Abstract
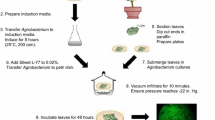
In this study, we present a method for transient expression of the type III effector AvrGf1 from Xanthomonas citri subsp. citri strain Aw in grapefruit leaves (Citrus paradisi) via Agrobacterium tumefaciens. The coding sequence of avrGf1 was placed under the control of the constitutive CaMV 35S promoter in the binary vectors pGWB2 and pGWB5. Infiltration of grapefruit leaves with A. tumefaciens carrying these constructs triggered a hypersensitive response (HR) in grapefruit 4 days after inoculation. When transiently expressed in grapefruit leaves, two mutants, AvrGf1ΔN116 and AvrGf1ΔC83, failed to induce an HR. Moreover, using bioinformatics tools, a chloroplast transit signal was predicted at the N terminus of AvrGf1. We demonstrated chloroplast localization by using an AvrGf1::GFP fusion protein, where confocal images revealed that GFP fluorescence was accumulating in the stomatal cells that are abundant in chloroplasts. Transient expression in citrus has the potential for aiding in the development of new disease defense strategies in citrus.



Similar content being viewed by others
References
Abramovitch RB, Kim YJ, Chen S, Dickman MB, Martin GB (2003) Pseudomonas type III effector AvrPtoB induces plant disease susceptibility by inhibition of host programmed cell death. EMBO J 22:60–69
Ahmad M, Mirza B (2005) An efficient protocol for transient transformation of intact fruit and transgene expression in Citrus. Plant Molecular Biology Reporter 23:419a–419k
An G (1987) Binary Ti vectors for plant transformation and promoter analysis. Methods Enzymol 153:292–305
Bespalhok JCF, Kobayashi AK, Pereira LFP, Galvão RM, Vieira LGE (2003) Transient gene expression of β-glucuronidase in citrus thin epicotyl transversal sections using particle bombardment. Braz Arch Biol Technol 46:1–6
Bhaskar PB, Venkateshwaran M, Wu L, Ane JM, Jiang J (2009) Agrobacterium-mediated transient gene expression and silencing: a rapid tool for functional gene assay in potato. PLoS One 6:e5812
Bonas U, Van den Ackerveken G (1999) Gene-for-gene interactions: bacterial avirulence proteins specify plant disease resistance. Curr Opin Microbiol 2:94–98
Brunings AM, Gabriel DW (2003) Xanthomonas citri: breaking the surface. Mol Plant Pathol 4:141–157
Büttner D, Bonas U (2010) Regulation and secretion of Xanthomonas virulence factors. FEMS Microbiol Rev 2:107–133
Catanzariti A, Dodds PN, Ve T, Kobe B, Ellis JG, Staskawicz BJ et al (2010) The AvrM effector from flax rust has a structured C-terminal domain and interacts directly with the M resistance protein. Mol Plant Microbe Interact 23:49–57
Cheng YQ, Yang J, Xu FP, An LJ, Liu JF, Chen ZW (2009) Transient expression of minimum linear gene cassettes in onion epidermal cells via direct transformation. Appl Biochem Biotechnol 159:739–749
Chisholm ST, Coaker G, Day B, Staskawicz BJ (2006) Host–microbe interactions: shaping the evolution of the plant immune response. Cell 124:803–814
Duan YP, Castañeda A, Zhao G, Erdos G, Gabriel DW (1999) Expression of a single, host-specific, bacterial pathogenicity gene in plant cells elicits division, enlargement, and cell death. Mol Plant Microbe Interact 12:556–560
Emanuelsson O, Nielsen H, von Heijne G (1999) ChloroP, a neural network-based method for predicting chloroplast transit peptides and their cleavage sites. Protein Sci 8:978–984
Folimonov AS, Folimonova SY, Bar-Joseph M, Dawson WO (2007) A stable RNA virus-based vector for citrus trees. Virology 368:205–216
Green BJ, Fujiki M, Mett V, Kaczmarczyk J, Shamloul M, Musiychuk K (2009) Transient protein expression in three Pisum sativum (green pea) varieties. Biotechnol J 4:230–237
Gurlebeck D, Jahn S, Gurlebeck N, Szczesny R, Szurek B, Hahn S (2009) Visualization of novel virulence activities of the Xanthomonas type III effectors AvrBs1, AvrBs3 and AvrBs4. Mol Plant Pathol 10:175–188
Guttman DS, Vinatzer BA, Sarkar SF, Ranall MV, Kettler G, Greenberg JT (2002) A functional screen for the type III (hrp) secretome of the plant pathogen Pseudomonas syringae. Science 295:1722–1726
Jelenska J, Yao N, Vinatzer BA, Wright CM, Brodsky JL, Greenberg JT (2007) A J domain virulence effector of Pseudomonas syringae remodels host chloroplasts and suppresses defenses. Curr Biol 17:499–508
Joensuu JJ, Conley AJ, Lienemann M, Brandle JE, Linder MB, Menassa R (2010) Hydrophobin fusions for high-level transient protein expression and purification in Nicotiana benthamiana. Plant Physiol 152:622–633
Johansen LK, Carrington JC (2001) Silencing on the spot induction and suppression of RNA silencing in the Agrobacterium-mediated transient expression system. Plant Physiol 126:930–938
Jones HD, Doherty A, Wu H (2005) Review of methodologies and a protocol for the Agrobacterium-mediated transformation of wheat. Plant Methods 1:5
Jones HD, Doherty A, Sparks CA (2009) Transient transformation of plants. Methods Mol Biol 513:131–152
Klement Z (1963) Rapid detection of the pathogenicity of phytopathogenic pseudomonads. Nature 199:299–300
Li JF, Park E, von Arnim AG, Nebenfuhr A (2009) The FAST technique: a simplified Agrobacterium-based transformation method for transient gene expression analysis in seedlings of Arabidopsis and other plant species. Plant Methods 5:6
Manavella PA, Chan RL (2009) Transient transformation of sunflower leaf discs via an Agrobacterium-mediated method: applications for gene expression and silencing studies. Nat Protoc 4:1699–1707
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nair R, Rost B (2005) Mimicking cellular sorting improves prediction of subcellular localization. J Mol Biol 348:85–100
Nakagawa T, Kurose T, Hino T, Tanaka K, Kawamukai M, Niwa Y (2007) Development of series of gateway binary vectors, pGWBs, for realizing efficient construction of fusion genes for plant transformation. J Biosci Bioeng 104:34–41
Porta C, Lomonossoff GP (1996) Use of viral replicons for the expression of genes in plants. Mol Biotechnol 5:209–221
Rybak M, Minsavage GV, Stall RE, Jones JB (2009) Identification of Xanthomonas citri ssp. citri host specificity genes in a heterologous expression host. Mol Plant Microbe Interact 2:249–262
Sambrook J, Fristsch E, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, NY
Santos-Rosa M, Poutaraud A, Merdinoglu D, Mestre P (2008) Development of a transient expression system in grapevine via agro-infiltration. Plant Cell Rep 27:1053–1063
Schein AI, Kissinger JC, Ungar LH (2001) Chloroplast transit peptide prediction: a peek inside the black box. Nucleic Acids Res 29:E82
Sparkes IA, Runions J, Kearns A, Hawes C (2006) Rapid, transient expression of fluorescent fusion proteins in tobacco plants and generation of stably transformed plants. Nat Protoc 1:2019–2025
Van Larebeke N, Engler G, Holsters M, Van den Elsacker S, Zaenen I, Schilperoort RA, Schell J (1974) Large plasmid in Agrobacterium tumefaciens essential for crown gall-inducing ability. Nature 252:169–170
Yang Y, Li R, Qi M (2000) In vivo analysis of plant promoters and transcription factors by agroinfiltration of tobacco leaves. Plant J 22:543–551
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Merkle.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Figueiredo, J.F.L., Römer, P., Lahaye, T. et al. Agrobacterium-mediated transient expression in citrus leaves: a rapid tool for gene expression and functional gene assay. Plant Cell Rep 30, 1339–1345 (2011). https://doi.org/10.1007/s00299-011-1045-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-011-1045-7