Abstract.
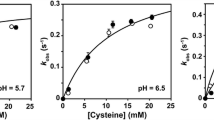
Cysteine 195 in isocitrate lyase from Escherichia coli has been replaced by directed mutagenesis. Substitution by Ser yields enzyme with a kcat that is 0.03% that of wild type, and substitution by Ala, Gly, Thr, or Val yields completely inactive enzyme. The present results are consistent with a functional role of Cys 195.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 26 March 1997 / Accepted: 29 April 1997
Rights and permissions
About this article
Cite this article
Rehman, A., McFadden, B. Cysteine 195 Has a Critical Functional Role in Catalysis by Isocitrate Lyase from Escherichia coli . Curr Microbiol 35, 267–269 (1997). https://doi.org/10.1007/s002849900251
Issue Date:
DOI: https://doi.org/10.1007/s002849900251