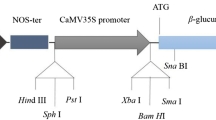
Abstract.
Transgenic basil (Ocimum spp.) was produced for the first time using an Agrobacterium tumefaciens-mediated transformation system. β-Glucuronidase (GUS) transient expression experiments showed a 96% or higher infection rate using Agrobacterium strains GV3101 and EHA105. Inoculated leaf explants were regenerated on Murashige and Skoog medium supplemented with thidiazuron, naphthaleneacetic acid and kanamycin. Two sweet basil (cvs SBA28 and Purple Ruffles, O. basilicum) and two lemon basil (cv Sweet Dani, O. citriodorum) plants were regenerated after in vitro cultivation. Stable transformation of both species was verified by polymerase chain reaction amplification (PCR) of both the gus (697 bp) and nptII (320 bp) gene fragments and by Southern blot analysis for the gus gene. Inheritance of the transgenes was verified by the GUS colorimetric assay and PCR analysis of the T1 generation of Ocimum transgenic plants, and the observed segregation ratio (3:1) indicated a single gene for GUS. Transgenic T1 plants exhibited identical phenotypic characteristics and accumulated high concentrations of linalool (118.98 µg g–1), 1.8-cineole (20.86 µg g–1), eugenol (69.3 µg g–1), neral (468.61 µg g–1) and geraniol (649.1 µg g–1) in volatile oil profiles similar to those of the non-transgenic controls.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Deschamps, .C., Simon, .J. Agrobacterium tumefaciens-mediated transformation of Ocimum basilicum and O. citriodorum . Plant Cell Rep 21, 359–364 (2002). https://doi.org/10.1007/s00299-002-0526-0
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00299-002-0526-0