Summary
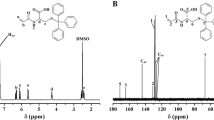
A new fluorescent initiator fluorescein 2-bromoisobutyrate (Flu-Br) was synthesized. The use of Flu-Br as initiator, Me6TREN as ligand and CuCl as catalyst by atom transfer radical polymerization allowed for chain-end fluoresceined poly(N-isopropylacrylamide) (Flu-PNIPAM) in one step. The polymerization reached high conversion (65%) and low polydispersity (PDI) (1.15∼1.28). The linearity plot of the Mn,GPC and Mn,NMR against conversion and the low PDI revealed the well-controlled polymerization by ATRP. In addition, N-isopropylacrylamide (NIPAM) was copolymerized with hydrophilic N,N-dimethylacrylamide (DMAM) using Flu-Br initiator by ATRP. By changing the feed ratio of NIPAM to DMAM, it was very easy to obtain thermo-sensitive fluorescent copolymers with proper lower critical solution temperature (36.0±0.2 °C, 38.0±0.2 °C). The pH dependence on fluorescence intensity of Flu-PNIPAM displayed a similar behavior to the parent fluorescein.
Similar content being viewed by others
References
Velonia K, Rowan AE, Nolte RJ (2002) Am Chem Soc 124:4224
Ding Z, Fong RB, Long CJ, Stayton PS, Hoffman AS (2001) Nature 411:59
Chiefari J, Chong YK, Ercole F, Krstina J, Jeffery J, Le TP, Mayadunne RT, Meijs GF, Moad CL, Moad G, Rizzardo E, Thang SH (1998) Macromolecules 31:5559
Chiefari J, Mayadunne RT, Moad CL, Moad G, Rizzardo E, Postma A, Skidmore, MA, Thang, SH (2003) Macromolecules 36:2273
Wang JS, Matyjaszewski K (1995) J Am Chem Soc 117:5614
Kamigaito M, Ando T, Sawamoto M (2001) Chem Rev 101:3689
Matyjaszewski K, Xia J (2001) Chem Rev 101:292
Hoffman AS, Stayton PS (2004) Macromol Symp 207:139
Stayton PS, Shimoboji T, Long C, Chilkoti A, Chen GH, Harris JM, Hoffman AS (1995) Nature 378:472
Ding Z, Fong RB, Long CJ, Stayton PS, Hoffman AS (2001) Nature 411:59
Chilkoti A, Dreher MR, Meyer DE, Raucher D (2002) Adv Drug Delivery Rev 54:613
Zhu MQ, Wang LQ, Exarhos GJ, Li AD (2004) J Am Chem Soc 126:2656
DiLoreto Jr DA, Das T, del Cerro C, Cox C, del Cerro M (1997) Curr Eye Res 16:1159
Wang S, Gaylord BS, Bazan GC (2003) J Am Chem Soc 126:5446
Wischke C, Borchert HH (2006) Pharmazie 61:770
Bontempo D, Li RC, Ly T, Brubaker CE, Maynard HD (2005) Chem Commun 37:4702
Disney M, Zheng J, Swager T, Seeberger P (2004) J Am Chem Soc 126:13343
Morawetz H, Allen, NS, Rabek JF, In New Trends in the Photochemistry of Polymers; Elsevier: London, 1985
Kim CS, Oh SM, Kim S, Cho CG (1998) Macromol Rapid Commun 19:91
Cheng ZP, Zhu XL, Zhu J, Lu JM, Yu JH (2005), J Macromol Sci Pure Appl Chem 42:34
Erdogan M, Hepuzer Y, Cianga I, Yagci Y, Pekcan O (2003) J Phys Chem 107:8363
Duan Q, Miura Y, Narumi A, Shen X, Sato S, Satoh T, Kakuchi T (2006) J Polym Sci Part- Polym Chem 44:1117
Ciampolini M, Nardi N (1966) Inorg Chem 5:41
Couet J, Biesalski M (2006) Macromolecules 39:7258
Fischer H (2001) Chem Rev 101:3581
Schilli C, Lanzendörfer MG, Müller AH (2002) Macromolecules 35:6819
Feil H, Bae YH, Feijen J, Kim SW (1993) Macromolecules 26:2496
Siu MH, Zhang G, Wu C (2002) Macromolecules 35:2723
Zhang G, Wu C, Jiang M (2005) Polym Bull 4:82
Khokhlov AR, Khalatur PG (1999) Phys Rev Lett 82:3456
Timoshenko EG, Kuznetsov YA (2000) Chem Phys 112:8163
Lee SB, Russell AJ, Matyjaszewski K (2003) Biomacromolecules 4:1386
Liu SQ, Tong YW, Yang Y (2005) Biomaterials 26:5064
Guan X, Liu X, Su Z, Liu P (2006) React & Func Polym 66:1227
Martin MM, Lindqvist L (1975), J Lumin 10:381
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, X., Zhang, L., Meng, L. et al. Synthesis of poly(N-isopropylacrylamide) by ATRP using a fluorescein-based initiator. Polym. Bull. 59, 195–206 (2007). https://doi.org/10.1007/s00289-007-0754-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-007-0754-0