Abstract
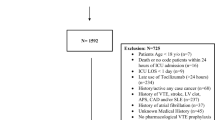
Acute myocardial infarction (MI) occurs when blood supply falls below critical levels and normal cellular maintenance mechanisms fail. Interleukin-6 (IL-6) is a proinflammatory cytokine released in MI and associated with poor clinical outcomes. Tocilizumab (TCZ) is a humanized monoclonal antibody against the IL-6 receptor. In a randomized, double-blinded, placebo controlled trial we assigned subjects admitted with MI a single TCZ dose of 162 mg subcutaneously vs. placebo in addition to standard of care medications and interventions. Primary outcome was a change in major adverse cardiac events (MACE) 30 days after enrollment. Secondary outcomes assessed changes in CRP 30 days after enrollment, changes in QT/QTc, and monitoring for trends in adverse events. Futility analysis was performed as subject enrollment slowed and no trends were noted in either the primary or secondary outcomes. Twenty-eight subjects were enrolled; 12 to TCZ and 16 to placebo. No statistically significant differences were noted between study arms regarding demographics, comorbidities, or medical/interventional therapies received. No statistically significant differences in MACE were observed. CRP increased after administration of TCZ but this was not statistically significant. No adverse events or safety signals were observed. Though futility analysis suggested that the primary outcome was not likely achievable as our recruitment slowed, we did not observe any adverse events or safety trends. Building on this information, future studies should be conducted to assess a true benefit from TCZ as adjunct therapy for MI. The work reported herein was performed under United States Air Force Surgeon General approved Clinical Investigation FKE20140029 and has been registered at ClinicalTrials.gov under identifier NCT02419937.

Similar content being viewed by others
References
Moran AE, Tzong KY, Forouzanfar MH et al (2014) Variations in ischemic heart disease burden by age, country, and income: the global burden of diseases, injuries and risk factors 2010 study. Glob Heart 9:91–99
Fox CS, Evans JC, Larson MG, Kannel WB, Levy D (2004) Temporal trends in coronary heart disease mortality and sudden cardiac death from 1950 to 1999: the Framingham heart study. Circulation 110:522–527
Rosamond WD, Chambless LE, Heiss G et al (2012) Twenty-two-year trends in incidence of myocardial infarction, coronary heart disease mortality, and case fatality in 4 US communities, 1987–2008. Circulation 125:1848–1857
Reddy K, Khaliq A, Henning RJ (2015) Recent advances in the diagnosis and treatment of acute myocardial infarction. World J Cardiol 7(5):243–276
Amsterdam EA, Wenger NK, Brindis RG et al (2014) American College of Cardiology, American Heart Association Task Force on Practice Guidelines, Society for Cardiovascular Angiography and Interventions, Society of Thoracic Surgeons, American Association for Clinical Chemistry. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 64(24):e139–e228
Tanaka T, Narazaki M, Kishimoto T (2014) IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol 6(10):a016295
Miyao Y, Yasue H, Ogawa H et al (1993) Elevated plasma interleukin-6 levels in patients with acute myocardial infarction. Am Heart J 126(6):1299–1304
Gabriel AS, Martinsson A, Wretlind B et al (2004) IL-6 levels in acute and post myocardial infarction: their relation to CRP levels, infarction size, left ventricular systolic function, and heart failure. Eur J Intern Med 15(8):523–528
López-Cuenca Á, Manzano-Fernández S, Lip GY et al (2013) Interleukin-6 and high-sensitivity C-reactive protein for the prediction of outcomes in non-ST-segment elevation acute coronary syndromes. Rev Esp Cardiol (English Edition) 66(3):185–192. doi:10.1016/j.rec.2012.07.019
Zamani P, Schwartz GG, Olsson AG et al (2013) Myocardial ischemia reduction with aggressive cholesterol lowering (MIRACL) study investigators. Inflammatory biomarkers, death, and recurrent nonfatal coronary events after an acute coronary syndrome in the MIRACL study. J Am Heart Assoc 2(1):e003103. doi:10.1161/JAHA.112.003103
Karpiński L, Płaksej R, Kosmala W, Witkowska M (2008) Serum levels of interleukin-6, interleukin-10 and C-reactive protein in relation to left ventricular function in patients with myocardial infarction treated with primary angioplasty. Kardiol Pol 66(12):1279–1285
Narazaki M, Tanaka T, Kishimoto T (2017) The role and therapeutic targeting of IL-6 in rheumatoid arthritis. Expert Rev Clin Immunol 13(6):535–551
van’t Hoog AH, Meme HK, Laserson KF et al (2012) Screening strategies for tuberculosis prevalence surveys: the value of chest radiography and symptoms. PLoS ONE 7(7):e38691
US Food and Drug Administration. Tocilizumab prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/125276s092lbl.pdf. Accessed 3 Oct 2013
Goldenberg I, Moss AJ, Zareba W (2006) QT interval: how to measure it and what is “normal”. J Cardiovasc Electrophysiol 17(3):333–336
Anderson JL, Morrow DA (2017) Acute myocardial infarction. N Engl J Med 376(21):2053–2064
Khanna D, McMahon M, Furst DE (2004) Anti-tumor necrosis factor alpha therapy and heart failure: what have we learned and where do we go from here? Arthritis Rheumatol 50(4):1040–1050
Singh JA, Saag KG, Bridges SL Jr et al (2016) 2015 American college of rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Rheumatol 68(1):1–26
Kanda J, Kawabata H, Yamaji Y et al (2007) Reversible cardiomyopathy associated with multicentric castleman disease: successful treatment with tocilizumab, an anti-interleukin 6 receptor antibody. Int J Hematol 85(3):207–211
Man L, Goudar RK (2013) Reversal of cardiomyopathy with tocilizumab in a case of HIV-negative castleman’s disease. Eur J Haematol 91(3):273–276
Kleveland O, Kunszt G, Bratlie M et al (2016) Effect of a single dose of the interleukin-6 receptor antagonist tocilizumab on inflammation and troponin T release in patients with non-ST-elevation myocardial infarction: a double-blind, randomized, placebo-controlled phase 2 trial. Eur Heart J 37(30):2406–2413
Myles PS (2014) Meaningful outcome measures in cardiac surgery. J Extra Corp Technol 46(1):23–27
Zhang X, Georgy A, Rowell L (2013) Pharmacokinetics and pharmacodynamics of tocilizumab, a humanized anti-interleukin-6 receptor monoclonal antibody, following single-dose administration by subcutaneous and intravenous routes to healthy subjects. Int J Clin Pharmacol Ther 51(6):443–455
Acknowledgements
We would like to thank Yolanda M. Moulds-Love, RPh, Dr. William Pomeroy, Dr. C. Bryan Ramsey, and Dr. Suizhao Wang for their assistance developing the protocol and assistance in preparing this manuscript.
Author information
Authors and Affiliations
Contributions
Each author has participated in the writing of the manuscript and has seen and approved the submitted version. Furthermore, each author has been involved in the conception and design of the study and the analysis of the data.
Corresponding author
Ethics declarations
Funding
We affirm that we have no financial support or other benefits from commercial sources to disclose. The authors have received no pharmaceutical or industry support in writing this manuscript.
Conflict of interest
This manuscript represents original work and is not under consideration for publication elsewhere. All authors meet the criteria for authorship and that the authors will sign a statement attesting authorship, disclosing all potential conflicts of interest, and releasing the copyright should the manuscript be accepted for publication.
Informed consent
The voluntary, fully informed consent of the subjects used in this research was obtained as required by 32 CFR 219 and AFI 40-402, Protection of Human Subjects in Biomedical and Behavioral Research.
Additional information
The views expressed in this article are those of the authors and do not reflect the official policy or position of the United States Air Force, Department of Defense, or the US Government.
Rights and permissions
About this article
Cite this article
Carroll, M.B., Haller, C. & Smith, C. Short-term application of tocilizumab during myocardial infarction (STAT-MI). Rheumatol Int 38, 59–66 (2018). https://doi.org/10.1007/s00296-017-3842-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-017-3842-y