Abstract
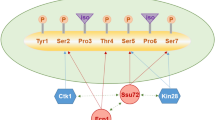
Despite the fact that the process of mRNA polyadenylation has been known for more than 40 years, a detailed understating of the mechanism underlying polyadenylation site selection is still far from complete. As 3′ end processing is intimately associated with RNA polymerase II (RNAPII) transcription, factors that can successively interact with the transcription machinery and recognize cis-acting sequences on the nascent pre-mRNA would be well suited to contribute to poly(A) site selection. Studies using the fission yeast Schizosaccharomyces pombe have recently identified Seb1, a protein that shares homology with Saccharomyces cerevisiae Nrd1 and human SCAF4/8, and that is critical for poly(A) site selection. Seb1 binds to the C-terminal domain (CTD) of RNAPII via a conserved CTD-interaction domain and recognizes specific sequence motifs clustered downstream of the polyadenylation site on the uncleaved pre-mRNA. In this short review, we summarize insights into Seb1-dependent poly(A) site selection and discuss some unanswered questions regarding its molecular mechanism and conservation.

Similar content being viewed by others
References
Ahn SH, Kim M, Buratowski S (2004) Phosphorylation of serine 2 within the RNA polymerase II C-terminal domain couples transcription and 3′ end processing. Mol Cell 13:67–76
Barilla D, Lee BA, Proudfoot NJ (2001) Cleavage/polyadenylation factor IA associates with the carboxyl-terminal domain of RNA polymerase II in Saccharomyces cerevisiae. Proc Natl Acad Sci USA 98:445–450. doi:10.1073/pnas.021545298
Becker R, Loll B, Meinhart A (2008) Snapshots of the RNA processing factor SCAF8 bound to different phosphorylated forms of the carboxyl-terminal domain of RNA polymerase II. J Biol Chem 283:22659–22669. doi:10.1074/jbc.M803540200
Boeing S et al (2016) Multiomic analysis of the UV-induced DNA damage response. Cell Rep. doi:10.1016/j.celrep.2016.04.047
Corden JL (2013) RNA polymerase II C-terminal domain: tethering transcription to transcript and template. Chem Rev 113:8423–8455. doi:10.1021/cr400158h
Curinha A, Oliveira Braz S, Pereira-Castro I, Cruz A, Moreira A (2014) Implications of polyadenylation in health and disease. Nucleus 5:508–519. doi:10.4161/nucl.36360
Davidson L, Muniz L, West S (2014) 3′ End formation of pre-mRNA and phosphorylation of Ser2 on the RNA polymerase II CTD are reciprocally coupled in human cells. Genes Dev 28:342–356. doi:10.1101/gad.231274.113
Derti A et al (2012) A quantitative atlas of polyadenylation in five mammals. Genome Res 22:1173–1183. doi:10.1101/gr.132563.111
Elkon R, Ugalde AP, Agami R (2013) Alternative cleavage and polyadenylation: extent, regulation and function. Nat Rev Genet 14:496–506. doi:10.1038/nrg3482
Erson-Bensan AE, Can T (2016) Alternative polyadenylation: another foe in cancer. Mol Cancer Res 14:507–517. doi:10.1158/1541-7786.MCR-15-0489
Grzechnik P, Gdula MR, Proudfoot NJ (2015) Pcf11 orchestrates transcription termination pathways in yeast. Genes Dev 29:849–861. doi:10.1101/gad.251470.114
Hollingworth D, Noble CG, Taylor IA, Ramos A (2006) RNA polymerase II CTD phosphopeptides compete with RNA for the interaction with Pcf11. RNA 12:555–560. doi:10.1261/rna.2304506
Hoque M et al (2013) Analysis of alternative cleavage and polyadenylation by 3′ region extraction and deep sequencing. Nat Methods 10:133–139. doi:10.1038/nmeth.2288
Hsin JP, Manley JL (2012) The RNA polymerase II CTD coordinates transcription and RNA processing. Genes Dev 26:2119–2137. doi:10.1101/gad.200303.112
Kubicek K et al (2012) Serine phosphorylation and proline isomerization in RNAP II CTD control recruitment of Nrd1. Genes Dev 26:1891–1896. doi:10.1101/gad.192781.112
Lemay JF et al (2016) The Nrd1-like protein Seb1 coordinates cotranscriptional 3′ end processing and polyadenylation site selection. Genes Dev 30:1558–1572. doi:10.1101/gad.280222.116
Lemieux C, Bachand F (2009) Cotranscriptional recruitment of the nuclear poly(A)-binding protein Pab2 to nascent transcripts and association with translating mRNPs. Nucl Acid Res 37:3418–3430
Licatalosi DD, Geiger G, Minet M, Schroeder S, Cilli K, McNeil JB, Bentley DL (2002) Functional interaction of yeast pre-mRNA 3′ end processing factors with RNA polymerase II. Mol Cell 9:1101–1111
Lunde BM et al (2010) Cooperative interaction of transcription termination factors with the RNA polymerase II C-terminal domain. Nat Struct Mol Biol 17:1195–1201. doi:10.1038/nsmb.1893
Mata J (2013) Genome-wide mapping of polyadenylation sites in fission yeast reveals widespread alternative polyadenylation. RNA Biol 10:1407–1414. doi:10.4161/rna.25758
Mayer A et al (2012) CTD tyrosine phosphorylation impairs termination factor recruitment to RNA polymerase II. Science 336:1723–1725. doi:10.1126/science.1219651
Meinhart A, Cramer P (2004) Recognition of RNA polymerase II carboxy-terminal domain by 3′-RNA-processing factors. Nature 430:223–226. doi:10.1038/nature02679
Muhlbacher W et al (2015) Structure of Ctk3, a subunit of the RNA polymerase II CTD kinase complex, reveals a noncanonical CTD-interacting domain fold. Proteins 83:1849–1858. doi:10.1002/prot.24869
Ogorodnikov A, Kargapolova Y, Danckwardt S (2016) Processing and transcriptome expansion at the mRNA 3′ end in health and disease: finding the right end. Pflugers Arch 468:993–1012. doi:10.1007/s00424-016-1828-3
O’Reilly D, Kuznetsova OV, Laitem C, Zaborowska J, Dienstbier M, Murphy S (2014) Human snRNA genes use polyadenylation factors to promote efficient transcription termination. Nucl Acid Res 42:264–275. doi:10.1093/nar/gkt892
Ozsolak F et al (2010) Comprehensive polyadenylation site maps in yeast and human reveal pervasive alternative polyadenylation. Cell 143:1018–1029. doi:10.1016/j.cell.2010.11.020
Patturajan M, Wei X, Berezney R, Corden JL (1998) A nuclear matrix protein interacts with the phosphorylated C-terminal domain of RNA polymerase II. Mol Cell Biol 18:2406–2415
Sadowski M, Dichtl B, Hubner W, Keller W (2003) Independent functions of yeast Pcf11p in pre-mRNA 3′ end processing and in transcription termination. EMBO J 22:2167–2177. doi:10.1093/emboj/cdg200
Schlackow M, Marguerat S, Proudfoot NJ, Bahler J, Erban R, Gullerova M (2013) Genome-wide analysis of poly(A) site selection in Schizosaccharomyces pombe. RNA 19:1617–1631. doi:10.1261/rna.040675.113
Schreieck A, Easter AD, Etzold S, Wiederhold K, Lidschreiber M, Cramer P, Passmore LA (2014) RNA polymerase II termination involves C-terminal-domain tyrosine dephosphorylation by CPF subunit Glc7. Natl Struct Mol Biol 21:175–179. doi:10.1038/nsmb.2753
Schwer B, Shuman S (2011) Deciphering the RNA polymerase II CTD code in fission yeast. Mol Cell 43:311–318. doi:10.1016/j.molcel.2011.05.024
Srivastava R, Ahn SH (2015) Modifications of RNA polymerase II CTD: connections to the histone code and cellular function. Biotechnol Adv 33:856–872. doi:10.1016/j.biotechadv.2015.07.008
Tian B, Manley JL (2013) Alternative cleavage and polyadenylation: the long and short of it. Trends Biochem Sci 38:312–320. doi:10.1016/j.tibs.2013.03.005
West ML, Corden JL (1995) Construction and analysis of yeast RNA polymerase II CTD deletion and substitution mutations. Genetics 140:1223–1233
Yuryev A, Patturajan M, Litingtung Y, Joshi RV, Gentile C, Gebara M, Corden JL (1996) The C-terminal domain of the largest subunit of RNA polymerase II interacts with a novel set of serine/arginine-rich proteins. Proc Natl Acad Sci USA 93:6975–6980
Zhang Z, Fu J, Gilmour DS (2005) CTD-dependent dismantling of the RNA polymerase II elongation complex by the pre-mRNA 3′-end processing factor, Pcf11. Genes Dev 19:1572–1580. doi:10.1101/gad.1296305
Acknowledgments
Work on polyadenylation site selection and transcription termination is supported by a grant from the Natural Sciences and Engineering Research Council of Canada (NSERC) to F.B. F.B. is the Canada Research Chair in Quality Control of Gene Expression.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
M. Larochelle and J. Hunyadkürti have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Larochelle, M., Hunyadkürti, J. & Bachand, F. Polyadenylation site selection: linking transcription and RNA processing via a conserved carboxy-terminal domain (CTD)-interacting protein. Curr Genet 63, 195–199 (2017). https://doi.org/10.1007/s00294-016-0645-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-016-0645-8