Abstract
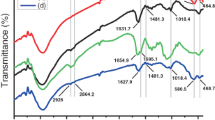
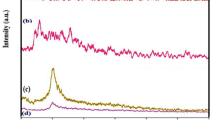
Recently, the green synthesizing methods of nanoparticles found their place in the center of attention. In this regard. the synthesis of useful nanoparticles such as the magnetic types, as well as the cases of Chia seeds that can form natural mucilage and function as a capping agent, are recognized of great importance. In this work, superparamagnetic (Fe3O4) nanoparticles were prepared by using the water extract of chia seeds for the first time, which was then coated with chitosan (CS), Fe3O4@CS core–shell, and finally, exerted for the drug delivery of oxaliplatin (OXA), and irinotecan (IRI) that were labeled as Fe3O4–OXA@CS core–shell and Fe3O4–IRI@CS core–shell, respectively. The nanoparticles were characterized through the means of XRD, FTIR, UV–Vis, TEM, FESEM, DLS, zeta potential, and VSM. The results of XRD analyses confirmed the successful synthesis of superparamagnetic nanoparticles. The observed crystallity, solid-phase, and hydrodynamic sizes were indicative of particle agglomeration in the solid phase, while in comparison to the crystallite sizes and particle diameters were increased up to more than 3-folds. The occurrence of agglomeration was more apparent in the case of Fe3O4–OXA@CS core–shell. Moreover, the cytotoxicities of nano-drugs were investigated against CT-26 cancer cells by the application of MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. The IC50 values of Fe3O4@CS core–shell, Fe3O4–OXA@CS core–shell, and Fe3O4–IRI@CS core–shell were reported to be 246.6, 79.6, and 61.1 ppm, respectively. The cytotoxicities of drug-loaded nanoparticles were exceedingly increased when being compared to the case of Fe3O4@CS core–shell.










Similar content being viewed by others
References
Hashemzadeh A, Drummen G, Avan A, Darroudi M, Khazaei M, Khajavian R, Rangrazi A, Mirzaei M (2021) When a metal-organic framework mediated smart drug delivery meets gastrointestinal cancers. J Mater Chem B
Asgharzadeh F, Hashemzadeh A, Yaghoubi A, Avan A, Nazari SE, Soleimanpour S, Hassanian SM, Ferns GA, Rahmani F, Khazaei M (2021) Therapeutic effects of silver nanoparticle containing sulfasalazine on DSS-induced colitis model. J Drug Deliv Sci Technol 61:102133
Fereydouni N, Movaffagh J, Amiri N, Darroudi S, Gholoobi A, Goodarzi A, Hashemzadeh A, Darroudi M (2021) Synthesis of nano-fibers containing nano-curcumin in zein corn protein and its physicochemical and biological characteristics. Sci Rep 11(1):1–15
Naeem M, Awan UA, Subhan F, Cao J, Hlaing SP, Lee J, Im E, Jung Y, Yoo J-W (2020) Advances in colon-targeted nano-drug delivery systems: challenges and solutions. Arch Pharmacal Res 43(1):153–169
Marques A, Costa P, Velho S, Amaral M (2020) Functionalizing nanoparticles with cancer-targeting antibodies: a comparison of strategies. J Control Release 320:180–200
Moballegh Nasery M, Abadi B, Poormoghadam D, Zarrabi A, Keyhanvar P, Khanbabaei H, Ashrafizadeh M, Mohammadinejad R, Tavakol S, Sethi G (2020) Curcumin delivery mediated by bio-based nanoparticles: a review. Molecules 25(3):689
Gong F, Yang N, Wang X, Zhao Q, Chen Q, Liu Z, Cheng L (2020) Tumor microenvironment-responsive intelligent nanoplatforms for cancer theranostics. Nano Today 32:100851
Kievit FM, Zhang M (2011) Surface engineering of iron oxide nanoparticles for targeted cancer therapy. Acc Chem Res 44(10):853–862
Dadfar SM, Roemhild K, Drude NI, von Stillfried S, Knüchel R, Kiessling F, Lammers T (2019) Iron oxide nanoparticles: diagnostic, therapeutic and theranostic applications. Adv Drug Deliv Rev 138:302–325
Ulbrich K, Hola K, Subr V, Bakandritsos A, Tucek J, Zboril R (2016) Targeted drug delivery with polymers and magnetic nanoparticles: covalent and noncovalent approaches, release control, and clinical studies. Chem Rev 116(9):5338–5431
Wahajuddin SA (2012) Superparamagnetic iron oxide nanoparticles: magnetic nanoplatforms as drug carriers. Int J Nanomed 7:3445
Dulińska-Litewka J, Łazarczyk A, Hałubiec P, Szafrański O, Karnas K, Karewicz A (2019) Superparamagnetic iron oxide nanoparticles—current and prospective medical applications. Materials 12(4):617
Mohammed L, Gomaa HG, Ragab D, Zhu J (2017) Magnetic nanoparticles for environmental and biomedical applications: a review. Particuology 30:1–14
Khan A, Sahu NK (2020) Folate encapsulation in PEG-diamine grafted mesoporous Fe3O4 nanoparticles for hyperthermia and in vitro assessment. IET Nanobiotechnol 14(9):881–888
Sullivan MV, Stockburn WJ, Hawes PC, Mercer T, Reddy SM (2020) Green synthesis as a simple and rapid route to protein modified magnetic nanoparticles for use in the development of a fluorometric molecularly imprinted polymer-based assay for detection of myoglobin. Nanotechnology 32(9):095502
Rahmani R, Gharanfoli M, Gholamin M, Darroudi M, Chamani J, Sadri K, Hashemzadeh A (2020) Plant-mediated synthesis of superparamagnetic iron oxide nanoparticles (SPIONs) using aloe vera and flaxseed extracts and evaluation of their cellular toxicities. Ceram Int 46(3):3051–3058
Park SB, Goldstein D, Krishnan AV, Lin CSY, Friedlander ML, Cassidy J, Koltzenburg M, Kiernan MC (2013) Chemotherapy‐induced peripheral neurotoxicity: a critical analysis. CA Cancer J Clin 63(6):419–437
Argyriou AA, Bruna J, Marmiroli P, Cavaletti G (2012) Chemotherapy-induced peripheral neurotoxicity (CIPN): an update. Crit Rev Oncol Hematol 82(1):51–77
Oun R, Moussa YE, Wheate NJ (2018) The side effects of platinum-based chemotherapy drugs: a review for chemists. Dalton Trans 47(19):6645–6653
Tabasi H, Hamed Mosavian MT, Sabouri Z, Khazaei M, Darroudi M (2021) pH-responsive and CD44-targeting by Fe3O4/MSNs-NH2 nanocarriers for Oxaliplatin loading and colon cancer treatment. Inorg Chem Commun 125:108430. https://doi.org/10.1016/j.inoche.2020.108430
Johnstone TC, Suntharalingam K, Lippard SJ (2016) The next generation of platinum drugs: targeted Pt (II) agents, nanoparticle delivery, and Pt (IV) prodrugs. Chem Rev 116(5):3436–3486
Gholoobi A, Meshkat Z, Abnous K, Ghayour-Mobarhan M, Ramezani M, Homaei Shandiz F, Verma KD, Darroudi M (2017) Biopolymer-mediated synthesis of Fe3O4 nanoparticles and investigation of their in vitro cytotoxicity effects. J Mol Struct 1141:594–599. https://doi.org/10.1016/j.molstruc.2017.04.024
Yew YP, Shameli K, Miyake M, Khairudin NBBA, Mohamad SEB, Naiki T, Lee KX (2020) Green biosynthesis of superparamagnetic magnetite Fe3O4 nanoparticles and biomedical applications in targeted anticancer drug delivery system: a review. Arab J Chem 13(1):2287–2308
Aisida SO, Alnasir MH, Botha S, Bashir A, Bucher R, Ahmad I, Zhao T-k, Maaza M, Ezema FI (2020) The role of polyethylene glycol on the microstructural, magnetic and specific absorption rate in thermoablation properties of Mn-Zn ferrite nanoparticles by sol–gel protocol. Eur Polym J 132:109739
Vangijzegem T, Stanicki D, Laurent S (2019) Magnetic iron oxide nanoparticles for drug delivery: applications and characteristics. Exp Opin Drug Deliv 16(1):69–78
Han H, Hou Y, Chen X, Zhang P, Kang M, Jin Q, Ji J, Gao M (2020) Metformin-induced stromal depletion to enhance the penetration of gemcitabine-loaded magnetic nanoparticles for pancreatic cancer targeted therapy. J Am Chem Soc 142(10):4944–4954
Zhang W, Taheri-Ledari R, Hajizadeh Z, Zolfaghari E, Ahghari MR, Maleki A, Hamblin MR, Tian Y (2020) Enhanced activity of vancomycin by encapsulation in hybrid magnetic nanoparticles conjugated to a cell-penetrating peptide. Nanoscale 12(6):3855–3870
Liu D, Hong Y, Li Y, Hu C, Yip T-C, Yu W-K, Zhu Y, Fong C-C, Wang W, Au S-K (2020) Targeted destruction of cancer stem cells using multifunctional magnetic nanoparticles that enable combined hyperthermia and chemotherapy. Theranostics 10(3):1181
Ebrahim SA, Ashtari A, Pedram MZ, Ebrahim NA (2019) Publication trends in drug delivery and magnetic nanoparticles. Nanoscale Res Lett 14(1):1–14
Ades S (2009) Adjuvant chemotherapy for colon cancer in the elderly: moving from evidence to practice. Oncology 23(2):162–162
Niu H, Meng Z, Cai Y (2012) Fast defluorination and removal of norfloxacin by alginate/Fe@ Fe3O4 core/shell structured nanoparticles. J Hazard Mater 227:195–203
Justin C, Samrot AV, Sahithya CS, Bhavya KS, Saipriya C (2018) Preparation, characterization and utilization of coreshell super paramagnetic iron oxide nanoparticles for curcumin delivery. PloS One 13(7):e0200440
Karimi M, Ghasemi A, Zangabad PS, Rahighi R, Basri SMM, Mirshekari H, Amiri M, Pishabad ZS, Aslani A, Bozorgomid M (2016) Smart micro/nanoparticles in stimulus-responsive drug/gene delivery systems. Chem Soc Rev 45(5):1457–1501
Dutta RK, Sahu S (2012) Development of oxaliplatin encapsulated in magnetic nanocarriers of pectin as a potential targeted drug delivery for cancer therapy. Res Pharma Sci 2:38–45
Munaweera I, Shi Y, Koneru B, Saez R, Aliev A, Di Pasqua AJ, Balkus KJ Jr (2015) Chemoradiotherapeutic magnetic nanoparticles for targeted treatment of nonsmall cell lung cancer. Mol Pharm 12(10):3588–3596
Fawcett D, Verduin JJ, Shah M, Sharma SB, Poinern GEJ (2017) A review of current research into the biogenic synthesis of metal and metal oxide nanoparticles via marine algae and seagrasses. J Nanosci 2017
Parsian M, Unsoy G, Mutlu P, Yalcin S, Tezcaner A, Gunduz U (2016) Loading of Gemcitabine on chitosan magnetic nanoparticles increases the anti-cancer efficacy of the drug. Eur J Pharmacol 784:121–128
Wu D, Zhu L, Li Y, Wang H, Xu S, Zhang X, Wu R, Yang G (2020) Superparamagnetic chitosan nanocomplexes for colorectal tumor-targeted delivery of irinotecan. Int J Pharm 584:119394
39. Zhang Y, Qi J, Chen H, Xiong C (2021) Corrigendum to “Amphiphilic diblock copolymers inhibit the formation of encrustation on the surface of biodegradable ureteral stents in vitro and in vivo” [Colloids Surf. A: Physicochem. Eng. Asp. 610 (2021) 125667]. Colloids Surf A Physicochem Eng Asp 625:126997. https://doi.org/10.1016/j.colsurfa.2021.126997
Hayashi K, Ono K, Suzuki H, Sawada M, Moriya M, Sakamoto W, Yogo T (2010) High-frequency, magnetic-field-responsive drug release from magnetic nanoparticle/organic hybrid based on hyperthermic effect. ACS Appl Mater Interfaces 2(7):1903–1911
Saboktakin MR, Tabatabaie R, Maharramov A, Ramazanov MA (2010) Synthesis and characterization of superparamagnetic chitosan–dextran sulfate hydrogels as nano carriers for colon-specific drug delivery. Carbohyd Polym 81(2):372–376
Denadai ÂM, Ianzer D, Alcântara AFdC, Santoro MM, Santos CF, Lula IS, de Camargo AC, Faljoni-Alario A, dos Santos RA, Sinisterra RD (2007) Novel pharmaceutical composition of bradykinin potentiating penta peptide with β-cyclodextrin: physical–chemical characterization and anti-hypertensive evaluation. Int J Pharm 336(1):90–98
Zambito Y, Pedreschi E, Di Colo G (2012) Is dialysis a reliable method for studying drug release from nanoparticulate systems?—a case study. Int J Pharm 434(1–2):28–34
Subbiah R, Ramalingam P, Ramasundaram S, Park K, Ramasamy MK, Choi KJ (2012) N, N, N-Trimethyl chitosan nanoparticles for controlled intranasal delivery of HBV surface antigen. Carbohyd Polym 89(4):1289–1297
Brito E, Gomes D, Cid CP, de Araújo J, Bohn F, Streck L, Fonseca J (2019) Superparamagnetic magnetite/IPEC particles. Colloids Surf A 560:376–383
Ag M, AM ES, MS S, (2013) Current situation of water pollution and its effect on aquatic life in Egypt. Egy J Occup Med 37(1):95–115
de Campo C, Dos Santos PP, Costa TMH, Paese K, Guterres SS, de Oliveira RA, Flôres SH (2017) Nanoencapsulation of chia seed oil with chia mucilage (Salvia hispanica L.) as wall material: Characterization and stability evaluation. Food Chem 234:1–9
Kuznetcova DV, Linder M, Jeandel C, Paris C, Desor F, Baranenko DA, Nadtochii LA, Arab-Tehrany E, Yen FT (2020) Nanoliposomes and nanoemulsions based on chia seed lipids: preparation and characterization. Int J Mol Sci 21(23):9079
da Silva SF, de Campo C, Paese K, Guterres SS, Costa TMH, Flôres SH (2019) Nanoencapsulation of linseed oil with chia mucilage as structuring material: characterization, stability and enrichment of orange juice. Food Res Int 120:872–879
Hernández-Morales L, Espinoza-Gómez H, Flores-López LZ, Sotelo-Barrera EL, Núñez-Rivera A, Cadena-Nava RD, Alonso-Núñez G, Espinoza KA (2019) Study of the green synthesis of silver nanoparticles using a natural extract of dark or white Salvia hispanica L. seeds and their antibacterial application. Appl Surf Sci 489:952–961
Al-Qasmi N (2021) Facial eco-friendly synthesis of copper oxide nanoparticles using chia seeds extract and evaluation of its electrochemical activity. Processes 9(11):2027
Sabouri Z, Rangrazi A, Amiri MS, Khatami M, Darroudi M (2021) Green synthesis of nickel oxide nanoparticles using Salvia hispanica L. (chia) seeds extract and studies of their photocatalytic activity and cytotoxicity effects. Bioprocess Biosyst Eng 44(11):2407–2415. https://doi.org/10.1007/s00449-021-02613-8
Silva VAJ, Andrade PL, Silva MPC, Bustamante DA, De Los Santos Valladares L, Albino Aguiar J (2013) Synthesis and characterization of Fe3O4 nanoparticles coated with fucan polysaccharides. J Magn Magn Mater 343:138–143. https://doi.org/10.1016/j.jmmm.2013.04.062
Matei E, Predescu C, Berbecaru A, Predescu A, Trusca R (2011) Leaching tests for synthesized magnetite nanoparticles used as adsorbent for metal ions from liquid solutions. Dig J Nanomater Biostruct 6(4):1701–1708
Řezníček R, Chlan V, Štěpánková H, Novák P, Maryško M (2012) Magnetocrystalline anisotropy of magnetite. J Phys Condensed Matter 24(5):055501
Sahu D, Panda N, Acharya B, Panda A (2014) Enhanced UV absorbance and photoluminescence properties of ultrasound assisted synthesized gold doped ZnO nanorods. Opt Mater 36(8):1402–1407
Nalbandian L, Patrikiadou E, Zaspalis V, Patrikidou A, Hatzidaki E, Papandreou NC (2016) Magnetic Nanoparticles in medical diagnostic applications: synthesis, characterization and proteins conjugation. Curr Nanosci 12(4):455–468
Sahoo Y, Goodarzi A, Swihart MT, Ohulchanskyy TY, Kaur N, Furlani EP, Prasad PN (2005) Aqueous ferrofluid of magnetite nanoparticles: fluorescence labeling and magnetophoretic control. J Phys Chem B 109(9):3879–3885
Machodi MJ, Daramola MO (2019) Synthesis and performance evaluation of PES/chitosan membranes coated with polyamide for acid mine drainage treatment. Sci Rep 9(1):1–14
Balamurugan M, Saravanan S, Soga T (2014) Synthesis of iron oxide nanoparticles by using Eucalyptus globulus plant extract. e-J Surf Sci Nanotechnol 12:363–367
Adewuyi A, Lau WJ (2021) Chapter 3—nanomaterial development and its applications for emerging pollutant removal in water. In: Lau WJ, Faungnawakij K, Piyachomkwan K, Ruktanonchai UR (eds) Handbook of nanotechnology applications. Elsevier, pp 67–97. https://doi.org/10.1016/B978-0-12-821506-7.00003-X
Elahi B, Mirzaee M, Darroudi M, Kazemi Oskuee R, Sadri K, Amiri MS (2019) Preparation of cerium oxide nanoparticles in salvia macrosiphon boiss seeds extract and investigation of their photo-catalytic activities. Ceram Int 45(4):4790–4797. https://doi.org/10.1016/j.ceramint.2018.11.173
Jaiswal MK, Gogoi M, Dev Sarma H, Banerjee R, Bahadur D (2014) Biocompatibility, biodistribution and efficacy of magnetic nanohydrogels in inhibiting growth of tumors in experimental mice models. Biomater Sci 2(3):370–380. https://doi.org/10.1039/C3BM60225G
Dorniani D, Hussein MZB, Kura AU, Fakurazi S, Shaari AH, Ahmad Z (2012) Preparation of Fe3O4 magnetic nanoparticles coated with gallic acid for drug delivery. Int J Nanomed 7:5745
Huxford RC, Della Rocca J, Lin W (2010) Metal–organic frameworks as potential drug carriers. Curr Opin Chem Biol 14(2):262–268
Horcajada P, Gref R, Baati T, Allan PK, Maurin G, Couvreur P, Ferey G, Morris RE, Serre C (2012) Metal–organic frameworks in biomedicine. Chem Rev 112(2):1232–1268
Horcajada P, Chalati T, Serre C, Gillet B, Sebrie C, Baati T, Eubank JF, Heurtaux D, Clayette P, Kreuz C (2010) Porous metal–organic-framework nanoscale carriers as a potential platform for drug delivery and imaging. Nat Mater 9(2):172–178
Cunha D, Ben Yahia M, Hall S, Miller SR, Chevreau H, Elkaïm E, Maurin G, Horcajada P, Serre C (2013) Rationale of drug encapsulation and release from biocompatible porous metal–organic frameworks. Chem Mater 25(14):2767–2776
Hashemzadeh A, Drummen GP, Avan A, Darroudi M, Khazaei M, Khajavian R, Mirzaei Shahrabi M, Rangrazi A (2021) When a metal-organic framework mediated smart drug delivery meets gastrointestinal cancers. J Mater Chem B 50
Abney CW, Taylor-Pashow KM, Russell SR, Chen Y, Samantaray R, Lockard JV, Lin W (2014) Topotactic transformations of metal–organic frameworks to highly porous and stable inorganic sorbents for efficient radionuclide sequestration. Chem Mater 26(18):5231–5243
Wu D, Zhu L, Li Y, Wang H, Xu S, Zhang X, Wu R, Yang G (2020) Superparamagnetic chitosan nanocomplexes for colorectal tumor-targeted delivery of irinotecan. Int J Pharm 584:119394. https://doi.org/10.1016/j.ijpharm.2020.119394
Kawassaki RK, Romano M, Dietrich N, Araki K (2021) Titanium and iron oxide nanoparticles for cancer therapy: surface chemistry and biological implications. Front Nanotechnol 3(68). https://doi.org/10.3389/fnano.2021.735434
Darroudi M, Gholami M, Rezayi M, Khazaei M (2021) An overview and bibliometric analysis on the colorectal cancer therapy by magnetic functionalized nanoparticles for the responsive and targeted drug delivery. J Nanobiotechnol 19(1):399. https://doi.org/10.1186/s12951-021-01150-6
Farzin A, Etesami SA, Quint J, Memic A, Tamayol A (2020) Magnetic nanoparticles in cancer therapy and diagnosis. Adv Healthc Mater 9(9):1901058
Acknowledgments
The technical support for this work was provided by Islamic Azad University of Quchan and Mashhad University of Medical Sciences based on the MS thesis of Ms. N. Farmanbar.
Funding
None.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
For this type of study, the ethical approval was not.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Farmanbar, N., Mohseni, S. & Darroudi, M. Green synthesis of chitosan-coated magnetic nanoparticles for drug delivery of oxaliplatin and irinotecan against colorectal cancer cells. Polym. Bull. 79, 10595–10613 (2022). https://doi.org/10.1007/s00289-021-04066-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-021-04066-1