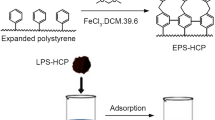
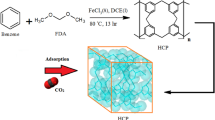
Abstract
In this investigation, a hypercrosslinked polymer based on polystyrene as an adsorbent was used for CO2 adsorption. Design of experiment with response surface methodology is considered to optimize the synthesis parameters for obtaining the maximum carbon dioxide adsorption capacity. The independent parameters including the crosslinker amount (mmol), synthesis time (h) and catalyst type (FeCl3 and AlCl3) and adsorption capacity (mg/g) are considered as the dependent parameter in design of experiment method. The optimum values of crosslinker amount, synthesis time, and catalyst type to maximize adsorption capacity achieved 17.7 mmol, 14.6 h, and FeCl3, respectively. Additionally, isotherm and kinetic modeling were carried out to determine the adsorbent behavior. The results showed that Hill and Elovich models have a better precision between other isotherm and kinetic models. Finally, thermodynamic modeling was accomplished using the optimized adsorbent and the results show that physisorption is the main of CO2 adsorption by polystyrene-based adsorbent. The values of − 13.498 kJ/mol, − 0.018 kJ/mol.K, and − 8.224 kJ/mol were obtained for enthalpy differences, entropy differences, and Gibbs free energy change at 293 K and 5 bar, respectively.













Similar content being viewed by others
Abbreviations
- q :
-
Adsorption capacity (mg/g)
- q e :
-
Equilibrium adsorption capacity (mg/g)
- m :
-
Mass of gas adsorbed (mg)
- w :
-
Mass of adsorbent (g)
- M w :
-
Molecular weight of gas (g/mol)
- R :
-
Universal gas constant (8.314 J/mol K)
- P :
-
Pressure (bar)
- P e :
-
Equilibrium pressure (bar)
- Z :
-
Compressibility factor
- B :
-
First virial coefficient
- T :
-
Temperature (K)
- V :
-
Rector volume (cm3)
- X :
-
Independent variable of RSM method
- β :
-
Coefficient of CCD-RSM polynomial
- R 2 :
-
Correlation coefficient
- i :
-
Subscripts refer to initial condition
- f :
-
Subscripts refer to final condition
- t :
-
Time (s)
- K d :
-
Distribution coefficient
- ΔH :
-
Enthalpy changes
- ΔG :
-
Gibbs free energy changes
- AARE:
-
Average absolute relative error
- q L :
-
Constant of Langmuir isotherm
- k L :
-
Constant of Langmuir isotherm
- k F :
-
Constant of Freundlich isotherm
- n F :
-
Constant of Freundlich isotherm
- q D :
-
Constant of Dubinin–Radushkevich isotherm
- β D :
-
Constant of Dubinin–Radushkevich isotherm
- ε D :
-
Constant of Dubinin–Radushkevich isotherm
- b T :
-
Constant of Temkin isotherm
- A T :
-
Constant of Temkin isotherm
- q H :
-
Constant of Hill isotherm
- K H :
-
Constant of Hill isotherm
- n H :
-
Constant of Hill isotherm
- q 1 :
-
Constant of first-order kinetic model
- k 1 :
-
Constant of first-order kinetic model
- q 2 :
-
Constant of second-order kinetic model
- k 2 :
-
Constant of second-order kinetic model
- q R :
-
Constant of Ritchie second-order kinetic model
- k R :
-
Constant of Ritchie second-order kinetic model
- α E :
-
Constant of Elovich kinetic model
- β E :
-
Constant of Elovich kinetic model
- ΔS :
-
Entropy changes
- N :
-
Number of experimental data points
References
Saeidi M, Ghaemi A, Tahvildari K, Derakhshi P (2018) Exploiting response surface methodology (RSM) as a novel approach for the optimization of carbon dioxide adsorption by dry sodium hydroxide. J Chin Chem Soc 65(12):1465–1475. https://doi.org/10.1002/jccs.201800012
Amiri M, Shahhosseini S, Ghaemi A (2017) Optimization of CO2 capture process from simulated flue gas by dry regenerable alkali metal carbonate-based adsorbent using response surface methodology (RSM). Energy Fuels 31(5):5286–5296. https://doi.org/10.1021/acs.energyfuels.6b03303
Mohammad NK, Ghaemi A, Tahvildari K (2019) Hydroxide modified activated alumina as an adsorbent for CO2 adsorption: experimental and modeling. Int J Greenh Gas Control 88:24–37. https://doi.org/10.1016/j.ijggc.2019.05.029
Li Y, Yang L, Zhu X, Hu J, Liu H (2017) Post-synthesis modification of porous organic polymers with amine: a task-specific microenvironment for co2 capture. Int J Coal Sci Technol 4(1):50–59. https://doi.org/10.1007/s40789-016-0148-8
Li J, Jia D, Guo Z, Liu Y, Lyu Y, Zhou Y, Wang J (2017) Imidazolinium based porous hypercrosslinked ionic polymers for efficient CO2 capture and fixation with epoxides. Green Chem 19:2675–2686
Ghaemi A, Shahhosseini S, Maragheh MG (2009) Nonequilibrium dynamic modeling of carbon dioxide absorption by partially carbonate d ammonia solutions. Chem Eng J 149:110–117. https://doi.org/10.1016/j.cej.2008.10.020
Penchah HR, Ghaemi A, Gilani HG (2019) Benzene-based hypercrosslinked polymer with enhanced adsorption capacity for CO2 capture. Energy Fuels 33(12):12578–12586. https://doi.org/10.1021/acs.energyfuels.9b03136
Penchah HR, Gilani HG, Ghaemi A (2020) CO2, N2, and H2 adsorption by hyper-cross-linked polymers and their selectivity evaluation by gas−solid equilibrium. J Chem Eng Data 65(10):4905–4913. https://doi.org/10.1021/acs.jced.0c00541
Rezakazemi M, Heydari I, Zhang Z (2017) Hybrid systems: combining membrane and absorption technologies leads to more efficient acid gases (CO2 and H2S) removal from natural gas. J CO2 Util 18:362–369. https://doi.org/10.1016/j.jcou.2017.02.006
Patil SB, Basavarajappa PS, Ganganagappa N, Jyothi MS, Raghu AV, Reddy KR (2019) Recent advances in non-metals-doped TiO2 nanostructured photocatalysts for visible-light driven hydrogen production, CO2 reduction and air purification. Int J Hydrogen Energy 44:13022–13039. https://doi.org/10.1016/j.ijhydene.2019.03.164
Khajeh M, Ghaemi A (2020) Nanocl ay montmorillon ite as an adsorbent for CO2 capture: experimental and modeling. J Chin Chem Soc 67(2):253–266. https://doi.org/10.1002/jccs.201900150
Khajeh M, Ghaemi A (2020) Exploiting response surface methodology for experimental modeling and optimization of CO2 adsorption onto NaOH-modified nanoclay montmorillonite. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2020.103663
Taheri FS, Ghaemi A, Maleki A, Shahhosseini S (2019) High CO2 adsorption on amine-functionalized improved mesoporous silica nanotube as an eco-friendly nanocomposite. Energy Fuels 33:5384–5397. https://doi.org/10.1021/acs.energyfuels.9b00703
Taheri FS, Ghaemi A, Maleki A (2019) High efficiency and eco-friendly TEPA-functionalized adsorbent with enhanced porosity for CO2 capture. Energy Fuels 33(11):11465–11476. https://doi.org/10.1021/acs.energyfuels.9b02636
Ghaemi A, Torab-Mostaedi M, Ghannadi-Maragheh M (2011) Characterizations of strontium(II) and barium(II) adsorption from aqueous solutions using dolomite powder. J Hazard Mater 190:916–921. https://doi.org/10.1016/j.jhazmat.2011.04.006
Niknafs H, Ghaemi A, Shahhosseini S (2015) Dynamic heat and mass transfer modeling and control in carbon dioxide reactive absorption process. Heat Mass Transf. https://doi.org/10.1007/s00231-014-1484-0
Trickett CA, Helal A, Al-Maythalony BA, Yamani ZH, Cordova KE, Yaghi OM (2017) The chemistry of metal–organic frameworks for CO2 capture, regeneration and conversion. Nat Rev Mater 2:17045. https://doi.org/10.1038/natrevmats.2017.45
Chena S, Zhu M, Tang Y, Fu Y, Li W, Xiao B (2018) Molecular simulation and experimental investigation of CO2 capture in a polymetallic cation-exchanged 13X zeolite. J Mater Chem A 6(40):19570–19583. https://doi.org/10.1039/C8TA05647A
Hauchhum L, Mahanta P (2014) Carbon dioxide adsorption on zeolites and activated carbon by pressure swing adsorption in a fixed bed. Int J Energy Environ Eng 5(4):349–356. https://doi.org/10.1007/s40095-014-0131-3
Das S, Heasman P, Ben T, Qiu S (2016) Porous organic materials: strategic design and structure−function correlation. Chem Rev 117(3):1515–1563. https://doi.org/10.1021/acs.chemrev.6b00439
Shafeeyan MS, Houshmand A, Arami-Niya A, Razaghizadeh H, Daud WMAW (2015) Modification of activated carbon using nitration followed by reduction for carbon dioxide capture. Bull Korean Chem Soc 36:533–538. https://doi.org/10.1002/bkcs.10100
Sumida K, Rogow DL, Mason JA, McDonald TM, Bloch ED, Herm ZR, Bae T-H, Long JR (2012) Carbon dioxide capture in metal-organic frameworks. J Am Chem Soc 112:724–781
Somy A, Mehrnia MR, Amrei HD, Ghanizadeh A, Safari M (2009) Adsorption of carbon dioxide using impregnated activated carbon promoted by Zinc. Int J Greenh Gas Control 3:249–254. https://doi.org/10.1016/j.ijggc.2008.10.003
McKeown NB, Budd PM, Book D (2007) Microporous polymers as potential hydrogen storage materials. Macromol Rapid Commun 28:995–1002. https://doi.org/10.1002/marc.200700054
Du X, Sun Y, Tan B, Teng Q, Yao X, Su C, Wang W (2010) Troger’s base-functionalised organic nanoporous polymerfor heterogeneous catalysis. Chem Commun 46:970–972. https://doi.org/10.1039/b920113k
Davankov VA, Tsyurupa MP (2010) Hypercrosslinked polymeric networks and adsorbing materials: synthesis, properties, structure, and applications, vol 56. Elsevier, Amsterdam
Li B, Yang X, Xia L, Majeed MI, Tan B (2013) Hollow microporous organic capsules. Sci Rep 3:2128. https://doi.org/10.1038/srep02128
Ren S, Dawson R, Laybourn A, Jiang J-x, Khimyak Y, Adams DJ, Cooper AI (2012) Functional conjugated microporous polymers: from 1,3,5-benzene to 1,3,5-triazine. Polym Chem 3:928–934
Ding S-Y, Wang W (2013) Covalent organic frameworks (cofs): from design to applications. Chem Soc Rev 42(2):548–568. https://doi.org/10.1039/c2cs35072f
Ren S, Bojdys MJ, Dawson R, Laybourn A, Khimyak YZ, Adams DJ, Cooper AI (2012) Porous, fluorescent, covalent triazine-based frameworks via room-temperature and microwave-assisted synthesis. Adv Mater 24:2357–2361. https://doi.org/10.1002/adma.201200751
Ben T, Qiu S (2013) Porous aromatic frameworks: synthesis, structure and functions. CrystEng Commun 15(1):17–26. https://doi.org/10.1039/c2ce25409c
Dawson R, Cooper AI, Adams DJ (2012) Nanoporous organic polymer networks. Prog Polym Sci 37:530–563. https://doi.org/10.1016/j.progpolymsci.2011.09.002
Penchah HR, Najafi P, Ghaemi A, Gilani HG (2021) Characterization of hypercrosslinked polymer adsorbent based on carbazole to achieve higher CO2 capture. Environ Prog Sustain Energy. https://doi.org/10.1002/ep.13586
Ratvijitvech T, Barrow M, Cooper AI, Adams DJ (2015) The effect of molecular weight on the porosity of hypercrosslinked polystyrene. Polym Chem 6(41):7280–7285. https://doi.org/10.1039/C5PY00668F
Yang Y, Tan B, Wood CD (2016) Solution-processable hypercrosslinked polymers by low cost strategies: a promising platform for gas storage and separation. J Mater Chem A. https://doi.org/10.1039/C6TA05226F
Ding L, Gao H, Xie F, Li W, Bai H, Li L (2017) Porosity-enhanced polymers from hyper-cross-linked polymer precursors. Macromolecules 50(3):956–962. https://doi.org/10.1021/acs.macromol.6b02715
Luo Y, Zhang S, Ma Y, Wang W, Tan B (2013) Microporous organic polymers synthesized by selfcondensation of aromatic hydroxymethyl monomers. Polym Chem 4(4):1126–1131. https://doi.org/10.1039/c2py20914d
Wang J, Sng W, Yi G, Zhang Y (2015) Imidazolium salts-modified porous hypercrosslinked polymers for a synergistic CO2 capture and conversion. Chem Commun 51(60):12076–12079. https://doi.org/10.1039/C5CC04702A
Wang S, Tan L, Zhang C, Hussain I, Tan B (2015) Novel POSS-based organic–inorganic hybrid porous materials by low cost strategies. J Mater Chem A 3(12):6542–6548. https://doi.org/10.1039/c4ta06963c
Luo Y, Li B, Wang W, Wu K, Tan B (2012) Hypercrosslinked aromatic heterocyclic microporous polymers: a new class of highly selective CO2 capturing materials. Adv Mater 24:5703–5707
Zhang Y, Li Y, Wang F, Zhao Y, Zhang C, Wang X, Jiang J-X (2014) Hypercrosslinked microporous organic polymer networks derived from silole-containing building blocks. Polymer 55:5746–5750. https://doi.org/10.1016/j.polymer.2014.09.014
Chen Q, Luo M, Hammershøj P, Zhou D, Han Y, Laursen BW, Yan C-G, Han B-H (2012) Microporous polycarbazole with high specific surface area for gas storage and separation. J Am Chem Soc 134:6084–6087. https://doi.org/10.1021/ja300438w
Zhou Q, Zhang M, Li Z, Li A (2011) New hypercrosslinked polymer adsorbents for solid phase extraction of polar compounds. Adv Mater Res 239–242:2076–2079
Dawson R, Stockel E, Holst JR, Adams DJ, Cooper AI (2011) Microporous organic polymers for carbon dioxide capture. Energy Environ Sci 4:4239–4245. https://doi.org/10.1039/c1ee01971f
Yao S, Yang X, Yu M, Zhang Y, Jiang J-X (2014) High surface area hypercrosslinked microporous organic polymer networks based on tetraphenylethylene for CO2 capture. J Mater Chem A 2:8054–8059. https://doi.org/10.1039/c4ta00375f
Jing X, Zou D, Cui P, Ren H, Zhu G (2013) Facile synthesis of cost-effective porous aromatic materials with enhanced carbon dioxide uptake. J Mater Chem A 1:13926–13931. https://doi.org/10.1039/C3TA13115G
Zhu X, Mahurin SM, An S-H, Do-Thanh C-L, Tian C, Li Y, Gill LW, Hagaman EW, Bian Z, Zhou J-H, Hu J, Liu H, Dai S (2014) Efficient CO2 capture by a task-specific porous organic polymer bifunctionalized with carbazole and triazine groups. Chem Commun 50:7933–7936. https://doi.org/10.1039/c4cc01588f
Zhu J-H, Chen Q, Sui Z-Y, Pan L, Yu J, Han B-H (2014) Preparation and adsorption performance of crosslinked porous polycarbazoles. J Mater Chem A 2:16181–16189. https://doi.org/10.1039/c4ta01537a
Yang X, Yu M, Zhao Y, Zhang C, Wang X, Jiang J-X (2014) Hypercrosslinked microporous polymers based on carbazole for gas storage and separation. RSC Adv 4:61051–61055. https://doi.org/10.1039/c4ra09394a
Zhai T-L, Tan L, Luo Y, Liu J-M, Tan B, Yang X-L, Xu H-B, Zhang C (2016) Microporous polymers from a carbazole-based triptycene monomer: synthesis and their applications for gas uptake. Chem Asian J 11:294–298. https://doi.org/10.1002/asia.201501105
Chang D, Yu M, Zhang C, Zhao Y, Kong R, Xie F, Jiang J-x (2016) Indolo[3,2-b]carbazole-containing hypercrosslinked microporous polymer networks for gas storage and separation. Microporous Mesoporous Mater 228:231–236. https://doi.org/10.1016/j.micromeso.2016.03.038
Yang X, Yu M, Zhao Y, Zhang C, Wang X, Jiang J-X (2014) Remarkable gas adsorption by carbonized nitrogen-rich hypercrosslinked porous organic polymers. J Mater Chem A 2:15139–15145. https://doi.org/10.1039/c4ta02782e
Li W, Zhang A, Gao H, Chen M, Liu A, Bai H, Li L (2016) Massive preparation of pitch-based organic microporous polymers for gas storage. Chem Commun 52:2780–2783. https://doi.org/10.1039/c5cc07908j
Castaldo R, Gentile G, Avella M, Carfagna C, Ambrogi V (2017) Microporous hyper-crosslinked polystyrenes and nanocomposites with high adsorption properties: a review. Polymers 9(12):651–672. https://doi.org/10.3390/polym9120651
Ahn J-H, Jang J-E, Oh C-G, Ihm S-K, Cortez J, Sherrington DC (2006) Rapid generation and control of microporosity, bimodal pore size distribution, and surface area in davankov-type hyper-cross-linked resins. Macromolecules 39:627–632
Li B, Gong R, Luo Y, Tan B (2011) 7, ) Tailoring the pore size of hypercrosslinked polymers. Soft Matter 7:10910–10916. https://doi.org/10.1039/c1sm06113e
Li B, Gong R, Wang W, Huang X, Zhang W, Li H, Hu C, Tan B (2011) A new strategy to microporous polymers: knitting rigid aromatic building blocks by external cross-linker. Macromolecules 44:2410–2414. https://doi.org/10.1021/ma200630s
Tsyurupa MP, Andreeva AI, Davankov VA (1978) On factors determining the swelling ability of crosslinked polymers. Die Angewandte Makromolekulare Chemie 70:179–187
Penner NA, Nesterenko RN, Ilyin MM, Tsyurupa MR, Davankov VA (1999) Investigation of the properties of hypercrosslinked polystyrene as a stationary phase for high-performance liquid chromatography. Chromatographia 50(9):611–620
Prausnitz JM, Lichtenthaler RN, Azevedo EGD (1999) Molecular thermodynamics of fluid-phase equilibria. 3rd edn
Myers RH, Montgomery DC, Anderson-Cook CM (2016) Response surface methodology: process and product optimization using designed experiments, 4th edn. Wiley, Hoboken
Houshmand A, Daud WMAW, Shafeeyan MS (2011) Tailoring the surface chemistry of activated carbon by nitric acid: study using response surface method. Bull Chem Soc Jpn 84(11):12511260. https://doi.org/10.1246/bcsj.20110145
Karadag D, Koc Y, Turan M, Ozturk M (2007) A comparative study of linear and non-linear regression analysis for ammonium exchange by clinoptilolite zeolite. J Hazard Mater 144(1):432–437
Falk M, Miller AG (1992) Infrared spectrum of carbon dioxide in aqueous solution. Hbrational Spectroscopy 4:105–108
Oliaee JN, Dehghany M, McKellar ARW, Moazzen-Ahmadi N (2011) High resolution infrared spectroscopy of carbon dioxide clusters up to (CO2)13. J Chem Phys 135:044315. https://doi.org/10.1063/1.3615543
Bashir MJK, Aziz HA, Yusoff MSa, Adlan MN, (2010) Application of response surface methodology (RSM) for optimization of ammoniacal nitrogen removal from semi-aerobic landfi ll leachate using ion exchange resin. Desalination 254:154–161. https://doi.org/10.1016/j.desal.2009.12.002
Ölmez T (2009) The optimization of Cr(VI) re duction and removal by electrocoagulation using response surface methodology. J Hazard Mater 162:1371–1378. https://doi.org/10.1016/j.jhazmat.2008.06.017
Vaidya PD, Kenig EY (2007) CO2-Alkanolamine reaction kinetics: a review of recent studies. Chem Eng Technol 30(11):1467–1474. https://doi.org/10.1002/ceat.200700268
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ramezanipour Penchah, H., Ghaemi, A. & Ghanadzadeh Gilani, H. Efficiency increase in hypercrosslinked polymer based on polystyrene in CO2 adsorption process. Polym. Bull. 79, 3681–3702 (2022). https://doi.org/10.1007/s00289-021-03678-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-021-03678-x