Abstract
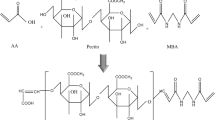
By manufacturing a pH-sensitive and biocompatible interpenetrating polymer network device, the targeting of BCS III drugs in the colon and improving their absorption can be achieved. Using N, N methylene bisacrylamide as a crosslinker and ammonium persulfate as an initiator, the formed hydrogel was evaluated using an in vitro and in vivo toxicity study of pectin–acrylamide fabricated hydrogels. Fourier transform infrared confirmed the fabrication of stable pectin/acrylamide hydrogel. Swelling studies have shown that swelling is preferably increased by increasing the acrylamide content of hydrogel structures in solutions of higher pH 7.5. For the formulated formulations, porosity and sol–gel fraction were also calculated. With a rise in pectin and acrylamide content, porosity and gel fraction increased, while porosity decreased and gel fraction improved with an increase in N, N methylene bisacrylamide content. At different pH strengths of 1.2, 5.5 and 7.5, the drug release profile was also analysed. With the increasing pH of the dissolution medium, maximum drug release was seen. By fitting experimental data to model equations and determining the corresponding parameters, the release mechanisms were studied. The maximum drug release of 90% was observed in the A6 formulation. The findings showed that the kinetics of drug release from the hydrogels in the respective buffer solutions were mainly due to Fickian diffusion. The in vivo toxicity studies confirmed the safety of the prepared hydrogels and showed no toxicity.





Similar content being viewed by others
References
Dwivedi S (2011) Hydrogel-a conceptual overview. Int J Pharm Biol Sci Arch 2(6):1588–1597
Godasu S, Sreenivas S (2017) A new validated RP-HPLC method for the determination of metformin HCL and empagliflozin in its bulk and pharmaceutical dosage forms. Int J Pharm Sci Res 8(5):2223
Maruthur NM et al (2016) Diabetes medications as monotherapy or metformin-based combination therapy for type 2 diabetes a systematic review and meta-analysis diabetes medications as monotherapy or metformin-based combination therapy. Ann Intern Med 164(11):740–751
Seifarth C, Schehler B, Schneider H (2013) Effectiveness of metformin on weight loss in non-diabetic individuals with obesity. Exp Clin Endocrinol Diabetes 121(01):27–31
Salsa T, Veiga F, Pina M (1997) Oral controlled-release dosage forms. I. Cellulose ether polymers in hydrophilic matrices. Drug Dev Ind Pharm 23(9):929–938
Mandal U et al (2008) Optimization of metformin HCl 500 mg sustained release matrix tablets using artificial neural network (ANN) based on multilayer perceptrons (MLP) model. Chem Pharm Bull 56(2):150–155
Balasubramaniam J et al (2007) Sodium alginate microspheres of metformin HCl: Formulation and in vitro evaluation. Curr Drug Deliv 4(3):249–256
El-Kamel A et al (2002) Chitosan and sodium alginate—based bioadhesive vaginal tablets. AAPS Pharm Sci 4(4):224–230
Buchholz FL, Graham AT (1998) Modern superabsorbent polymer technology. Wiley, 605 Third Ave, New York, NY 10016, USA, 1998. 279
Li Y et al (2013) Magnetic hydrogels and their potential biomedical applications. Adv Func Mater 23(6):660–672
Hoare TR, Kohane DS (2008) Hydrogels in drug delivery: progress and challenges. Polymer 49(8):1993–2007
Latif IA, Abdullah HM, Saleem MH (2016) Electrical and swelling study of different prepared hydrogel. Am J Polym Sci 6(2):50–57
Rehman S et al (2020) Enteric-coated Ca-alginate hydrogel beads: a promising tool for colon targeted drug delivery system. Polym Bull 78:5103–5117
Ahmed EM (2015) Hydrogel: preparation, characterization, and applications: a review. J Adv Res 6(2):105–121
Burkert S et al (2007) Cross-linking of poly (N-vinyl pyrrolidone) films by electron beam irradiation. Radiat Phys Chem 76(8):1324–1328
Ashford M et al (1993) An evaluation of pectin as a carrier for drug targeting to the colon. J Control Release 26(3):213–220
Wakerly Z et al (1997) Studies on drug release from pectin/ethylcellulose film-coated tablets: a potential colonic delivery system. Int J Pharm 153(2):219–224
Visser J, Voragen AGJ (1996) Pectins and pectinases, vol 14. Elsevier
May CD (1990) Industrial pectins: sources, production and applications. Carbohyd Polym 12(1):79–99
Mishra R, Banthia A, Majeed A (2012) Pectin based formulations for biomedical applications: a review. Asian J Pharm Clin Res 5(4):1–7
Imeson AP (2012) Thickening and gelling agents for food. Springer Science & Business Media, India
Rolin C, De Vries J (1990) Pectin. Food gels. Springer, pp 401–434
Liu L, Fishman ML, Hicks KB (2007) Pectin in controlled drug delivery–a review. Cellulose 14(1):15–24
Watts P, Smith A (2009) PecSys: in situ gelling system for optimised nasal drug delivery. Expert Opin Drug Deliv 6(5):543–552
Beneke CE, Viljoen AM, Hamman JH (2009) Polymeric plant-derived excipients in drug delivery. Molecules 14(7):2602–2620
Mahdavinia G et al (2009) Synthesis of porous poly (acrylamide) hydrogels using calcium carbonate and its application for slow release of potassium nitrate. Express Polym Lett 3:279–285
Fuhr U et al (2006) Toxicokinetics of acrylamide in humans after ingestion of a defined dose in a test meal to improve risk assessment for acrylamide carcinogenicity. Cancer Epidemiol Prev Biomark 15(2):266–271
Kozulic B, MosbachK (1994) Polymers, and their use as gels for electrophoresis, Google Patents
Ranjha NM, Mudassir J, Sheikh ZZ (2011) Synthesis and characterization of pH-sensitive pectin/acrylic acid hydrogels for verapamil release study. Iran Polym J 20(2):147–159
Maitra J, Shukla VK (2014) Cross-linking in hydrogels-a review. Am J Polym Sci 4(2):25–31
Kuo CK, Ma PX (2001) Ionically crosslinked alginate hydrogels as scaffolds for tissue engineering: part 1. Structure, gelation rate and mechanical properties. Biomaterials 22(6):511–521
Mudassir J, Ranjha NM (2008) Dynamic and equilibrium swelling studies: crosslinked pH sensitive methyl methacrylate-co-itaconic acid (MMA-co-IA) hydrogels. J Polym Res 15(3):195–203
Nizam El-Din HM, Abd Alla SG, El-Naggar AWM (2007) Radiation synthesis and characterization of hydrogels composed of poly (vinyl alcohol) and acrylamide mixtures. J Macromol Sci Part A Pure Appl Chem 44(1):47–54
Yin L et al (2007) Superporous hydrogels containing poly (acrylic acid-co-acrylamide)/O-carboxymethyl chitosan interpenetrating polymer networks. Biomaterials 28(6):1258–1266
Gemeinhart RA et al (2000) pH-sensitivity of fast responsive superporous hydrogels. J Biomater Sci Polym Ed 11(12):1371–1380
Ahn J-S et al (2002) Release of triamcinolone acetonide from mucoadhesive polymer composed of chitosan and poly (acrylic acid) in vitro. Biomaterials 23(6):1411–1416
Higuchi HT (1963) Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci 52(12):1145–1149
Li S et al (2006) A common profile for polymer-based controlled releases and its logical interpretation to general release process. J Pharm Pharm Sci 9(2):238–244
Korsmeyer RW et al (1983) Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm 15(1):25–35
Sharma R, Walker RB, Pathak K (2011) Evaluation of the kinetics and mechanism of drug release from econazole nitrate nanosponge loaded carbapol hydrogel. Indian J Pharm Educ Res 45(1):25–31
Naushad M, Sharma G, Alothman ZA (2019) Photodegradation of toxic dye using Gum Arabic-crosslinked-poly (acrylamide)/Ni (OH)2/FeOOH nanocomposites hydrogel. J Clean Prod 241:118263
Patil SB et al (2019) Novel biocompatible poly (acrylamide)-grafted-dextran hydrogels: synthesis, characterization and biomedical applications. J Microbiol Methods 159:200–210
Murthy PK et al (2006) Semi-IPNs of starch and poly (acrylamide-co-sodium methacrylate): preparation, swelling and diffusion characteristics evaluation. React Funct Polym 66(12):1482–1493
Öztop HN, Akyildiz F, Saraydin D (2020) Poly (acrylamide/vinylsulfonic acid) hydrogel for invertase immobilization. MicroscRes Tech. https://doi.org/10.1002/jemt.23542
Bhuyan MM et al (2019) Selective adsorption of trivalent metal ions from multielement solution by using gamma radiation-induced pectin-acrylamide-(2-Acrylamido-2-methyl-1-propanesulfonic acid) hydrogel. J Environ Chem Eng 7(1):102844
Chauhan GS, Kumari A, Sharma R (2007) Pectin and acrylamide based hydrogels for environment management technologies: synthesis, characterization, and metal ions sorption. J Appl Polym Sci 106(4):2158–2168
Saraydin D et al (1999) Influence of some amino acids on the dynamic swelling behavior of radiation-induced acrylamide hydrogel. Appl Biochem Biotechnol 82(2):115–125
Sutar PB et al (2008) Development of pH sensitive polyacrylamide grafted pectin hydrogel for controlled drug delivery system. J Mater Sci Mater Med 19(6):2247–2253
Akhtar MF, Ranjha NM, Hanif M (2015) Effect of ethylene glycol dimethacrylate on swelling and on metformin hydrochloride release behavior of chemically crosslinked pH–sensitive acrylic acid–polyvinyl alcohol hydrogel. DARU J Pharm Sci 23(1):41
Mishra RK, Datt M, Banthia AK (2008) Synthesis and characterization of pectin/PVP hydrogel membranes for drug delivery system. AAPS Pharm Sci Tech 9(2):395–403
Abbasi M et al (2019) Novel biodegradable pH-sensitive hydrogels: An efficient controlled release system to manage ulcerative colitis. Int J Biol Macromol 136:83–96
Markov PA et al (2019) Mechanical properties of the pectin hydrogels and inflammation response to their subcutaneous implantation. J Biomed Mater Res Part A 107(9):2088–2098
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Siddiqua, A., Ranjha, N.M., Rehman, S. et al. Preparation and characterization of methylene bisacrylamide crosslinked pectin/acrylamide hydrogels. Polym. Bull. 79, 7655–7677 (2022). https://doi.org/10.1007/s00289-021-03870-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-021-03870-z