Abstract
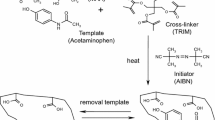
In view of technological significance of molecularly imprinted polymers (MIPs) in drug delivery, the ‘grafting from’ approach was employed to develop surface-imprinted silica particles for selective recognition and sustained delivery of a pharmaceutical drug antipyrine (ANP). ANP–MIP was fabricated using methacrylic acid (MAA) as functional monomer and ethylene glycol dimethacrylate (EGDMA) as cross-linker by UV irradiation via iniferter approach. Voltammetric measurements was used to scale the extraction and rebinding of ANP on glassy carbon electrode with Pt wire as counter electrode, Ag/AgCl as reference electrode. Various parameters were optimized for ascertaining the performance of ANP–MIP such as time, temperature, and pH. Grafted MIP was characterized by FTIR, thermogravimetry, elemental analysis, surface morphology (AFM) besides the recognition, rebinding and selectivity studies. Calibration curve linearly increases in concentration range of 0.1–2.0 mM with correlation coefficient R2 = 0.976 and limit of detection (LOD) as 0.448 µg mL−1. Thus, fabricated ANP–MIP was studied for controlled release of drug under varying conditions.













Similar content being viewed by others
References
Brodie BB, Axelrod J (1950) The fate of antipyrine in man. J Pharmacol Exp Ther 98:97–104
Hoberman A, Paradise JL, Reynolds EA, Urkin J (1997) Efficacy of Auralgan for treating ear pain in children with acute otitis media. Arch Pediatr Adolsc Med 151:675–678
Marmura MJ, Stephen SD, Schwedt TJ (2015) The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache 55:3–20
Zuehlke S, Duennbier U, Heberer T (2007) Investigation of the behavior and metabolism of pharmaceutical residues during purification of contaminated ground water used for drinking water supply. Chemosphere 69:1673–1680
Wiegel S, Aulinger A, Brockmeyer R, Harms H, Löffler J, Reincke H, Schmidt R, Stachel B, Von Tümpling W, Wanke A (2004) Pharmaceuticals in the river Elbe and its tributaries. Chemosphere 57:107–126
Reddersen K, Heberer T, Dünnbier U (2002) Identification and significance of phenazone drugs and their metabolites in ground- and drinking water. Chemosphere 49:539–544
Cai M, Zhang L, Qi F, Feng L (2013) Influencing factors and degradation products of antipyrine chlorination in water with free chlorine. J Environ Sci 25:77–84
Sakamoto JH, van de Ven AL, Godin B, Blanco E, Serda RE, Grattoni A, Ziemys A, Bouamrani A, Hu T, Ranganathan SI, De Rosa E, Martinez JO, Smid CA, Buchanan RM, Lee S-Y, Srinivasan S, Landry M, Meyn A, Tasciotti E, Liu X, Decuzzia P, Ferrari M (2010) Enabling individualized therapy through nanotechnology. Pharmacol Res 62:57–89
Lulinski P (2017) Molecularly imprinted polymers based drug delivery devices: a way to application in modern pharmacotherapy. Mater Sci Eng C 76:1344–1353
Wulff G, Gross T, Schonfeld R (1997) Enzyme models based on molecularly imprinted polymers with strong esterase activity. Angew Chem Int Ed 36:1962–1964
Toorisaka E, Yoshida M, Uezu K, Goto M, Furusaki S (1999) Artificial biocatalyst prepared by the surface molecular imprinting technique. Chem Lett 28:387–388
Mosbach K, Ramstom O (1996) The emerging technique of molecular imprinting and its future impact on biotechnology. Nat Biotechnol 14:163–170
Vidyasankar S, Dhal PK, Plunkett SD, Arnold FH (1995) Selective ligand-exchange adsorbents prepared by template polymerization. Biotechnol Bioeng 48:431–436
Kempe M, Mosbach K (1995) Molecular imprinting used for chiral separations. J Chromatogr A 694:3–13
Saha D, Warren KE, Naskar AK (2014) Controlled release of antipyrine from mesoporous carbons. Microporous Mesoporous Mater 196:327–334
Salonen J, Laitinen L, Kaukonen AM, Tuura J, Bjorkqvist M, Heikkila T, Vaha-Heikkila K, Hirvonen J, Lehto V-P (2005) Mesoporous silicon microparticles for oral drug delivery: loading and release of five model drugs. J Control Release 108:362–374
Goscianska J, Olejnik A, Nowak I (2017) APTES-functionalized mesoporous silica as a vehicle for antipyrine–adsorption and release studies. Colloids Surf A 533:187–196
Sangalli ME, Maroni A, Zema L, Busetti C, Giordano F, Gazzaniga A (2001) In vitro and in vivo evaluation of an oral system for time and/or site-specific drug delivery. J Control Release 73:103–110
Gazzaniga A, Sangalli M, Giordano F (1994) Oral Chronotopic® drug delivery systems: achievement of time and/or site specificity. Eur J Pharm Biopharm 40:246–250
Gazzaniga A, Iamartino P, Maffione G, Sangalli ME (1994) Oral delayed-release system for colonic specific delivery. Int J Pharm 108:77–83
Gazzaniga A, Busetti C, Sangalli ME, Moro L, Giordano F (1995) Oral delayed-release system for colonic specific delivery. Int J Pharm 5:83–88
Yang G, Wang D, Li Z, Zhou S, Chen Y (2003) Adsorption isotherms on aminoantipyrine imprinted polymer stationary phase. Chromatographia 58(1):53–58
Otsu T, Yoshida M (1982) Role of initiator–transfer agent–terminator (iniferter) in radical polymerization: polymer design by organic disulfides as iniferters. Macromol Rapid Commun 3:127–132
Otsu T, Matsunaga T, Doi T, Matsumoto A (1995) Features of living radical polymerization of vinyl monomers in homogeneous system using N, N-diethyldithiocarbamate derivatives as photoiniferters. Eur Polym J 31:67–78
Caldorera-Moore M, Peppas NA (2009) Micro- and nanotechnologies for intelligent and responsive biomaterial-based medical systems. Adv Drug Deliv Rev 61:1391–1401
Skoog DA, Holler FT, Nieman TA (1998) Principles of instrumental analysis, 5th edn. Harcourt Brace College Publishers, Florida, pp 13–14
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE., Robb MA, Cheeseman JR, Montgomery JA, Vreven JT, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian03 Revision D01. Gaussian Inc, Walling ford
Dennington R, Keith T, Millam J (2007) GaussView 4.1. Semichem, Inc. Shawnee Mission
Boys SF, Bernardi FD (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566
Cossi M, Barone V, Cammi R, Tomasi J (1996) Ab initio study of solvated molecules: a new implementation of the polarizable continuum model. Chem Phys Lett 255(4–6):327–335
Salonen J, Laitinen L, Kaukonen AM, Tuura J, Björkqvist M, Heikkilä T, Vähä-Heikkilä K, Hirvonen J, Lehto V-P (2005) Mesoporous silicon microparticles for oral drug delivery: loading and release of five model drugs. J Control Release 108(2):362–374
Sellergren B, Lepisto M, Mosbach K (1988) Highly enantioselective and substrate-selective polymers obtained by molecular imprinting utilizing noncovalent interactions. NMR and chromatographic studies on the nature of recognition. J Am Chem Soc 110:5853–5860
Mosbach K, Haupt K (1998) Some new developments and challenges in non-covalent molecular imprinting technology. J Mol Recognit 11:62–68
Zhang H, Ye L, Mosbach K (2006) Non-covalent molecular imprinting with emphasis on its application in separation and drug development. J Mol Recognit 19:248–259
Norell MC, Andersson HS, Nicholls IA (1998) Theophylline molecularly imprinted polymer dissociation kinetics: a novel sustained release drug dosage mechanism. J Mol Recognit 11:98–102
Cunliffe D, Kirby A, Alexander C (2005) Molecularly imprinted drug delivery systems. Adv Drug Deliv Rev 57:1836–1853
Huang X, Brazel CS (2001) On the importance and mechanisms of burst release in matrix-controlled drug delivery systems. J Control Release 73:121–136
Zaidi SA (2016) Molecular imprinted polymers as drug delivery vehicles. Drug Deliv 23:2262–2271
Acknowledgements
Authors acknowledge Department of Chemistry, Banaras Hindu University for AFM analysis. AK acknowledges UGC and BHU for university research fellowship. Financial assistance from DST, New Delhi is acknowledged (Grant no. EMR/2016/005245).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kushwaha, A., Singh, S., Gupta, N. et al. Synthesis and characterization of antipyrine-imprinted polymers and their application for sustained release. Polym. Bull. 75, 5235–5252 (2018). https://doi.org/10.1007/s00289-018-2326-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-018-2326-x