Abstract
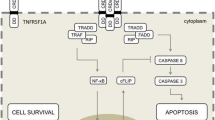
Tumor necrosis factor receptor-associated periodic syndrome (TRAPS) is an autosomal dominantly inherited autoinflammatory disease caused by mutations in the TNFRSF1A gene. Treatment is aimed at preventing acute disease attacks, improving quality of life, and preventing long-term complications such as systemic reactive amyloidosis. Biologic agents have significantly improved TRAPS management. In particular, interleukin 1 (IL-1) inhibition either with the recombinant IL-1 receptor antagonist anakinra or with the human IgG1 anti-IL-1β monoclonal antibody canakinumab has recently shown to induce a prompt and stable disease remission. Conversely, the successful experience with IL-6 inhibition is nowadays limited to a single patient. Anyway, introduction of new treatment options for patients requiring a lifelong therapy is desirable. We describe two TRAPS patients (son and father) successfully treated with canakinumab and tocilizumab, respectively. In particular, we highlight the clinical and laboratory efficacy as well as the good safety profile of tocilizumab during a 42-month follow-up period.
Similar content being viewed by others
References
McDermott MF, Aksentijevich I, Galon J et al (1999) Germline mutations in the extracellular domains of the 55 kDa TNF receptor, TNFR1, define a family of dominantly inherited autoinflammatory syndromes. Cell 97:133–144
Cantarini L, Lucherini OM, Muscari I et al (2012) Tumour necrosis factor receptor-associated periodic syndrome (TRAPS): state of the art and future perspectives. Autoimmun Rev 12:38–43
Magnotti F, Vitale A, Rigante D et al (2013) The most recent advances in pathophysiology and management of tumour necrosis factor receptor-associated periodic syndrome (TRAPS): personal experience and literature review. Clin Exp Rheumatol 31:141–149
Vitale A, Rigante D, Lucherini OM et al (2013) Biological treatments: new weapons in the management of monogenic autoinflammatory disorders. Mediat Inflamm 2013:939847. doi:10.1155/2013/939847
Ter Haar N, Lachmann H, Özen S et al (2013) Treatment of, autoinflammatory diseases: results from the Eurofever Registry and a literature review. Ann Rheum Dis 72:678–685
Vaitla PM, Radford PM, Tighe PJ et al (2011) Role of interleukin-6 in a patient with tumor necrosis factor receptor-associated periodic syndrome: assessment of outcomes following treatment with the anti-interleukin-6 receptor monoclonal antibody tocilizumab. Arthritis Rheum 63:1151–1155
Aganna E, Hawkins PN, Ozen S et al (2004) Allelic variants in genes associated with hereditary periodic fever syndromes as susceptibility factors for reactive systemic AA amyloidosis. Genes Immun 5:289–293
Cantarini L, Rigante D, Lucherini OM et al (2010) Role of etanercept in the treatment of tumor necrosis factor receptor-associated periodic syndrome: personal experience and review of the literature. Int J Immunopathol Pharmacol 23:701–707
Cantarini L, Lucherini OM, Galeazzi M et al (2009) Tumour necrosis factor receptor-associated periodic syndrome caused by a rare mutation in the TNFRSF1A gene, and with excellent response to etanercept treatment. Clin Exp Rheumatol 27:890–891
Drewe E, McDermott EM, Powell PT, Isaacs JD, Powell RJ (2003) Prospective study of anti-tumour necrosis factor receptor superfamily 1B fusion protein, and case study of anti-tumour necrosis factor receptor superfamily 1A fusion protein, in tumour necrosis factor receptor associated periodic syndrome (TRAPS): clinical and laboratory findings in a series of seven patients. Rheumatol 42:235–239
Bulua AC, Mogul DB, Aksentijevich I et al (2012) Efficacy of etanercept in the tumor necrosis factor receptor-associated periodic syndrome: a prospective, open-label, dose-escalation study. Arthritis Rheum 64:908–913
Drewe E, Powell RJ, McDermott EM (2007) Comment on: failure of anti-TNF therapy in TNF receptor 1-associated periodic syndrome (TRAPS). Rheumatol 46:1865–1866
Jacobelli S, André M, Alexandra JF, Dodé C, Papo T (2007) Failure of anti-TNF therapy in TNF receptor 1-associated periodic syndrome (TRAPS). Rheumatol 46:1211–1212
Nedjai B, Hitman GA, Quillinan N et al (2009) Proinflammatory action of the antiinflammatory drug infliximab in tumor necrosis factor receptor-associated periodic syndrome. Arthritis Rheum 60:619–625
Simon A, Bodar EJ, van der Hilst JC et al (2004) Beneficial response to interleukin 1 receptor antagonist in TRAPS. Am J Med 117:208–210
Gattorno M, Pelagatti MA, Meini A et al (2008) Persistent efficacy of anakinra in patients with tumor necrosis factor receptor–associated periodic syndrome. Arthritis Rheum 58:1516–1520
Lopalco G, Rigante D, Vitale A, Frediani B, Iannone F, Cantarini L (2014) Tumor necrosis factor receptor-associated periodic syndrome managed with the couple canakinumab-alendronate. Clin Rheumatol. doi:10.1007/s10067-014-2556-8
Brizi MG, Galeazzi M, Lucherini OM, Cantarini L, Cimaz R (2012) Successful treatment of tumor necrosis factor receptor-associated periodic syndrome with canakinumab. Ann Intern Med 156:907–908
Gattorno M, Obici L, Meini A et al (2012) Efficacy and safety of canakinumab in patients with TNF receptor associated periodic syndrome. Ann Rheum Dis 71(Suppl 3), Art ID 289
Cantarini L, Lucherini OM, Cimaz R, Galeazzi M (2010) Recurrent pericarditis caused by a rare mutation in the TNFRSF1A gene and with excellent response to anakinra treatment. Clin Exp Rheumatol 28:802
Newburger JW, Takahashi M, Gerber MA (2004) Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 110:2747–2771
Kimberley FC, Lobito AA, Siegel RM, Screaton GR (2007) Falling into TRAPS—receptor misfolding in the TNF receptor 1-associated periodic fever syndrome. Arthritis Res Ther 9:217
Aksentijevich I, Galon J, Soares M et al (2001) The tumor-necrosis-factor receptor-associated periodic syndrome: new mutations in TNFRSF1A, ancestral origins, genotype-phenotype studies, and evidence for further genetic heterogeneity of periodic fevers. Am J Hum Genet 69:301–314
Cantarini L, Rigante D, Merlini G et al (2014) The expanding spectrum of low-penetrance TNFRSF1A gene variants in adults presenting with recurrent inflammatory attacks: clinical manifestations and long-term follow-up. Semin Arthritis Rheum 43:818–823
Chan FK, Chun HJ, Zheng L, Siegel RM, Bui KL, Lenardo MJ (2000) A domain in TNF receptors that mediates ligand-independent receptor assembly and signaling. Science 288:2351–2354
Rebelo SL, Bainbridge SE, Amel-Kashipaz MR et al (2006) Modeling of tumor necrosis factor receptor superfamily 1A mutants associated with tumor necrosis factor receptor-associated periodic syndrome indicates misfolding consistent with abnormal function. Arthritis Rheum 54:2674–2687
Ravet N, Rouaghe S, Dodé C et al (2006) Clinical significance of P46L and R92Q substitutions in the tumour necrosis factor superfamily 1A gene. Ann Rheum Dis 65:1158–1162
Tchernitchko D, Chiminqgi M, Galactéros F et al (2005) Unexpected high frequency of P46L TNFRSF1A allele in sub-Saharan West African populations. Eur J Hum Genet 13:513–515
Simon A, Park H, Maddipati R et al (2010) Concerted action of wild-type and mutant TNF receptors enhances inflammation in TNF receptor 1-associated periodic fever syndrome. Proc Natl Acad Sci USA 107:9801–9806
Cantarini L, Lucherini OM, Brucato A et al (2012) Clues to detect tumor necrosis factor receptor-associated periodic syndrome (TRAPS) among patients with idiopathic recurrent acute pericarditis: results of a multicentre study. Clin Res Cardiol 101:525–531
Cantarini L, Lucherini OM, Baldari CT, Laghi Pasini F, Galeazzi M (2010) Familial clustering of recurrent pericarditis may disclose tumour necrosis factor receptor-associated periodic syndrome. Clin Exp Rheumatol 28:405–407
Cantarini L, Lucherini OM, Cimaz R et al (2009) Idiopathic recurrent pericarditis refractory to colchicine treatment can reveal tumor necrosis factor receptor-associated periodic syndrome. Int J Immunopathol Pharmacol 22:1051–1058
Cantarini L, Lucherini OM, Cimaz R, Baldari CT, Laghi Pasini F, Galeazzi M (2010) Sacroileitis and pericarditis: atypical presentation of tumor necrosis factor receptor-associated periodic syndrome and response to etanercept therapy. Clin Exp Rheumatol 28:290–291
Obici L, Merlini G (2012) Amyloidosis in autoinflammatory syndromes. Autoimmun Rev 12:14–17
Nowlan ML, Drewe E, Bulsara H et al (2006) Systemic cytokine levels and the effects of etanercept in TNF receptor-associated periodic syndrome (TRAPS) involving a C33Y mutation in TNFRSF1A. Rheumatol 45:31–37
Dickie LJ, Aziz AM, Savic S et al (2012) Involvement of X-box binding protein 1 and reactive oxygen species pathways in the pathogenesis of tumour necrosis factor receptor-associated periodic syndrome. Ann Rheum Dis 71:2035–2043
Kamata H, Honda S, Maeda S, Chang L, Hirata H, Karin M (2005) Reactive oxygen species promote TNFalpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell 120:649–661
Takada Y, Mukhopadhyay A, Kundu GC, Mahabeleshwar GH, Singh S, Aggarwal BB (2003) Hydrogen peroxide activates NF-kappa B through tyrosine phosphorylation of I kappa B alpha and serine phosphorylation of p65: evidence for the involvement of I kappa B alpha kinase and Syk protein-tyrosine kinase. J Biol Chem 278:24233–24241
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J (2010) Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol 11:136–140
Bulua AC, Simon A, Maddipati R et al (2011) Mitochondrial reactive oxygen species promote production of proinflammatory cytokines and are elevated in TNFR1-associated periodic syndrome (TRAPS). J Exp Med 208:519–533
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
La Torre, F., Muratore, M., Vitale, A. et al. Canakinumab efficacy and long-term tocilizumab administration in tumor necrosis factor receptor-associated periodic syndrome (TRAPS). Rheumatol Int 35, 1943–1947 (2015). https://doi.org/10.1007/s00296-015-3305-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-015-3305-2