Abstract
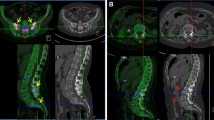
The goal of this study was to demonstrate whether increased 18F-fluoride uptake lesions on positron emission tomography (PET) scan can predict new syndesmophyte development in patients with ankylosing spondylitis (AS). In 12 AS patients, 18F-fluoride PET and magnetic resonance imaging (MRI) was performed at baseline, and radiography was performed at baseline and the 2-year follow-up. The following data were recorded: the presence of increased 18F-fluoride uptake lesions on PET defined as an uptake greater than the uptake in the adjacent normal vertebral body; acute (type A) and advanced (type B) corner inflammatory lesions (CILs) and fat lesions on MRI; and syndesmophytes on radiography. Of 231 anterior vertebral corners without syndesmophyte at baseline, 13 type A CILs (5.5%), 2 type B CILs (0.9%), and 20 fat lesions (8.7%) on MRI and six increased fluoride uptake lesions (2.6%) on PET were observed. At the 2-year follow-up, 16 new syndesmophytes (6.9%) in eight AS patients (66.7%) occurred. New syndesmophytes developed significantly more frequently in anterior vertebral corners with increased 18F-fluoride uptake lesions (50%) or fat lesions (25%) at baseline than in those without such lesions (5.8 and 5.2%; p = 0.005 and p = 0.007, respectively). After adjusting confounding factors, baseline increased 18F-fluoride uptake lesions was independently associated with new syndesmophytes development (OR 13.8, 95% CI 1.5–124.3, p = 0.019). Fat lesions were also associated with new syndesmophytes formation. Our data suggest that 18F-fluoride PET may be applied to identify AS patients with high risk of future syndesmophyte formation.

Similar content being viewed by others
References
Lories RJ, Haroon N (2014) Bone formation in axial spondyloarthritis. Best Pract Res Clin Rheumatol 28 (5):765–777. doi:10.1016/j.berh.2014.10.008
Louie GH, Ward MM (2014) Measurement and treatment of radiographic progression in ankylosing spondylitis: lessons learned from observational studies and clinical trials. Curr Opin Rheumatol 26(2):145–150. doi:10.1097/BOR.0000000000000025
Baraliakos X, Listing J, Rudwaleit M, Brandt J, Sieper J, Braun J (2005) Radiographic progression in patients with ankylosing spondylitis after 2 years of treatment with the tumour necrosis factor alpha antibody infliximab. Ann Rheum Dis 64(10):1462–1466. doi:10.1136/ard.2004.033472
van der Heijde D, Landewe R, Einstein S, Ory P, Vosse D, Ni L, Lin SL, Tsuji W, Davis JC Jr (2008) Radiographic progression of ankylosing spondylitis after up to two years of treatment with etanercept. Arthritis Rheum 58(5):1324–1331. doi:10.1002/art.23471
van der Heijde D, Salonen D, Weissman BN, Landewe R, Maksymowych WP, Kupper H, Ballal S, Gibson E, Wong R (2009) Assessment of radiographic progression in the spines of patients with ankylosing spondylitis treated with adalimumab for up to 2 years. Arthritis Res Ther 11(4):R127. doi:10.1186/ar2794
Rudwaleit M, Baraliakos X, Listing J, Brandt J, Sieper J, Braun J (2005) Magnetic resonance imaging of the spine and the sacroiliac joints in ankylosing spondylitis and undifferentiated spondyloarthritis during treatment with etanercept. Ann Rheum Dis 64(9):1305–1310. doi:10.1136/ard.2004.032441
Maksymowych WP, Chiowchanwisawakit P, Clare T, Pedersen SJ, Ostergaard M, Lambert RG (2009) Inflammatory lesions of the spine on magnetic resonance imaging predict the development of new syndesmophytes in ankylosing spondylitis: evidence of a relationship between inflammation and new bone formation. Arthritis Rheum 60(1):93–102. doi:10.1002/art.24132
Pedersen SJ, Chiowchanwisawakit P, Lambert RG, Ostergaard M, Maksymowych WP (2011) Resolution of inflammation following treatment of ankylosing spondylitis is associated with new bone formation. J Rheumatol 38(7):1349–1354. doi:10.3899/jrheum.100925
Chiowchanwisawakit P, Lambert RG, Conner-Spady B, Maksymowych WP (2011) Focal fat lesions at vertebral corners on magnetic resonance imaging predict the development of new syndesmophytes in ankylosing spondylitis. Arthritis Rheum 63(8):2215–2225. doi:10.1002/art.30393
van der Heijde D, Machado P, Braun J, Hermann KG, Baraliakos X, Hsu B, Baker D, Landewe R (2012) MRI inflammation at the vertebral unit only marginally predicts new syndesmophyte formation: a multilevel analysis in patients with ankylosing spondylitis. Ann Rheum Dis 71(3):369–373. doi:10.1136/annrheumdis-2011-200208
Baraliakos X, Heldmann F, Callhoff J, Listing J, Appelboom T, Brandt J, Van den Bosch F, Breban M, Burmester G, Dougados M, Emery P, Gaston H, Grunke M, Van Der Horst-Bruinsma IE, Landewe R, Leirisalo-Repo M, Sieper J, De Vlam K, Pappas D, Kiltz U, Van Der Heijde D, Braun J (2013) Which spinal lesions are associated with new bone formation in patients with ankylosing spondylitis treated with anti-TNF agents? A long-term observational study using MRI and conventional radiography. Ann Rheum Dis doi:10.1136/annrheumdis-2013-203425
Maksymowych WP, Morency N, Conner-Spady B, Lambert RG (2013) Suppression of inflammation and effects on new bone formation in ankylosing spondylitis: evidence for a window of opportunity in disease modification. Ann Rheum Dis 72(1):23–28. doi:10.1136/annrheumdis-2011-200859
Machado PM, Baraliakos X, van der Heijde D, Braun J, Landewe R (2015) MRI vertebral corner inflammation followed by fat deposition is the strongest contributor to the development of new bone at the same vertebral corner: a multilevel longitudinal analysis in patients with ankylosing spondylitis. Ann Rheum Dis. doi:10.1136/annrheumdis-2015-208011
Maksymowych WP (2010) Disease modification in ankylosing spondylitis. Nat Rev Rheumatol 6(2):75–81. doi:10.1038/nrrheum.2009.258
Baraliakos X, Listing J, Rudwaleit M, Sieper J, Braun J (2008) The relationship between inflammation and new bone formation in patients with ankylosing spondylitis. Arthritis Res Ther 10(5):R104. doi:10.1186/ar2496
Appel H, Loddenkemper C, Grozdanovic Z, Ebhardt H, Dreimann M, Hempfing A, Stein H, Metz-Stavenhagen P, Rudwaleit M, Sieper J (2006) Correlation of histopathological findings and magnetic resonance imaging in the spine of patients with ankylosing spondylitis. Arthritis Res Ther 8(5):R143. doi:10.1186/ar2035
Li Y, Schiepers C, Lake R, Dadparvar S, Berenji GR (2012) Clinical utility of (18)F-fluoride PET/CT in benign and malignant bone diseases. Bone 50(1):128–139. doi:10.1016/j.bone.2011.09.053
Hsu WK, Virk MS, Feeley BT, Stout DB, Chatziioannou AF, Lieberman JR (2008) Characterization of osteolytic, osteoblastic, and mixed lesions in a prostate cancer mouse model using 18F-FDG and 18F-fluoride PET/CT. J Nucl Med 49(3):414–421. doi:10.2967/jnumed.107.045666
Tan AL, Tanner SF, Waller ML, Hensor EM, Burns A, Jeavons AP, Bury RF, Emery P, McGonagle D (2013) High-resolution [18F]fluoride positron emission tomography of the distal interphalangeal joint in psoriatic arthritis–a bone-enthesis-nail complex. Rheumatology 52(5):898–904. doi:10.1093/rheumatology/kes384
Bruijnen ST, van der Weijden MA, Klein JP, Hoekstra OS, Boellaard R, van Denderen JC, Dijkmans BA, Voskuyl AE, van der Horst-Bruinsma IE, van der Laken CJ (2012) Bone formation rather than inflammation reflects ankylosing spondylitis activity on PET-CT: a pilot study. Arthritis Res Ther 14(2):R71. doi:10.1186/ar3792
Darrieutort-Laffite C, Ansquer C, Maugars Y, Le Goff B, Bodere F, Berthelot JM (2015) Sodium (18)F-sodium fluoride PET failed to predict responses to TNFalpha antagonist therapy in 31 patients with possible spondyloarthritis not meeting ASAS criteria. Jt Bone Spine 82 (6):411–416. doi:10.1016/j.jbspin.2015.08.012
Lee SG, Kim IJ, Kim KY, Kim HY, Park KJ, Kim SJ, Park EK, Jeon YK, Yang BY, Kim GT (2015) Assessment of bone synthetic activity in inflammatory lesions and syndesmophytes in patients with ankylosing spondylitis: the potential role of 18F-fluoride positron emission tomography-magnetic resonance imaging. Clin Exp Rheumatol 33(1):90–97
Buchbender C, Ostendorf B, Ruhlmann V, Heusch P, Miese F, Beiderwellen K, Schneider M, Braun J, Antoch G, Baraliakos X (2015) Hybrid 18F-labeled fluoride positron emission tomography/magnetic resonance (MR) imaging of the sacroiliac joints and the spine in patients with axial spondyloarthritis: a pilot study exploring the link of mr bone pathologies and increased osteoblastic activity. J Rheumatol 42(9):1631–1637. doi:10.3899/jrheum.150250
Toussirot E, Caoduro C, Ungureanu C, Michel F, Runge M, Boulahdour H (2015) 18F- fluoride PET/CT assessment in patients fulifilling the clinical arm of the ASAS criteria for axial spondyloarthritis. A comparative study with ankylosing spondylitis. Clin Exp Rheumatol 33(4):588
van der Linden S, Valkenburg HA, Cats A (1984) Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum 27(4):361–368
Dougados M, Simon P, Braun J, Burgos-Vargas R, Maksymowych WP, Sieper J, van der Heijde D (2011) ASAS recommendations for collecting, analysing and reporting NSAID intake in clinical trials/epidemiological studies in axial spondyloarthritis. Ann Rheum Dis 70(2):249–251. doi:10.1136/ard.2010.133488
Lambert RGW, Pedersen SJ, Maksymowych WP, Chiowchanwisawakit P, Ostergaard M (2009) Active inflammatory lesions detected by magnetic resonance imaging in the spine of patients with spondyloarthritis - definitions, assessment system, and reference image set. J Rheumatol 84(0):3–17. doi:10.3899/jrheum.090616
Creemers MC, Franssen MJ, van’t Hof MA, Gribnau FW, van de Putte LB, van Riel PL (2005) Assessment of outcome in ankylosing spondylitis: an extended radiographic scoring system. Ann Rheum Dis 64(1):127–129. doi:10.1136/ard.2004.020503
Wong KK, Piert M (2013) Dynamic bone imaging with 99mTc-labeled diphosphonates and 18F-NaF: mechanisms and applications. J Nucl Med 54(4):590–599. doi:10.2967/jnumed.112.114298
Raynor W, Houshmand S, Gholami S, Emamzadehfard S, Rajapakse CS, Blomberg BA, Werner TJ, Hoilund-Carlsen PF, Baker JF, Alavi A (2016) Evolving role of molecular imaging with (18)F-sodium fluoride PET as a biomarker for calcium metabolism. Curr Osteoporos Rep 14(4):115–125. doi:10.1007/s11914-016-0312-5
Jadvar H, Desai B, Conti PS (2015) Sodium 18F-fluoride PET/CT of bone, joint, and other disorders. Semin Nucl Med 45(1):58–65. doi:10.1053/j.semnuclmed.2014.07.008
Strobel K, Fischer DR, Tamborrini G, Kyburz D, Stumpe KD, Hesselmann RG, Johayem A, von Schulthess GK, Michel BA, Ciurea A (2010) 18F-fluoride PET/CT for detection of sacroiliitis in ankylosing spondylitis. Eur J Nucl Med Mol Imaging 37(9):1760–1765. doi:10.1007/s00259-010-1464-7
Lories RJ, Luyten FP, de Vlam K (2009) Progress in spondylarthritis. Mechanisms of new bone formation in spondyloarthritis. Arthritis Res Ther 11(2):221. doi:10.1186/ar2642
Haroon N (2015) Ankylosis in ankylosing spondylitis: current concepts. Clin Rheumatol 34(6):1003–1007. doi:10.1007/s10067-015-2956-4
Maksymowych WP, Wichuk S, Chiowchanwisawakit P, Lambert RG, Pedersen SJ (2014) Fat metaplasia and backfill are key intermediaries in the development of sacroiliac joint ankylosis in patients with ankylosing spondylitis. Arthritis Rheumatol 66 (11):2958–2967. doi:10.1002/art.38792
Kang KY, Kim IJ, Yoon MA, Hong YS, Park SH, Ju JH (2015) Fat metaplasia on sacroiliac joint magnetic resonance imaging at baseline is associated with spinal radiographic progression in patients with axial spondyloarthritis. PloS one 10(8):e0135206. doi:10.1371/journal.pone.0135206
Neumann E, Junker S, Schett G, Frommer K, Muller-Ladner U (2016) Adipokines in bone disease. Nat Rev Rheumatol 12(5):296–302. doi:10.1038/nrrheum.2016.49
Syrbe U, Callhoff J, Conrad K, Poddubnyy D, Haibel H, Junker S, Frommer KW, Muller-Ladner U, Neumann E, Sieper J (2015) Serum adipokine levels in patients with ankylosing spondylitis and their relationship to clinical parameters and radiographic spinal progression. Arthritis Rheumatol 67 (3):678–685. doi:10.1002/art.38968
Tan S, Wang R, Ward MM (2015) Syndesmophyte growth in ankylosing spondylitis. Curr Opin Rheumatol 27(4):326–332. doi:10.1097/BOR.0000000000000179
Wanders A, Heijde D, Landewe R, Behier JM, Calin A, Olivieri I, Zeidler H, Dougados M (2005) Nonsteroidal antiinflammatory drugs reduce radiographic progression in patients with ankylosing spondylitis: a randomized clinical trial. Arthritis Rheum 52(6):1756–1765. doi:10.1002/art.21054
Sieper J, Listing J, Poddubnyy D, Song IH, Hermann KG, Callhoff J, Syrbe U, Braun J, Rudwaleit M (2016) Effect of continuous versus on-demand treatment of ankylosing spondylitis with diclofenac over 2 years on radiographic progression of the spine: results from a randomised multicentre trial (ENRADAS). Ann Rheum Dis 75(8):1438–1443. doi:10.1136/annrheumdis-2015-207897
Acknowledgements
We specially thank the late Professor Sung-Il Kim who devoted himself to education, research, and patient care in the Division of Rheumatology, Department of Internal Medicine, Pusan National University School of Medicine (1963–2011).
Funding
This work was supported by clinical research grant from Pusan National University Hospital 2016.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflicts of interest.
Additional information
Eun-Kyoung Park and Kyoungjune Pak have equally contributed on this work.
Rights and permissions
About this article
Cite this article
Park, EK., Pak, K., Park, JH. et al. Baseline increased 18F-fluoride uptake lesions at vertebral corners on positron emission tomography predict new syndesmophyte development in ankylosing spondylitis: a 2-year longitudinal study. Rheumatol Int 37, 765–773 (2017). https://doi.org/10.1007/s00296-017-3660-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-017-3660-2