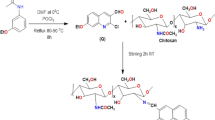
Abstract
The druggability of the Schiff bases and dithiocarbamate derivatives of chitosan was examined; the oral bioavailability and bioactivity of all these molecules against selected drug targets were investigated, as well as ADME/Tox studies were conducted. It was observed that the Lipinski’s rule of five was satisfied by all the molecules. The Schiff bases and dithiocarbamate derivatives of chitosan also show good bioactivity score for protease and enzyme inhibition. The ADME/Tox studies conducted show that almost all the derivatives are free from toxicity risks, except citral- and sulfur-containing derivatives. Substitution of the –SH group by –NH2 gives better positive results in toxicology studies. From this study, it has been observed that these molecules exhibit fairly good drug score and are orally viable molecules. The mechanism driving their bioactivity might be chelation of chitosan and its derivatives with essential metal ions. Chitosan and the derivatives studied can serve as good lead molecules for further research.




Similar content being viewed by others
Abbreviations
- C:
-
Chitosan
- CC:
-
Chitosan Schiff base of citral
- CnC:
-
Chitosan Schiff base of cinnamaldehyde
- DC:
-
Dithiocarbamate derivatives of chitosan
- DFT:
-
Density functional theory
- GC:
-
Chitosan Schiff base of glyoxylic acid
- GPCR:
-
G protein-coupled receptors
- HC:
-
Chitosan Schiff base of heptaldehyde
- PaC:
-
Chitosan Schiff base of pyruvic acid
- PC:
-
Chitosan Schiff base of pyridoxal hydrochloride
- PcC:
-
Chitosan Schiff base of 2-pyridine carbaldehyde
- PSA:
-
Polar surface area
- SC:
-
Chitosan Schiff base of salicylaldehyde
- SMILES:
-
Simplified molecular-input line-entry system
References
Lahlou M, (2013) The success of natural products in drug discovery. Pharmacol. Pharmacy 417–31
Mohamed NA, Sabaa MW, El-Ghandour AH, Abdel-Aziz MM, Abdel-Gawad OF (2013) Quaternized N-substituted carboxymethyl chitosan derivatives as antimicrobial agents. Int J Bio Macromol 60:156–164
Xia W, Liu P, Zhang J, Chen J (2011) Biological activities of chitosan and chitooligosaccharides. Food Hydrocoll 25:170–179
Kim SK, Rajapakse N (2005) Enzymatic production and biological activities of chitosan oligosaccharides (COS): a review. Carbohydr Polym 62:357–368
Lim SH, Hudson SM (2003) Review of chitosan and its derivatives as antimicrobial Agents and their uses as textile chemicals. J Macromol Sci C 43:223–269
Li DH, Liu LM, Tian KL, Liu JC, Fan XQ (2007) Synthesis, biodegradability and cytotoxicity of water-soluble isobutylchitosan. Carbohydr Polym 67:40–45
Zhao D, Huang J, Hu S, Mao J, Mei L (2011) Biochemical activities of N, O-carboxymethyl chitosan from squid cartilage. Carbohydr Polym 85:832–837
Shelma R, Sharma CP (2011) Development of lauroyl sulfated chitosan for enhancing hemocompatibility of chitosan. Coll surf B 84:561–570
de Britto D, Assis OBG (2007) A novel method for obtaining a quaternary salt of chitosan. Carbohydr Polym 69:305–310
Yang J, Cai J, Hu Y, Li D, Du Y (2012) Preparation, characterization and antimicrobial activity of 6-amino-6-deoxychitosan. Carbohydr Polym 87:202–209
Peng Y, Han B, Liu W, Xu X (2005) Preparation and antimicrobial activity of hydroxypropyl chitosan. Carbohydr Res 340:1846–1851
Jin X, Wang J, Bai J (2009) Synthesis and antimicrobial activity of the Schiff base from chitosan and citral. Carbohydr Res 344:825–829
Guo Z, Xing R, Liu S, Zhong Z, Ji X, Wang L (2007) Antifungal properties of Schiff bases of chitosan, N-substituted chitosan and quaternized chitosan. Carbohydr Res 342:1329–1332
Kajal A, Bala S, Kamboj S, Sharma N, Saini V (2013) Schiff bases: a versatile pharmacophore. J Catalysts, Article ID 893512
Kumar S, Dhar DN, Saxena PN (2009) Application of metal complexes of schiff bases-a review. J Sci Ind Res 68:181–187
Przybylski PHA, Pyta K, Brzezinski B, Bartl F (2009) Biological properties of schiff bases and azo derivatives of phenols. Curr Org Chem 13:124
da Silva CM, da Silva DL, Modolo LV, Alves RB, de Resende MA, Martins CVB (2011) Schiff bases: a short review of their antimicrobial activities. J Adv Res 2:1–8
Tirkistani FAA (1998) Thermal analysis of some chitosan Schiff bases. Poly Degrad Stab 60:67–70
dos Santos JE, Dockal ER, Cavalheiro ÉTG (2005) Synthesis and characterization of Schiff bases from chitosan and salicylaldehyde derivatives. Carbohydr Polym 60:277–282
Dash M, Chiellini F, Ottenbrite RM, Chiellini E (2011) Chitosan—a versatile semi- synthetic polymer in biomedical Applications. Progr Polym Sci 36:981–1014
Alexandra Mu˜noz-Bonilla, Marta Fernández-García (2012) Polymeric materials with antimicrobial activity. Progr Polym Sci 37:281–339
Sharma A (2012) Extraction of lead by dithiocarbmate derivative of chitosan. Int J Chem Environm Pharmaceut Res 3:170–173
Qin Y, Liu S, Xing R, Yu H, Li K, Meng X, Li R, Li P (2012) Synthesis and characterization of dithiocarbamate chitosan derivatives with enhanced antifungal activity. Carbohydr Polym 89:388–393
Frisch MJ et al (2004) Gaussian 03. Revision C02. Gaussian, Inc, Wallingford CT
Becke AD (1993) Density functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Hehre WJ, Ditchfield R, Pople JA (1972) Self-consistent molecular orbital methods. XII. Further extensions of Gaussian-type Basis sets for use in molecular orbital studies of organic molecules. J Chem Phy 56:2257–2261
Weininger D (1988) SMILES, a chemical language and information system. 1. Introduction to methodology and encoding rules. J Chem Infor Comp Sci 28:31–36
O’Boyle NM, Banck M, James CA, Morley C, Vandermeersch T, Hutchison GR (2011) Open Babel: an open chemical toolbox. J Cheminform 3:33
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (2001) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Delivery Rev 46:3–26
Veber DF, Johnson SR, Cheng HY, Smith BR, Ward KW, Kopple KD (2002) Molecular properties that influence the oral bioavailability of drug candidates. J Med Chem 45:2615–2623
Ertl P, Rohde B, Selzer P (2000) Fast calculation of molecular polar surface area as a sum of fragment-based contributions and its application to the prediction of drug transport properties. J Med Chem 43:3714–3717
Verma A (2012) Lead finding from phyllanthus debelis with hepatoprotective potentials. Asian Pacific J Tropical Biomed 2:S1735–S7
Zhao YH, Abraham MH, Le J, Hersey A, Luscombe CN, Beck G et al (2002) Rate-limited steps of human oral absorption and QSAR studies. Pharmaceut Res 19:1446–1457
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noushad, T., Alikutty, P., Basila, H. et al. A comparative study on the druggability of Schiff bases and dithiocarbamate derivatives of chitosan. Polym. Bull. 73, 2165–2177 (2016). https://doi.org/10.1007/s00289-016-1601-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-016-1601-y