Abstract
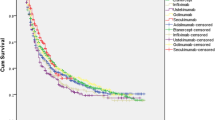
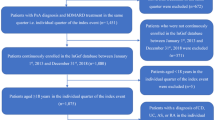
To study retention of biologic disease-modifying anti-rheumatic drugs (bDMARDs) or apremilast and potential predictors of lack of response in patients with psoriatic arthritis (PsA). A single-center retrospective analysis of PsA patients who received ≥ 1 bDMARD or apremilast during 2000–2018. The main endpoint was lack of response (primary or secondary failure). Analyses included retention of DMARDs (Kaplan–Meier curves) and potential predictors of lack of response (bivariate and multivariate logistic regression models). A total of 159 patients with PsA received up to 8 DMARDs: etanercept (34%), adalimumab (30%), infliximab (9%), golimumab (9%), apremilast (7%), ustekinumab (5%), certolizumab (4%), and secukinumab (2%). Therapy was discontinued in 96 cases (60%), mainly owing to secondary failure (37%), followed by primary failure (25%) and adverse effects (24%). Retention was analyzed based on 313 units of analysis. Duration of follow-up was 846.1 treatment-years (maximum 14.8 years, median 2.75 years). A total of 172 DMARDs were discontinued. The probability of continuing the initial treatment was 37% at 5 years, 22% at 10 years, and 12% at 14 years. The longest medium retention time was observed for infliximab (6.2 years) and etanercept (4.5 years). Predictors of lack of response included male sex, number of swollen joints, and, especially, depression (OR = 35.2). The sensitivity and specificity of the model were 86.4% and 85.7%, respectively, with a coefficient of determination (R2) of 45.6 (ROC, 0.912). Rates of discontinuation due to primary and secondary failure are high in PsA. Retention is better for anti-TNF agents than for other agents.


Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
Glintborg B, Østergaard M, Dreyer L et al (2011) Treatment response, drug survival, and predictors thereof in 764 patients with psoriatic arthritis treated with anti-tumor necrosis factor α therapy: results from the nationwide Danish DANBIO registry. Arthritis Rheum 63:382–390. https://doi.org/10.1002/art.30117
Stober C, Ye W, Guruparan T et al (2018) Prevalence and predictors of tumour necrosis factor inhibitor persistence in psoriatic arthritis. Rheumatology 57:158–163. https://doi.org/10.1093/rheumatology/kex387
Carmona L, Gómez-Reino JJ (2006) Survival of TNF antagonists in spondyloarthritis is better than in rheumatoid arthritis. Data from the Spanish registry BIOBADASER. Arthritis Res Ther 8:72. https://doi.org/10.1186/ar1941
Cañete JD, Naranjo A, Calvo J et al (2020) Biological treatment patterns in patients with inflammatory joint diseases. Retrospective study with 4 years follow-up. Reumatol Clínica Engl Ed 16:447–454. https://doi.org/10.1016/j.reumae.2018.11.016
Saad AA, Ashcroft DM, Watson KD et al (2009) Persistence with anti-tumor necrosis factor therapies in patients with psoriatic arthritis: observational study from the British Society of Rheumatology Biologics Register. Arthritis Res Ther 11:R52. https://doi.org/10.1186/ar2670
Thomas ML, Shaddick G, Charlton R et al (2020) Tumour necrosis factor inhibitor monotherapy versus combination therapy for the treatment of psoriatic arthritis: combined analysis of European biologics databases. J Rheumatol. https://doi.org/10.3899/jrheum.190815
Nissen M (2020) CO-medication with a conventional synthetic DMARD in patients with axial spondyloarthritis is associated with improved retention of TNF inhibitors: results from The Eurospa Collaboration. Ann Rheum Dis 79:71
Aaltonen K, Heinonen A, Joensuu J et al (2017) Effectiveness and drug survival of TNF-inhibitors in the treatment of psoriatic arthritis: a prospective cohort study. Semin Arthritis Rheum 46:732–739. https://doi.org/10.1016/j.semarthrit.2016.09.005
Mease PJ, Collier DH, Saunders KC et al (2015) Comparative effectiveness of biologic monotherapy versus combination therapy for patients with psoriatic arthritis: results from the CORRONA registry. RMD Open 1:e000181. https://doi.org/10.1136/rmdopen-2015-000181
Glintborg B, Østergaard M, Krogh NS et al (2013) Clinical response, drug survival, and predictors thereof among 548 patients with psoriatic arthritis who switched tumor necrosis factor α inhibitor therapy: results from the Danish Nationwide DANBIO Registry. Arthritis Rheum 65:1213–1223. https://doi.org/10.1002/art.37876
Gossec L, Siebert S, Bergmans P et al (2020) SAT0398 persistence of ustekinumab (UST) or TNF inhibitor (TNFI) treatment in psoriatic arthritis (PsA): insights from the large, prospective, multinational, real-world PsABio cohort. Ann Rheum Dis 79:1149–1150. https://doi.org/10.1136/annrheumdis-2020-eular.2127
Chimenti MS, Fonti GL, Conigliaro P et al (2020) Fri0270 one-year effectiveness, retention rate and safety of secukinumab in ankylosing spondylitis and psoriatic arthritis: a real-life multicentre study. Ann Rheum Dis 79:720–720. https://doi.org/10.1136/annrheumdis-2020-eular.3777
Favalli EG, Marchesoni A, Balduzzi S, Montecucco C, Lomater C, Crepaldi G (2020) Effectiveness and retention rate of secukinumab for psoriatic arthritis and axial spondyloarthritis: real-life data from the Italian LORHEN registry. Ann Rheum Dis 79:718
Cheila M, Douglas K, Koutsianas C (2020) Fri0280 comparative drug survival of TNF inhibitors and secukinumab in biologic naïve patients with ankylosing spondylitis and psoriatic arthritis. Ann Rheum Dis 79:727–728. https://doi.org/10.1136/annrheumdis-2020-eular.4969
Villa-Blanco I, Castro SA, Fernández S et al (2020) Fri0291 safety and survival of secukinumab in spondyloarthritis and psoriatic arthritis: real-life data. a multicenter study. Ann Rheum Dis 79:734–734. https://doi.org/10.1136/annrheumdis-2020-eular.5970
Letarouilly J-G, Flachaire B, Labadie C et al (2020) Secukinumab and ustekinumab treatment in psoriatic arthritis: results of a direct comparison. Rheumatol Oxf Engl. https://doi.org/10.1093/rheumatology/keaa710
Deza G, Notario J, Ferran M et al (2018) Long-term etanercept survival in patients with psoriatic arthritis: a multicenter retrospective analysis in daily clinical practice in Spain. Rheumatol Int 38:2037–2043. https://doi.org/10.1007/s00296-018-4144-8
Biggioggero M, Favalli EG (2014) Ten-year drug survival of anti-TNF agents in the treatment of inflammatory arthritides: anti-TNF in inflammatory arthritides. Drug Dev Res 75:S38–S41. https://doi.org/10.1002/ddr.21192
Iannone F, Salaffi F, Fornaro M et al (2018) Influence of baseline modified Rheumatic Disease Comorbidity Index (mRDCI) on drug survival and effectiveness of biological treatment in patients affected with rheumatoid arthritis, spondyloarthritis and psoriatic arthritis in real-world settings. Eur J Clin Invest 48:e13013. https://doi.org/10.1111/eci.13013
Funding
There is no financial support for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Document included.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mateo Soria, L., Prior-Español, Á., Grigorov, M.M. et al. Long-term survival of biological therapy in psoriatic arthritis: 18-year analysis of a cohort in a tertiary hospital. Rheumatol Int 42, 1043–1051 (2022). https://doi.org/10.1007/s00296-021-04928-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-021-04928-x