discussions and valuable comments. This work was supported by grants from the National Sciences Council (NSC95-2311-B-415-004 and NSC96-2311-B-415 -003-MY3) of the Republic of China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Jones.
Electronic supplementary material
Below is the link to the electronic supplementary material.
299_2013_1555_MOESM2_ESM.jpg
Supplementary material 2 (JPEG 1177 kb) Histochemical staining analysis of OsMT2b::GUS fusion gene in independent transgenic rice lines during seed germination and later stage of seedling growth and development. a. T2 seeds of homozygous transgenic lines MT-2-1, MT-5-1, and MT-7-1 carrying the OsMT2b::GUS gene were germinated and grown for 6 days and stained for GUS activity. b. T2 seeds of transgenic line MT-2-1 were germinated and grown for 10, 15, and 20 days and stained for GUS activity
299_2013_1555_MOESM3_ESM.jpg
Supplementary material 3 (JPEG 743 kb) Identification of the 5′-end of OsMT2b mRNA by primer extension analysis. Rice Seeds were germinated and grown in a 14-hour light/10-hour dark cycle at 28℃ for 6 days. Total RNA was purified from the embryo and subjected to primer extension analysis. A 25-nt MT-PE primer was 5′-end labeled with γ-32P ATP and used to synthesize cDNA fragment from total RNA isolated from germinated rice embryo. The extended products were electrophoresed and the resulting autoradiograph was shown. Lanes C, T, A, G provide the sequence ladders of the 5′-flanking region of OsMT2b cDNA using the MT-PE primer as a sequencing primer. The transcription start sites are indicated by the bold letters
299_2013_1555_MOESM4_ESM.jpg
Supplementary material 4 (JPEG 780 kb) The activity of αAmy8-ubi(In) promoter is much higher than the αAmy8-mt(In) promoter in rice embryo. a. Schematic representation of constructs for αAmy8-mt(In) and αAmy8-ubi(In) promoter analysis. b. Transient expression assay of luciferase activity in rice embryos. Rice embryos were transfetced with plasmids pαAmy8(+mt intron1)-Luc and pαAmy8(+ubi intron1)-Luc by particle bombardment. Transfected embryos were incubated at 28℃ for 24 h in the MS liquid medium lacking sucrose, and then pooled for luciferase activity assays. The luciferase activity reading obtained from αAmy8-ubi(In) were normalized to the reading obtained from αAmy8-mt(In), which was set to 1X. Error bars indicate the standard deviation of three replicate experiments for each construct
Rights and permissions
About this article
Cite this article
Wu, CS., Chen, DY., Chang, CF. et al. The promoter and the 5′-untranslated region of rice metallothionein OsMT2b gene are capable of directing high-level gene expression in germinated rice embryos. Plant Cell Rep 33, 793–806 (2014). https://doi.org/10.1007/s00299-013-1555-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-013-1555-6