Abstract
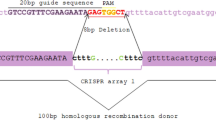
Gene-targeting is one of the most important molecular tools for genomic manipulations for research and industrial purposes. However, many factors influence targeting fidelity undermining the efforts for accurate, fast, and reliable construction of genetically modified yeast strains. Therefore, it is of great academic interest that we uncover as many as possible parameters affecting the recombination mechanisms that enable targeting. Since usually, researchers choose the orientation of the insertion (marker) within the module at random, it seemed interesting to see whether the same module will achieve essentially the same targeting efficiency when the same marker was oriented alternatively concerning the same target gene. Thus, two loci (URA3 and LEU2) and one allele (ura3-52) in a haploid yeast genetic background were targeted by artificial modules bearing homologous insertions in alternative orientations being flanked by long asymmetrical targeting homology to either replace or disrupt a genomic target. Results showed that insertion orientation within the targeting module strongly influences targeting in yeast, regardless of the targeting approach.




Similar content being viewed by others
Abbreviations
- HR:
-
Homologous recombination
- EOGT:
-
Ends-out gene-targeting
- NHEJ:
-
Non-homologous end-joining
- BIR:
-
Break-induced DNA replication
- FH:
-
Flanking homology
- THet:
-
Terminal heterology
- GTC:
-
Gene-targeting cassette
- TS:
-
Targeting success
- TC:
-
Targeting coefficient
- rQTE:
-
Relative quantitative targeting efficiency
- TGE:
-
Targeted genetic event
- nTGE:
-
Non-targeted genetic event
- YTE:
-
Yeast transformation efficiency
- SC:
-
Synthetic complete media
- ds:
-
Double-stranded
- nt:
-
Nucleotide(s)
References
Aguilera A, Gaillard H (2014) Transcription and recombination: when RNA meets DNA. Cold Spring Harb Perspect Biol 6:a016543. https://doi.org/10.1101/cshperspect.a016543
Amberg DC, Botstein D, Beasley EM (1995) Precise gene disruption in Saccharomyces cerevisiae by double fusion polymerase chain reaction. Yeast 11:1275–1280. https://doi.org/10.1002/yea.320111307
Astromskas E, Cohn M (2009) Ends-in vs. ends-out targeted insertion mutagenesis in Saccharomyces castellii. Curr Genet 55:339–347. https://doi.org/10.1007/s00294-009-0248-8
Baganz F, Hayes A, Marren D, Gardner DC, Oliver SG (1997) Suitability of replacement markers for functional analysis studies in Saccharomyces cerevisiae. Yeast 13:1563–1573. https://doi.org/10.1002/(SICI)1097-0061(199712)13:16%3c1563::AID-YEA240%3e3.0.CO;2-6
Bailis AM, Maines S (1996) Nucleotide excision repair gene function in short-sequence recombination. J Bacteriol 178:2136–2140. https://doi.org/10.1128/jb.178.7.2136-2140.1996
Baldari C, Cesareni G (1985) Plasmids pEMBLY: new single-stranded shuttle vectors for the recovery and analysis of yeast DNA sequences. Gene 35:27–32. https://doi.org/10.1016/0378-1119(85)90154-4
Baudin A, Ozier-Kalogeropoulos O, Denouel A, Lacroute F, Cullin C (1993) A simple and efficient method for direct gene deletion in Saccharomyces cerevisiae. Nucleic Acids Res 21:3329–3330. https://doi.org/10.1093/nar/21.14.3329
Cross FR (1997) “Marker swap” plasmids: convenient tools for budding yeast molecular genetics. Yeast 13:647–653. https://doi.org/10.1002/(SICI)1097-0061(19970615)13:7%3c647::AID-YEA115%3e3.0.CO;2-#
Delneri D, Tomlin GC, Wixon JL, Hutter A, Sefton M, Louis EJ, Oliver SG (2000) Exploring redundancy in the yeast genome: an improved strategy for use of the cre-loxP system. Gene 252:127–135. https://doi.org/10.1016/s0378-1119(00)00217-1
Edlind TD, Henry KW, Vermitsky J-P, Edlind MP, Raj S, Katiyar SK (2005) Promoter-dependent disruption of genes: simple, rapid, and specific PCR-based method with application to three different yeast. Curr Genet 48:117–125. https://doi.org/10.1007/s00294-005-0008-3
Estruch F, Prieto JA (2003) Construction of a Trp– commercial baker’s yeast strain by using food-safe-grade dominant drug resistance cassettes. FEMS Yeast Res 4:329–338. https://doi.org/10.1016/S1567-1356(03)00164-8
Giaever G, Chu AM, Ni L, Connelly C, Riles L, Véronneau S, Dow S, Lucau-Danila A, Anderson K, André B, Arkin AP, Astromoff A, El-Bakkoury M, Bangham R, Benito R, Brachat S, Campanaro S, Curtiss M, Davis K, Deutschbauer A, Entian KD, Flaherty P, Foury F, Garfinkel DJ, Gerstein M, Gotte D, Güldener U, Hegemann JH, Hempel S, Herman Z, Jaramillo DF, Kelly DE, Kelly SL, Kötter P, LaBonte D, Lamb DC, Lan N, Liang H, Liao H, Liu L, Luo C, Lussier M, Mao R, Menard P, Ooi SL, Revuelta JL, Roberts CJ, Rose M, Ross-Macdonald P, Scherens B, Schimmack G, Shafer B, Shoemaker DD, Sookhai-Mahadeo S, Storms RK, Strathern JN, Valle G, Voet M, Volckaert G, Wang CY, Ward TR, Wilhelmy J, Winzeler EA, Yang Y, Yen G, Youngman E, Yu K, Bussey H, Boeke JD, Snyder M, Philippsen P, Davis RW, Johnston M (2002) Functional profiling of the Saccharomyces cerevisiae genome. Nature 418:387–391. https://doi.org/10.1038/nature00935
Gjuračić K, Zgaga Z (1996) Illegitimate integration of single-stranded DNA in Saccharomyces cerevisiae. Mol Gen Genet 253:173–181. https://doi.org/10.1007/s004380050310
Glantz SA (1997) Primer of biostatistics. McGRAW-HILL, New York
Goldstein AL, Pan X, McCusker JH (1999) Heterologous URA3MX cassettes for gene replacement in Saccharomyces cerevisiae. Yeast 15:507–511. https://doi.org/10.1002/(SICI)1097-0061(199904)15:6%3c507::AID-YEA369%3e3.0.CO;2-P
Gray M, Honigberg SM (2001) Effect of chromosomal locus, GC content and length of homology on PCR-mediated targeted gene replacement in Saccharomyces. Nucleic Acids Res 29:5156–5162. https://doi.org/10.1093/nar/29.24.5156
Hashimoto S, Ogura M, Aritomi K, Hoshida H, Nishizawa Y, Akada R (2005) Isolation of auxatrophic mutants of diploid industrial yeast strains after UV mutagenesis. Appl Environ Microbiol 71:312–319. https://doi.org/10.1128/AEM.71.1.312-319.2005
Hegemann JH, Gldener U, Köhler GJ (2006) Gene disruption in the budding yeast Saccharomyces cerevisiae. Methods Mol Biol 313:129–144. https://doi.org/10.1385/1-59259-958-3:129
Jaquet L, Jauniaux JC (1999) Disruption and basic functional analysis of five chromosome X novel ORFs of Saccharomyces cerevisiae reveals YJL125c as an essential gene for vegetative growth. Yeast 15:51–61. https://doi.org/10.1002/(SICI)1097-0061(19990115)15:1%3c51::AID-YEA330%3e3.0.CO;2-1
Kegel A, Sjöstrand JO, Åström SU (2001) Nej1p, a cell type-specific regulator of nonhomologous end joining in yeast. Curr Biol 11:1611–1617. https://doi.org/10.1016/s0960-9822(01)00488-2
Klinner U, Schäfer B (2004) Genetic aspects of targeted insertion mutagenesis in yeasts. FEMS Microbiol Rev 28:201–223. https://doi.org/10.1016/j.femsre.2003.10.002
Koren P, Svetec IK, Mitrikeski PT, Zgaga Z (2000) Influence of homology size and polymorphism on plasmid integration in the yeast CYC1 DNA region. Curr Genet 37:292–297. https://doi.org/10.1007/s002940050530
Koyama H, Sumiya E, Ito T, Sekimizu K (2008) Improved method for the PCR-based gene disruption in Saccharomyces cerevisiae. FEMS Yeast Res 8:193–194. https://doi.org/10.1111/j.1567-1364.2007.00334.x
Längle-Rouault F, Jacobs E (1995) A method for performing precise alterations in the yeast genome using a recyclable selectable marker. Nucleic Acids Res 23:3079–3081. https://doi.org/10.1093/nar/23.15.3079
Lundblad V, Hartzog G, Moqtaderi Z (2001) Manipulation of cloned yeast DNA. Curr Protoc Mol Biol Chapter 13(Unit13):10. https://doi.org/10.1002/0471142727.mb1310s39
Manivasakam P, Weber SC, McElver J, Schiestl RH (1995) Micro-homology mediated PCR targeting in Saccharomyces cerevisiae. Nucleic Acids Res 23:2799–2800. https://doi.org/10.1093/nar/23.14.2799
Miklenić M, Štafa A, Bajić A, Žunar B, Lisnić B, Svetec IK (2013) Genetic transformation of the yeast Dekkera/Brettanomyces bruxellensis with non-homologous DNA. J Microbiol Biotechnol 23:674–680. https://doi.org/10.4014/jmb.1211.11047
Mitrikeski PT, Šimatović A, Brčić-Kostić K (2014) Simultaneous plasmid integration: a unifying model of multiple plasmid integration into the yeast chromosome. Period Biol 116:241–247
Nicolas A, Treco D, Schultes NP, Szostak JW (1989) An initiation site for meiotic gene conversion in the yeast Saccharomyces cerevisiae. Nature 338:35–39. https://doi.org/10.1038/338035a0
Puig O, Rutz B, Luukkonen BG, Kandels-Lewis S, Bragado-Nilsson E, Séraphin B (1998) New constructs and strategies for efficient PCR-based gene manipulations in yeast. Yeast 14:1139–1146. https://doi.org/10.1002/(SICI)1097-0061(19980915)14:12%3c1139::AID-YEA306%3e3.0.CO;2-B
Rieger KJ, Kaniak A, Coppée JY, Aljinovic G, Baudin-Baillieu A, Orlowska G, Gromadka R, Groudinsky O, Di Rago JP, Slonimski PP (1997) Large-scale phenotypic analysis-the pilot project on yeast chromosome III. Yeast 13:1547–1562. https://doi.org/10.1002/(SICI)1097-0061(199712)13:16%3c1547::AID-YEA230%3e3.0.CO;2-Y
Rose M, Winston F (1984) Identification of a Ty insertion within the coding sequence of the S. cerevisiae URA3 gene. Mol Gen Genet 193:557–560. https://doi.org/10.1007/BF00382100
Rothstein RJ (1983) One-step gene disruption in yeast. Methods Enzymol 101:202–211. https://doi.org/10.1016/0076-6879(83)01015-0
Rothstein R (1991) Targeting, disruption, replacement, and allele rescue: integrative DNA transformation in yeast. Methods Enzymol 194:281–301. https://doi.org/10.1016/0076-6879(91)94022-5
Sambrook J, Russell DW (2001) Molecular Cloning – A Laboratory Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Shen P, Huang HV (1986) Homologous recombination in Escherichia coli: dependence on substrate length and homology. Genetics 112:441–457. https://doi.org/10.1093/genetics/112.3.441
Sherman F, Fink GR, Hicks JB (1986) Methods in yeast genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Sofyanovich OA, Nishiuchi H, Yamagishi K, Maekawa K, Serebryanyy VA (2011) A new method for repeated “self-cloning” promoter replacement in Saccharomyces cerevisiae. Mol Biotechnol 48:218–227. https://doi.org/10.1007/s12033-010-9362-6
Štafa A, Miklenić M, Žunar B, Lisnić B, Symington LS, Svetec IK (2014) Sgs1 and Exo1 suppress targeted chromosome duplication during ends-in and ends-out gene targeting. DNA Repair 22:12–23. https://doi.org/10.1016/j.dnarep.2014.07.004
Štafa A, Svetec Miklenić M, Zandona A, Žunar B, Čadež N, Petković H, Svetec IK (2017) In Saccharomyces cerevisiae gene targeting fidelity depends on a transformation method and proportion of the overall length of the transforming and targeted DNA. FEMS Yeast Res 17:fox041. https://doi.org/10.1093/femsyr/fox041
Svetec IK, Štafa A, Zgaga Z (2007) Genetic side effects accompanying gene targeting in yeast: the influence of short heterologous termini. Yeast 24:637–652. https://doi.org/10.1002/yea.1497
Wach A (1996) PCR-synthesis of marker cassettes with long flanking homology regions for gene disruptions in S. cerevisiae. Yeast 12:259–265. https://doi.org/10.1002/(SICI)1097-0061(19960315)12:3%3C259::AID-YEA901%3E3.0.CO;2-C
Wach A, Brachat A, Poehlmann R, Philippsen P (1994) New heterologous modules for classical or PCR-based gene disruptions in Saccharomyces cerevisiae. Yeast 10:1793–1808. https://doi.org/10.1002/yea.320101310
Wach A, Brachat A, Alberti-Segui C, Rebischung C, Philippsen P (1997) Heterologous HIS3 marker and GFP reporter modules for PCR-targeting in Saccharomyces cerevisiae. Yeast 13:1065–1075. https://doi.org/10.1002/(SICI)1097-0061(19970915)13:11%3c1065::AID-YEA159%3e3.0.CO;2-K
Wang TT, Choi YL, Lee BH (2001) Transformation systems of non-Saccharomyces yeasts. Crit Rev Biotechnol 21:177–218. https://doi.org/10.1080/20013891081719
Wilson JH, Leung WY, Bosco G, Dieu D, Haber JE (1994) The frequency of gene targeting in yeast depends on the number of target copies. Proc Natl Acad Sci USA 91:177–181. https://doi.org/10.1073/pnas.91.1.177
Xiao W, Samson L (1993) In vivo evidence for endogenous DNA alkylation damage as a source of spontaneous mutation in eukaryotic cells. Proc Natl Acad Sci USA 90:2117–2121. https://doi.org/10.1073/pnas.90.6.2117
Zgaga Z (1991) Transformation of Saccharomyces cerevisiae with UV-irradiated single-stranded plasmid. Mutat Res 263:211–215. https://doi.org/10.1016/0165-7992(91)90003-m
Acknowledgements
This article is dedicated to the living memory of Professor Zoran Zgaga (1956-2011) under whose mentorship these experiments were conducted. I thank Dr. Alan Nicolas and Associate Professor Tomislav Domazet-Lošo for the courtesy of helping me with the genetic background of the ORD-2-7B strain and for providing the PCR primers, respectively. Special thanks go to the tragically deceased Mrs. Nataša Tomašević (1971-2020) and Mr. Vlado Bartovsky for skillful technical assistance. I also thank Dr. Mary Sopta for proofreading the manuscript.
Funding
This research was funded by grant 0058014 from the Croatian Ministry of Science, Education, and Sports to Zoran Zgaga.
Author information
Authors and Affiliations
Contributions
This research was conceptualized by ZZ (see Acknowledgments; replacement-of approach) and PTM (deletion-from and insertion-in approaches), the methodology was chosen by ZZ and PTM, formal analysis, investigation, data curation was executed by PTM, writing, and visualization were conceptualized by PTM, and funding acquisition was done by ZZ.
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Michael Polymenis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary Information
The online version contains supplementary material available at. (PDF 982 KB)
Rights and permissions
About this article
Cite this article
Mitrikeski, P.T. Insertion orientation within the cassette affects gene-targeting success during ends-out recombination in the yeast Saccharomyces cerevisiae. Curr Genet 68, 551–564 (2022). https://doi.org/10.1007/s00294-022-01246-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-022-01246-y