Summary
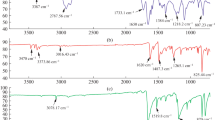
Bis(m-aminophenyl)diselenide was synthesized by diazotation of m-nitroaniline followed by incorporation of potassium selenium cyanate, and the reduction of the nitro groups by addition of tin and concentrated HCl. This dihydrochloride monomer was polymerised using ammonium persulphate in 0.25 M HCl as oxidizing agent. Copolymers of aniline with bis(m-aminophenyl)diselenide were prepared by oxidation of diselenide and aniline mixtures, at several mole ratios of aniline in the feed (f1), with the same oxidizing agent. In the all the range of polymers analysed there are more diselenide comonomer units than aniline units. The polymer and copolymers were characterized by FTIR, elemental analysis, thermal stability and electrical conductivity, showing a high thermal stability, with a weight loss of 10% at 400 °C and there is an important effect of groups diselenide on the electrical properties, because conductivities are highly modified when the substitution is in m-position in the aniline ring. Therefore, it is necessary to add a high mole ratio of aniline in the feed to obtain copolymers with conductivities within the semi-conduction range.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sánchez, C.O., Bustos, C.J., Del Valle, M.A. et al. Synthesis and Characterization of Poly[bis(3-aminophenyl) diselenide] and Poly[bis(3-aminophenyl) diselenide-co-aniline]. Polymer Bulletin 51, 95–102 (2003). https://doi.org/10.1007/s00289-003-0208-2
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00289-003-0208-2