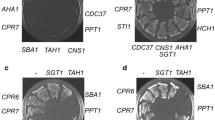
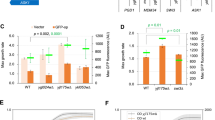
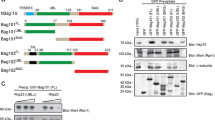
Abstract
J-domain proteins (JDPs) partner with Hsp70s to oversee proper synthesis, folding, transport and turnover of proteins in the cell. In any subcellular compartment, often multiple JDPs collaborate with a single Hsp70 to perform a variety of functions. Being co-localized, JDPs may exhibit complex genetic and physical interactions with each other, their clients as well as the Hsp70 partners. Even though most JDPs are highly specialized, redundancy between them is possible, making their functional analysis challenging. In the absence of assayable deletion phenotypes, protein overexpression appears to be a powerful alternative strategy to study JDP function. Here, we show that high levels of Caj1, one of the cytosolic JDPs, cause filamentous growth and G2/M arrest in yeast cells. Mutation in the critical HPD motif in the J-domain of Caj1 completely abolished these phenotypes, suggesting that Hsp70 co-chaperone function is important for the dominant-negative phenotypes exhibited by Caj1 overexpression. In this paper, we discuss the possible underlying mechanisms responsible for the pleiotropic phenotypes displayed by Caj1 overexpression in the light of current models proposed for dosage-sensitive genes (DSGs). Finally, we present generalized mechanisms of JDP overexpression-mediated dominant-negative phenotypes in budding yeast.



Similar content being viewed by others
References
Abe F, Horikoshi K (2000) Tryptophan permease gene TAT2 confers high-pressure growth in Saccharomyces cerevisiae. Mol Cell Biol 20:8093–8102. https://doi.org/10.1128/mcb.20.21.8093-8102.2000
Abe F, Iida H (2003) Pressure-induced differential regulation of the two tryptophan permeases Tat1 and Tat2 by ubiquitin ligase Rsp5 and its binding proteins, Bul1 and Bul2. Mol Cell Biol 23:7566–7584. https://doi.org/10.1128/mcb.23.21.7566-7584.2003
Beck T, Schmidt A, Hall MN (1999) Starvation induces vacuolar targeting and degradation of the tryptophan permease in yeast. J Cell Biol 146:1227–1238. https://doi.org/10.1083/jcb.146.6.1227
Belgareh-Touzé N, Léon S, Erpapazoglou Z et al (2008) Versatile role of the yeast ubiquitin ligase Rsp5p in intracellular trafficking. Biochem Soc Trans 36:791–796. https://doi.org/10.1042/BST0360791
Bolognesi B, Lehner B (2018) Reaching the limit. Elife. https://doi.org/10.7554/eLife.39804
Craig EA, Marszalek J (2017) How Do J-Proteins Get Hsp70 to Do So Many Different Things? Trends Biochem Sci 42:355–368. https://doi.org/10.1016/j.tibs.2017.02.007
Cullen PJ, Sprague GF (2000) Glucose depletion causes haploid invasive growth in yeast. Proc Natl Acad Sci 97:13619–13624. https://doi.org/10.1073/pnas.240345197
Cullen PJ, Sprague GF (2012) The regulation of filamentous growth in yeast. Genetics 190:23–49. https://doi.org/10.1534/genetics.111.127456
Dekel E, Alon U (2005) Optimality and evolutionary tuning of the expression level of a protein. Nature 436:588–592. https://doi.org/10.1038/nature03842
Dobriyal N, Sagarika P, Shrivastava A et al (2020) Over-expression of Caj1, a plasma membrane associated J-domain protein in Saccharomyces cerevisiae, stabilizes amino acid permeases. Biomembranes. https://doi.org/10.1016/j.bbamem.2020.183435
Dobriyal N, Tripathi P, Sarkar S et al (2017) Partial dispensability of Djp1’s J domain in peroxisomal protein import in Saccharomyces cerevisiae results from genetic redundancy with another class II J protein, Caj1. Cell Stress Chaperones 22:445–452. https://doi.org/10.1007/s12192-017-0779-8
Fan C-Y, Lee S, Ren H-Y, Cyr DM (2004) Exchangeable chaperone modules contribute to specification of type I and type II Hsp40 cellular function. Mol Biol Cell 15:761–773. https://doi.org/10.1091/mbc.e03-03-0146
Fang NN, Chan GT, Zhu M et al (2014) Rsp5/Nedd4 is the main ubiquitin ligase that targets cytosolic misfolded proteins following heat stress. Nat Cell Biol 16:1227–1237. https://doi.org/10.1038/ncb3054
Finley D, Ulrich HD, Sommer T, Kaiser P (2012) The Ubiquitin-Proteasome System of Saccharomyces cerevisiae. Genetics 192:319–360. https://doi.org/10.1534/genetics.112.140467
G R Fink SJK, (1994) Symmetric cell division in pseudohyphae of the yeast Saccharomyces cerevisiae. Mol Biol Cell 5:1003–1022. https://doi.org/10.1091/mbc.5.9.1003
Gancedo JM (2001) Control of pseudohyphae formation in Saccharomyces cerevisiae. FEMS Microbiol Rev 25:107–123. https://doi.org/10.1111/j.1574-6976.2001.tb00573.x
Ghaemmaghami S, Huh W-K, Bower K et al (2003) Global analysis of protein expression in yeast. Nature 425:737–741. https://doi.org/10.1038/nature02046
Gietz RD, Schiestl RH (2007) Frozen competent yeast cells that can be transformed with high efficiency using the LiAc/SS carrier DNA/PEG method. Nat Protoc 2:1–4. https://doi.org/10.1038/nprot.2007.17
Hernández-López MJ, García-Marqués S, Randez-Gil F, Prieto JA (2011) Multicopy suppression screening of Saccharomyces cerevisiae identifies the ubiquitination machinery as a main target for improving growth at low temperatures. Appl Environ Microbiol 77:7517–7525. https://doi.org/10.1128/AEM.00404-11
Ho B, Baryshnikova A, Brown GW (2018) Unification of protein abundance datasets yields a quantitative Saccharomyces cerevisiae Proteome. Cell Systems 6:192-205.e3. https://doi.org/10.1016/j.cels.2017.12.004
Ho E, Webber R, Wilkins MR (2008) Interactive three-dimensional visualization and contextual analysis of protein interaction networks. J Proteome Res 7:104–112. https://doi.org/10.1021/pr070274m
Jin R, Dobry CJ, McCown PJ, Kumar A (2008) Large-scale analysis of yeast filamentous growth by systematic gene disruption and overexpression. Mol Biol Cell 19:284–296. https://doi.org/10.1091/mbc.e07-05-0519
Johnson C, Kweon HK, Sheidy D et al (2014) The yeast Sks1p kinase signaling network regulates pseudohyphal growth and glucose response. PLoS Genet 10:e1004183. https://doi.org/10.1371/journal.pgen.1004183
Johnson JL, Craig EA (2001) An essential role for the substrate-binding region of Hsp40s in Saccharomyces cerevisiae. J Cell Biol 152:851–856. https://doi.org/10.1083/jcb.152.4.851
Kaake RM, Milenković T, Pržulj N et al (2010) Characterization of cell cycle specific protein interaction networks of the yeast 26S proteasome complex by the QTAX Strategy. J Proteome Res 9:2016–2029. https://doi.org/10.1021/pr1000175
Kabani M, Martineau CN (2008) Multiple hsp70 isoforms in the eukaryotic cytosol: mere redundancy or functional specificity? Curr Genomics 9:338–248. https://doi.org/10.2174/138920208785133280
Kaliszewski P, Zoładek T (2008) The role of Rsp5 ubiquitin ligase in regulation of diverse processes in yeast cells. Acta Biochim Pol 55:649–662
Kampinga HH, Craig EA (2010) The HSP70 chaperone machinery: J proteins as drivers of functional specificity. Nat Rev Mol Cell Biol 11:579–592. https://doi.org/10.1038/nrm2941
Kim J, Rose MD (2015) Stable pseudohyphal growth in budding yeast induced by synergism between septin defects and altered MAP-kinase signaling. PLoS Genet 11:e1005684. https://doi.org/10.1371/journal.pgen.1005684
Lin CH, MacGurn JA, Chu T et al (2008) Arrestin-related ubiquitin-ligase adaptors regulate endocytosis and protein turnover at the cell surface. Cell 135:714–725. https://doi.org/10.1016/j.cell.2008.09.025
Lo WS, Dranginis AM (1996) FLO11, a yeast gene related to the STA genes, encodes a novel cell surface flocculin. J Bacteriol 178:7144–7151. https://doi.org/10.1128/JB.178.24.7144-7151.1996
Lorenz MC (1998) The MEP2 ammonium permease regulates pseudohyphal differentiation in Saccharomyces cerevisiae. The EMBO Journal 17:1236–1247. https://doi.org/10.1093/emboj/17.5.1236
Lorenz MC, Cutler NS, Heitman J (2000) Characterization of alcohol-induced filamentous growth in Saccharomyces cerevisiae. MBoC 11:183–199. https://doi.org/10.1091/mbc.11.1.183
Magtanong L, Ho CH, Barker SL et al (2011) Dosage suppression genetic interaction networks enhance functional wiring diagrams of the cell. Nat Biotechnol 29:505–511. https://doi.org/10.1038/nbt.1855
Makanae K, Kintaka R, Makino T et al (2013) Identification of dosage-sensitive genes in Saccharomyces cerevisiae using the genetic tug-of-war method. Genome Res 23:300–311. https://doi.org/10.1101/gr.146662.112
Mandal AK, Nillegoda NB, Chen JA, Caplan AJ (2008) Ydj1 protects nascent protein kinases from degradation and controls the rate of their maturation. MCB 28:4434–4444. https://doi.org/10.1128/MCB.00543-08
Martineau CN, Beckerich J-M, Kabani M (2007) Flo11p-independent control of “mat” formation by hsp70 molecular chaperones and nucleotide exchange factors in yeast. Genetics 177:1679–1689. https://doi.org/10.1534/genetics.107.081141
Martinez-Anaya C (2003) In yeast, the pseudohyphal phenotype induced by isoamyl alcohol results from the operation of the morphogenesis checkpoint. J Cell Sci 116:3423–3431. https://doi.org/10.1242/jcs.00634
Mayer MP, Bukau B (2005) Hsp70 chaperones: Cellular functions and molecular mechanism. CMLS, Cell Mol Life Sci 62:670–684. https://doi.org/10.1007/s00018-004-4464-6
Meyer AE, Hung N-J, Yang P et al (2007) The specialized cytosolic J-protein, Jjj1, functions in 60S ribosomal subunit biogenesis. Proc Natl Acad Sci 104:1558–1563. https://doi.org/10.1073/pnas.0610704104
Moriya H (2015) Quantitative nature of overexpression experiments MBoC 26:3932–3939. https://doi.org/10.1091/mbc.E15-07-0512
Mumberg D, Müller R, Funk M (1995) Yeast vectors for the controlled expression of heterologous proteins in different genetic backgrounds. Gene 156:119–122. https://doi.org/10.1016/0378-1119(95)00037-7
Niu W, Li Z, Zhan W et al (2008) Mechanisms of cell cycle control revealed by a systematic and quantitative overexpression screen in S. cerevisiae. PLoS Genet. https://doi.org/10.1371/journal.pgen.1000120
Papp B, Pál C, Hurst LD (2003) Dosage sensitivity and the evolution of gene families in yeast. Nature 424:194–197. https://doi.org/10.1038/nature01771
Prelich G (2012) Gene overexpression: uses, mechanisms, and interpretation. Genetics 190:841–854. https://doi.org/10.1534/genetics.111.136911
Rosenzweig R, Nillegoda NB, Mayer MP, Bukau B (2019) The Hsp70 chaperone network. Nat Rev Mol Cell Biol 20:665–680. https://doi.org/10.1038/s41580-019-0133-3
Sahi C, Craig EA (2007) Network of general and specialty J protein chaperones of the yeast cytosol. Proc Natl Acad Sci 104:7163–7168. https://doi.org/10.1073/pnas.0702357104
Sahi C, Kominek J, Ziegelhoffer T et al (2013) Sequential duplications of an ancient member of the DnaJ-family expanded the functional chaperone network in the eukaryotic cytosol. Mol Biol Evol 30:985–998. https://doi.org/10.1093/molbev/mst008
Sarode N, Miracle B, Peng X et al (2011) Vacuolar protein sorting genes regulate mat formation in Saccharomyces cerevisiae by Flo11p-dependent and -independent mechanisms. Eukaryot Cell 10:1516–1526. https://doi.org/10.1128/EC.05078-11
Song Q, Kumar A (2012) An overview of autophagy and yeast pseudohyphal growth: integration of signaling pathways during nitrogen stress. Cells 1:263–283. https://doi.org/10.3390/cells1030263
Sopko R, Huang D, Preston N et al (2006) Mapping pathways and phenotypes by systematic gene overexpression. Mol Cell 21:319–330. https://doi.org/10.1016/j.molcel.2005.12.011
Stevenson LF, Kennedy BK, Harlow E (2001) A large-scale overexpression screen in Saccharomyces cerevisiae identifies previously uncharacterized cell cycle genes. Proc Natl Acad Sci 98:3946–3951. https://doi.org/10.1073/pnas.051013498
Suzuki A, Mochizuki T, Uemura S et al (2013) Pressure-induced endocytic degradation of the Saccharomyces cerevisiae low-affinity tryptophan permease Tat1 Is Mediated by Rsp5 ubiquitin ligase and functionally redundant PPxY motif proteins. Eukaryot Cell 12:990–997. https://doi.org/10.1128/EC.00049-13
Thomas BJ, Rothstein R (1989) The genetic control of direct-repeat recombination in Saccharomyces: the effect of rad52 and rad1 on mitotic recombination at GAL10, a transcriptionally regulated gene. Genetics 123:725–738
Vavouri T, Semple JI, Garcia-Verdugo R, Lehner B (2009) Intrinsic protein disorder and interaction promiscuity are widely associated with dosage sensitivity. Cell 138:198–208. https://doi.org/10.1016/j.cell.2009.04.029
Veitia RA (2009) On gene dosage balance in protein complexes: a comment on Semple JI, Vavouri T, Lehner B. A simple principle concerning the robustness of protein complex activity to changes in gene expression. BMC Syst Biol 3:16. https://doi.org/https://doi.org/10.1186/1752-0509-3-16
Veitia RA (2004) Gene dosage balance in cellular pathways: implications for dominance and gene duplicability. Genetics 168:569–574. https://doi.org/10.1534/genetics.104.029785
Xu T, Shively CA, Jin R et al (2010) A profile of differentially abundant proteins at the yeast cell periphery during pseudohyphal growth. J Biol Chem 285:15476–15488. https://doi.org/10.1074/jbc.M110.114926
Yoshikawa K, Tanaka T, Ida Y et al (2011) Comprehensive phenotypic analysis of single-gene deletion and overexpression strains of Saccharomyces cerevisiae. Yeast 28:349–361. https://doi.org/10.1002/yea.1843
Youker RT, Walsh P, Beilharz T et al (2004) Distinct Roles for the Hsp40 and Hsp90 Molecular Chaperones during Cystic Fibrosis Transmembrane Conductance Regulator Degradation in Yeast. MBoC 15:4787–4797. https://doi.org/10.1091/mbc.e04-07-0584
Zaslaver A, Mayo AE, Rosenberg R et al (2004) Just-in-time transcription program in metabolic pathways. Nat Genet 36:486–491. https://doi.org/10.1038/ng1348
Acknowledgements
CS Lab is supported by funding from the Science and Engineering Research Board (SERB/BIO/2019041), Department of Biotechnology (BT/PR12149/BRB/10/1348/2014) and intramural funds from IISER Bhopal.PS and ND thank CSIR and DBT, respectively, for fellowships. We are grateful to Prof. Rahguvir Singh Tomar for sharing anti-TBP antibody. We thank CS Lab members for their discussion and their insightful suggestions.
Author information
Authors and Affiliations
Contributions
PS and ND performed the experiments. CS and PS wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Michael Polymenis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sagarika, P., Dobriyal, N. & Sahi, C. Dosage sensitivity of JDPs, a valuable tool for understanding their function: a case study on Caj1 overexpression-mediated filamentous growth in budding yeast. Curr Genet 67, 407–415 (2021). https://doi.org/10.1007/s00294-021-01153-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-021-01153-8