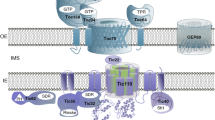
Abstract
Aminoacyl-tRNA synthetases (AARSs) play a critical role in translation and are thus required in three plant protein-synthesizing compartments: cytosol, mitochondria and plastids. A systematic study had previously shown extensive sharing of organellar AARSs from Arabidopsis thaliana, mostly between mitochondria and chloroplasts. However, distribution of AARSs from monocot species, such as maize, has never been experimentally investigated. Here we demonstrate dual targeting of maize seryl-tRNA synthetase, SerZMo, into both mitochondria and chloroplasts using combination of complementary methods, including in vitro import assay, transient expression analysis of green fluorescent protein (GFP) fusions and immunodetection. We also show that SerZMo dual localization is established by the virtue of an ambiguous targeting peptide. Full-length SerZMo protein fused to GFP is targeted to chloroplast stromules, indicating that SerZMo protein performs its function in plastid stroma. The deletion mutant lacking N-terminal region of the ambiguous SerZMo targeting peptide was neither targeted into mitochondria nor chloroplasts, indicating the importance of this region in both mitochondrial and chloroplastic import.




Similar content being viewed by others
Abbreviations
- AARS:
-
Aminoacyl-tRNA synthetase
- SerRS:
-
Seryl-tRNA synthetase
- GFP:
-
Green fluorescent protein
References
Ambroggio EE, Austen B, Fidelio GD (2007) Biophysical properties of a synthetic transit peptide from wheat chloroplast ribulose 1,5-bisphosphate carboxylase. J Pept Sci 13:245–252
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Beardslee TA, Roy-Chowdhury S, Jaiswal P, Buhot L, Lerbs-Mache S, Stern DB, Allison LA (2002) A nucleus-encoded maize protein with sigma factor activity accumulates in mitochondria and chloroplasts. Plant J 31:199–209
Berg M, Rogers R, Muralla R, Meinke D (2005) Requirement of aminoacyl-tRNA synthetases for gametogenesis and embryo development in Arabidopsis. Plant J 44:866–878
Bhushan S, Kuhn C, Berglund AK, Roth C, Glaser E (2006) The role of the N-terminal domain of chloroplast targeting peptides in organellar protein import and miss-sorting. FEBS Lett 580:3966–3972
Bruce BD (2000) Chloroplast transit peptides: structure, function and evolution. Trends Cell Biol 10:440–447
Bruce BD, Perry S, Froelich J, Keegstra K (1994) In vitro import of proteins into chloroplasts. In: Gelvin SB, Schilferoot RA (eds) Plant molecular biology manual. Kluwer, Dordrecht, pp J1:1–15
Chen MH, Huang LF, Li HM, Chen YR, Yu SM (2004) Signal peptide-dependent targeting of a rice alpha-amylase and cargo proteins to plastids and extracellular compartments of plant cells. Plant Physiol 135:1367–1377
Chew O, Rudhe C, Glaser E, Whelan J (2003) Characterization of the targeting signal of dual-targeted pea glutathione reductase. Plant Mol Biol 53:341–356
Chigri F, Soll J, Vothknecht UC (2005) Calcium regulation of chloroplast protein import. Plant J 42:821–831
Chiu W, Niwa Y, Zeng W, Hirano T, Kobayashi H, Sheen J (1996) Engineered GFP as a vital reporter in plants. Curr Biol 6:325–330
Cho HS, Lee SS, Kim KD, Hwang I, Lim JS, Park YI, Pai HS (2004) DNA gyrase is involved in chloroplast nucleoid partitioning. Plant Cell 16:2665–2682
Combet C, Blanchet C, Geourjon C, Deléage G (2000) NPS@: network protein sequence analysis. Trends Biochem Sci 25:147–150
Duchene AM, Giritch A, Hoffmann B, Cognat V, Lancelin D, Peeters NM, Zaepfel M, Marechal-Drouard L, Small ID (2005) Dual targeting is the rule for organellar aminoacyl-tRNA synthetases in Arabidopsis thaliana. Proc Natl Acad Sci USA 102:16484–16489
Emanuelsson O, Brunak S, von Heijne G, Nielsen H (2007) Locating proteins in the cell using TargetP, SignalP, and related tools. Nat Protoc 2:953–971
Fulgosi H, Soll J (2002) The chloroplast protein import receptors Toc34 and Toc159 are phosphorylated by distinct protein kinases. J Biol Chem 277:8934–8940
Glaser E, Sjoling S, Tanudji M, Whelan J (1998) Mitochondrial protein import in plants. Signals, sorting, targeting, processing and regulation. Plant Mol Biol 38:311–338
Hörmann F, Soll J, Bölter B (2007) The chloroplast protein import machinery: a review. Methods Mol Biol 390:179–194
Ibba M, Söll D (2000) Aminoacyl-tRNA synthesis. Annu Rev Biochem 69:617–650
Kim YK, Lee JY, Cho HS, Lee SS, Ha HJ, Kim S, Choi D, Pai HS (2005) Inactivation of organellar glutamyl- and seryl-tRNA synthetases leads to developmental arrest of chloroplasts and mitochondria in higher plants. J Biol Chem 280:37098–37106
Kwok EY, Hanson MR (2004) Stromules and the dynamic nature of plastid morphology. J Microsc 214:124–137
Lister R, Chew O, Lee MN, Heazlewood JL, Clifton R, Parker KL, Millar AH, Whelan J (2004) A transcriptomic and proteomic characterization of the Arabidopsis mitochondrial protein import apparatus and its response to mitochondrial dysfunction. Plant Physiol 134:777–789
Mackenzie SA (2005) Plant organellar protein targeting: a traffic plan still under construction. Trends Cell Biol 15:548–554
Moberg P, Nilsson S, Stahl A, Eriksson AC, Glaser E, Maler L (2004) NMR solution structure of the mitochondrial F1beta presequence from Nicotiana plumbaginifolia. J Mol Biol 336:1129–1140
Mocibob M, Weygand-Durasevic I (2008) The proximal region of a noncatalytic eukaryotic seryl-tRNA synthetase extension is required for protein stability in vitro and in vivo. Arch Biochem Biophys 470:129–138
Nakai K, Horton P (1999) PSORT: a program for detecting the sorting signals of proteins and predicting their subcellular localization. Trends Biochem Sci 24:34–35
Natesan SK, Sullivan JA, Gray JC (2005) Stromules: a characteristic cell-specific feature of plastid morphology. J Exp Bot 56:787–797
Neuburger M, Journet EP, Bligny R, Carde JP, Douce R (1982) Purification of plant mitochondria by isopycnic centrifugation in density gradients of Percoll. Arch Biochem Biophys 217:312–323
Neupert W, Herrmann JM (2007) Translocation of proteins into mitochondria. Annu Rev Biochem 76:723–49
Peeters N, Small I (2001) Dual targeting to mitochondria and chloroplasts. Biochim Biophys Acta 1541:54–63
Peltier JB, Cai Y, Sun Q, Zabrouskov V, Giacomelli L, Rudella A, Ytterberg AJ, Rutschow H, van Wijk KJ (2006) The oligomeric stromal proteome of Arabidopsis thaliana chloroplasts. Mol Cell Proteomics 5:114–133
Petsalaki EI, Bagos PG, Litou ZI, Hamodrakas SJ (2006) PredSL: a tool for the N-terminal sequence-based prediction of protein subcellular localization. Genomics Proteomics Bioinformatics 4:48–55
Pujol C, Marechal-Drouard L, Duchene AM (2007) How can organellar protein N-terminal sequences be dual targeting signals? In silico analysis and mutagenesis approach. J Mol Biol 369:356–367
Regev-Rudzki N, Pines O (2007) Eclipsed distribution: a phenomenon of dual targeting of protein and its significance. Bioessays 29:772–782
Rokov J, Soll D, Weygand-Durasevic I (1998) Maize mitochondrial seryl-tRNA synthetase recognizes Escherichia coli tRNA(Ser) in vivo and in vitro. Plant Mol Biol 38:497–502
Rokov-Plavec J, Lesjak S, Landeka I, Mijakovic I, Weygand-Durasevic I (2002) Maize seryl-tRNA synthetase: specificity of substrate recognition by the organellar enzyme. Arch Biochem Biophys 397:40–50
Rudhe C, Clifton R, Whelan J, Glaser E (2002) N-terminal domain of the dual-targeted pea glutathione reductase signal peptide controls organellar targeting efficiency. J Mol Biol 324:577–585
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, pp A8.52–A8.55
Silva-Filho MC (2003) One ticket for multiple destinations: dual targeting of proteins to distinct subcellular locations. Curr Opin Plant Biol 6:589–595
Small I, Peeters N, Legeai F, Lurin C (2004) Predotar: a tool for rapidly screening proteomes for N-terminal targeting sequences. Proteomics 4:1581–1590
Tanudji M, Sjoling S, Glaser E, Whelan J (1999) Signals required for the import and processing of the alternative oxidase into mitochondria. J Biol Chem 274:1286–1293
Wischmann C, Schuster W (1995) Transfer of rps10 from the mitochondrion to the nucleus in Arabidopsis thaliana: evidence for RNA-mediated transfer and exon shuffling at the integration site. FEBS Lett 374:152–156
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572
Acknowledgments
We thank Claire Pujol for initial help with in vitro import studies, Dr Igor Weber for inital help with confocal microscopy and Dr Hrvoje Fulgosi for the gift of pTH-2 plasmid. J.R.P thanks Dr Laurence Marechal-Drouard for hospitality. Visit of J.R.P to Strasbourg was supported by short-term scholarship of the French government and Croatian Ministry of Science, Education and Sports. This work was supported by grant from the Ministry of Science, Education and Sports of the Republic of Croatia (119-0982913-1358).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J.R. Liu.
Rights and permissions
About this article
Cite this article
Rokov-Plavec, J., Dulic, M., Duchêne, AM. et al. Dual targeting of organellar seryl-tRNA synthetase to maize mitochondria and chloroplasts. Plant Cell Rep 27, 1157–1168 (2008). https://doi.org/10.1007/s00299-008-0542-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-008-0542-9