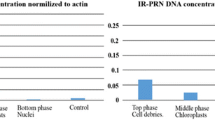
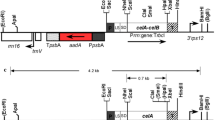
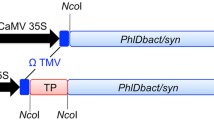
Abstract
The pathway for synthesis of polyhydroxybutyrate (PHB), a polyester produced by three bacterial enzymes, was transferred to the tobacco plastid genome by the biolistic transformation method. The polycistronic phb operon encoding this biosynthetic pathway was cloned into plastome transformation vectors. Following selection and regeneration, the content and structure of plant-produced hydroxybutyrate was analysed by gas chromatography. Significant PHB synthesis was limited to the early stages of in vitro culture. Within the transformants, PHB synthesis levels were highly variable. In the early regeneration stage, single regenerates reached up to 1.7% PHB in dry weight. At least 70% of plant-produced hydroxybutyric acid was proven to be polymer with a molecular mass of up to 2,500 kDa. PHB synthesis levels of the transplastomic lines were decreasing when grown autotrophically but their phb transcription levels remained stable. Transcription of the three genes is divided into two transcripts with phbB being transcribed separately from phbC and phbA. In mature plants even low amounts of PHB were associated with male sterility. Fertility was only observed in a mutant carrying a defective phb operon. These results prove successful expression of the entire PHB pathway in plastids, concomitant, however, with growth deficiency and male sterility.












Similar content being viewed by others
Abbreviations
- 3HB:
-
3-Hydroxybutyric acid
- d.w.:
-
Dry weight
- CoA:
-
Coenzyme A
- GC:
-
Gas chromatography
- GPC:
-
Gel permeation chromatography
- HPLC:
-
High performance liquid chromatography
- Mn:
-
Molecular number average
- Mw:
-
Molecular weight average
- nt:
-
Nucleotide
- PHA:
-
Polyhydroxyalkanoate
- PHB:
-
Polyhydroxybutyrate
- UTR:
-
Untranslated region
References
Anderson AJ, Dawes EA (1990) Occurrence, metabolism, metabolic role and industrial uses of bacterial polyhydroxy alkanoates. Microbiol Rev 54:450–472
Bohmert K, Balbo I, Kopka J, Mittendorf V, Nawrath C, Poirier Y, Tischendorf G, Trethewey R, Willmitzer L (2000) Transgenic Arabidopsis plants can accumulate polyhydroxybutyrate to up to 4% of their fresh weight. Planta 211:841–845
Bohmert K, Balbo I, Steinbuchel A, Tischendorf G, Willmitzer L (2002) Constitutive expression of the beta-ketothiolase gene in transgenic plants. A major obstacle for obtaining polyhydroxybutyrate-producing plants. Plant Physiol 128:1282–1290
Brandl H, Gross RA, Lenz RW, Fuller RC (1988) Pseudomonas oleovorans as a source of poly(ß-) hydroxyalkanoates for potential applications as biodegradable polyesters. Appl Environ Microbiol 54:1977–1982
Bruick R, Mayfield S (1999) Light-activated translation of chloroplast mRNAs. Trends Plant Sci 4:190–195
Corriveau JL, Coleman A (1988) Rapid screening method to detect potential biparental in-heritance of plastid DNA: results for over 200 angiosperm species. Am J Bot 75:1443–1458
De Cosa B, Moar W, Lee SB, Miller M, Daniell H (2001) Overexpression of the Bt cry2Aa2 operon in chloroplasts leads to formation of insecticidal crystals. Nat Biotechnol 19:71–74
Eibl C, Zhurong Z, Beck A, Minkyun K, Mullet J, Koop HU (1999) In vivo analysis of plastid psbA, rbcL and rpl32 UTR elements by chloroplast transformation: tobacco plastid gene expression is controlled by modulation of transcript levels and translation efficiency. Plant J 19:333–345
Estelle M (2001) Proteases and cellular regulation in plants. Curr Opin Plant Biol 4:254–260
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158
Hai T, Hein S, Steinbüchel A (2001) Multiple evidence for widespread and general occurrence of type-III PHA synthases in cyanobacteria and molecular characterization of the PHA synthases from two thermophilic cyanobacteria: Chlorogloeopsis fritschii PCC 6912 and Synechocystis strain MA19. Microbiology 147:3047–3060
Houmiel KL, Slater S, Broyles D, Casagrande L, Colburn S, Gonzalez K, Mitsky TA, Reiser SE, Shah D, Taylor NB, Tran M, Valentin HE, Gruys KJ (1999) Poly(beta-hydroxybutyrate) production in oilseed leucoplasts of Brassica napus. Planta 209:547–550
Huang FC, Klaus SM, Herz S, Zou Z, Koop HU, Golds TJ (2002) Efficient plastid transformation in tobacco using the aphA-6 gene and kanamycin selection. Mol Genet Genomics 268:19–27
Huffaker R (1990) Proteolytic activity during senescence of plants. New Phytol 116:199–231
Kempken F, Pring DR (1999) Male sterility in higher plants: fundamentals and applications. Prog Bot 60:139–166
Koop HU, Steinmüller K, Wagner H, Roessler C, Eibl C, Sacher L (1996) Integration of foreign sequences into the tobacco plastome via polyethylene glycol-mediated protoplast transformation. Planta 199:193–201
Kulkarni RD, Golden S (1994) Adaptation to high light intensity in Synechococcus sp. strain PCC 7942: regulation of three psbA genes and two forms of the D1 protein. J Bacteriol 176: 959–965
Lössl A, Eibl C, Dovzhenko A, Winterholler P, Koop HU (2000) Production of polyhydroxybutyric acid (PHB) using chloroplast transformation. The 8th international symposium on biological polyesters (ISBP 2000), 11 September 2000–15 September 2000, Massachusetts Institute of Technology, Cambridge, Mass., USA
Menzel G, Harloff HJ, Jung C (2002) Expression of bacterial poly(3-hydroxybutyrate) synthesis genes in hairy roots of sugar beet (Beta vulgaris L.) Appl Microbiol Biotechnol (in press)
Nakashita H, Arai Y, Yoshioka K, Fukui T, Doi Y, Usami R, Horikoshi K, Yamaguchi I (1999) Production of biodegradable polyester by a transgenic tobacco. Biosci Biotechnol Biochem 63:870–874
Nakashita H, Arai Y, Shikanai T, Doi Y, Yamaguchi I (2001) Introduction of bacterial metabolism into higher plants by polycistronic transgene expression. Biosci Biotechnol Biochem 65:1688–1691
Nawrath C, Poirier Y, Somerville C (1994) Targeting of the hydroxybutyrate biosynthetic pathway to the plastids of A. thaliana results in high levels of polymer accumulation. Proc Natl Acad Sci USA 91:12760–12764
Oelmüller UN, Krüger N, Steinbüchel A and Friedrich C (1990) Isolation of prokaryotic RNA and detection of specific mRNA with biotinylated probes. J Microbiol Methods 11:73–81
Peoples OP, Sinskey AJ (1989) Poly-beta-hydroxybutyrate PHB biosynthesis in Alcaligenes eutrophus H16. Identification and characterization of the PHB polymerase gene phbC. J Biol Chem 264:15298–15303
Poirier Y (1999) Production of new polymeric compounds in plants. Curr Opin Biotechnol 10:181–185
Poirier Y, Dennis DE, Klomparens K, Somerville C (1992) Polyhydroxybutyrate, a biodegradable thermoplastic in transgenic plants. Science 256:520–523
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Saruul P, Srienc F, Somers DA, Samac DA (2002) Production of a biodegradable plastic polymer, poly-ß-hydroxybutyrate, in transgenic alfalfa. Crop Sci 42:919–927
Schubert P, Steinbüchel A, Schlegel HG (1988) Cloning of the A. eutrophus genes for synthesis of poly-ß-hydroxybutyric acid (PHB) and synthesis of PHB in E. coli. J Bacteriol 170:5837–5847
Seebach D, Fritz M (1999) Detection, synthesis, structure, function of oligo(3-hydroxyalkanoates): contributions by synthetic organic chemists. Int J Biol Macromol 25:217–236
Shinozaki K, Ohme M, Tanaka M, Wakasugi T, Hayashida N, Matsubayashi T, Zaita N, Chunwongse J, Obokata J, Yamaguchi-Shinozaki K, Ohto C, Torazawa K, Meng BY, Sugita M, Deno H, Kamogashira T, Yamada K, Kusuda J, Takaiwa F, Kato A, Tohdoh N, Shimada H, Sugiura M (1986) The complete nucleotide sequence of the tobacco chloroplast genome: its gene organization and expression. EMBO J 5:2043–2049
Slater SC, Voige WH, Dennis DE (1988) Cloning and expression in E. coli of the A. eutrophus H16 poly-ß-hydroxybutyrate biosynthetic pathway. J Bacteriol 170:4431–4436
Staub JM, Garcia B, Graves J, Hajdukiewicz PTJ, Hunter P, Nehra N, Paradkar V, Schlittler M, Carroll JA, Spatola L, Ward D, Ye G, Russell DA (2000) High-yield production of a human therapeutic protein in tobacco chloroplasts. Nat Biotechnol 18:333–338
Stern DB, Higgs DC, Yang J (1997) Transcription and translation in chloroplasts. Trends Plant Sci 2:308–315
Svab Z, Hajdukiewicz P, Maliga P (1990) Stable transformation of plastids in higher plants. Proc Natl Acad Sci USA 87:8526–8530
Ye GN, Hajdukiewicz PT, Broyles D, Rodriguez D, Xu CW, Nehra N, Staub JM (2001) Plastid-expressed 5-enolpyruvylshikimate-3-phosphate synthase genes provide high level glyphosate tolerance in tobacco. Plant J 25:261–270
Zou Z (2001) Analysis of cis-acting expression determinants of the tobacco psbA 5′UTR in vivo. Ph.D. thesis, Ludwig-Maximilians-Universität München, Munich
Acknowledgements
This work was supported through the Fachagentur für nachwachsende Rohstoffe (grant no. 99NR074). The excellent technical assistance by Katharina Yeiser, Stefan Kirchner and Petra Winterholler is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Lörz
Rights and permissions
About this article
Cite this article
Lössl, A., Eibl, C., Harloff, HJ. et al. Polyester synthesis in transplastomic tobacco (Nicotiana tabacum L.): significant contents of polyhydroxybutyrate are associated with growth reduction. Plant Cell Rep 21, 891–899 (2003). https://doi.org/10.1007/s00299-003-0610-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-003-0610-0