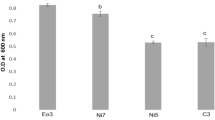
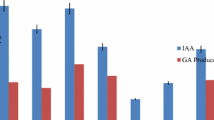
Abstract
In the present study, bacterial and fungal endophytes are isolated from Calotropis procera, a drought-resistant plant and studied for their role in plant growth promotion. Among bacterial sp. Enterobacter cloacae subsp. cloacae strain CPR5B and fungus, Penicillium citrinum strain CPL1F, were identified as potent endophytes as both strains were able to produce Indole Acetic Acid (IAA) and 1-aminocyclopropane-1-carboxylic acid (ACC) deaminase and solubilize phosphate. Penicillium citrinum CPL1F also been shown to produce siderophore. The IAA production was observed to be 94.28 μg/mL and 17.1 μg/mL for bacterial and fungal sp., respectively. The phosphate solubilization was observed to be 76.41 μg/mL and 114.57 μg/mL, respectively. The in vitro plant treatment studies with bacterium and fungus under irrigated and non-irrigated conditions showed that both strains had promoted plant growth in both conditions with respect to their control. Both the strains showed significant changes in most of the growth parameters under endophyte-treated irrigated and non-irrigated conditions, suggesting their stress-dependent plant growth promotion. The present findings will contribute to exploring endophytes that enhance plant growth in adverse conditions and act as plant growth-promoting endophytes.






Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
Abbreviations
- ROS:
-
Reactive oxygen species
- NaCl:
-
Sodium chloride
- NA:
-
Nutrient agar
- PDA:
-
Potato dextrose agar
- PDB:
-
Potato dextrose broth
- NBRIP:
-
National Botanical Research Institute’s Phosphate growth medium
- PKV:
-
Pikovskaya agar medium
- CAS:
-
Chrome azurol S
- HDTMA:
-
Hexadecyltrimethylammonium bromide
- KH2PO4 :
-
Monopotassium phosphate
- FeCl3 :
-
Ferric chloride
- Pmol:
-
Picomole
- μl:
-
Microliter
- μg:
-
Microgram
- mM:
-
Millimolar
- g/L:
-
Gram per liter
- 16S rRNA:
-
16S ribosomal Ribonucleic acid
- BLAST:
-
Basic local alignment search tool
- bp:
-
Base pair
- cm:
-
Centimeter
- DNA:
-
Deoxyribonucleic acid
- g:
-
Gram
- IAA:
-
Indole-3-acetic acid
- ACC:
-
1-Aminocyclopropane-1-carboxylate (ACC)
- NCBI:
-
National Center for Biotechnology Information
- nm:
-
Nanometer
- PCR:
-
Polymerase chain reaction
- PGPE:
-
Plant growth-promoting endophytes
References
Khare E, Mishra J, Arora NK (2018) Multifaceted interactions between endophytes and plant: developments and prospects. Front Microbiol 9:2732. https://doi.org/10.3389/fmicb.2018.02732
Kaur T, Devi R, Kour D et al (2021) Plant growth promoting soil microbiomes and their potential implications for agricultural and environmental sustainability. Biologia 76:2687–2709. https://doi.org/10.1007/s11756-021-00806-w
Arnold AE, Henk DA, Eells RL, Lutzoni F, Vilgalys R (2007) Diversity and phylogenetic affinities of foliar fungal endophytes in loblolly pine inferred by culturing and environmental PCR. Mycologia 99:185–206. https://doi.org/10.1080/15572536.2007.11832578
Eid AM, Fouda A, Abdel-Rahman MA, Salem SS, Elsaied A, Oelmüller R, Hijri M, Bhowmik A, Elkelish A, Hassan SE (2021) Harnessing bacterial endophytes for promotion of plant growth and biotechnological applications: an overview. Plants 10:935. https://doi.org/10.3390/plants10050935
Patel S, Jinal HN, Amaresan N (2017) Isolation and characterization of drought resistance bacteria for plant growth promoting properties and their effect on chilli (Capsicum annuum) seedling under salt stress. Biocatal Agric Biotechnol 12:85–89. https://doi.org/10.1016/j.bcab.2017.09.002
Etesami H, Maheshwari DK (2018) Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: action mechanisms and future prospects. Ecotoxicol Environ Saf 156:225–246. https://doi.org/10.1016/j.ecoenv.2018.03.013
Fahad S, Bajwa AA, Nazir U, Anjum SA, Farooq A, Zohaib A, Sadia S, Nasim W, Adkins S, Saud S, Ihsan MZ (2017) Crop production under drought and heat stress: plant responses and management options. Front Plant Sci 8:1147. https://doi.org/10.3389/fpls.2017.01147
Cassán F, Vanderleyden J, Spaepen S (2014) Physiological and agronomical aspects of phytohormone production by model plant-growth-promoting rhizobacteria (PGPR) belonging to the genus Azospirillum. J Plant Growth Regul 33:440–459. https://doi.org/10.1007/s00344-013-9362-4
Dudeja SS, Suneja-Madan P, Paul M, Maheswari R, Kothe E (2021) Bacterial endophytes: molecular interactions with their hosts. J Basic Microbiol 61:475–505. https://doi.org/10.1002/jobm.202000657
Baron NC, Rigobelo EC (2022) Endophytic fungi: a tool for plant growth promotion and sustainable agriculture. Mycology 13:39–55. https://doi.org/10.1080/21501203.2021.1945699
Khan AL, Halo BA, Elyassi A, Ali S, Al-Hosni K, Hussain J, Al-Harrasi A, Lee IJ (2016) Indole acetic acid and ACC deaminase from endophytic bacteria improves the growth of Solanum lycopersicum. Electron J Biotechnol 21:58–64. https://doi.org/10.1016/j.ejbt.2016.02.001
Chandwani S, Amaresan N (2022) Role of ACC deaminase producing bacteria for abiotic stress management and sustainable agriculture production. Environ Sci Pollut Res 20:1–7. https://doi.org/10.1007/s11356-022-18745-7
Gamalero E, Glick BR (2015) Bacterial modulation of plant ethylene levels. Plant Physiol 169:13–22. https://doi.org/10.1104/pp.15.00284
Yadav LS, Kushwaha V, Jain A (2020) Isolation and screening of phosphate solubilizing fungi from okra rhizosphere soil and their effect on the growth of okra plant (Abelmoschous esculentus L.). Trop Plant Res 7:277–284. https://doi.org/10.22271/tpr.2020.v7.i2.033
Raymond NS, Gómez-Muñoz B, van der Bom FJ, Nybroe O, Jensen LS, Müller-Stöver DS, Oberson A, Richardson AE (2021) Phosphate-solubilising microorganisms for improved crop productivity: a critical assessment. New Phytol 229:1268–1277. https://doi.org/10.1111/nph.16924
Singh KP (2023) Comparative analysis: physiological and biochemical characterization of phosphate solubilization by Pseudomonas and Bacillus species from various agricultural areas of Dehradun, Uttarakhand, India. Int J Life Sci 11(3):219–227. https://doi.org/10.5281/zenodo.8395512
Ahmad A, Moin SF, Liaqat I, Saleem S, Muhammad F, Mujahid T, Zafar U (2023) Isolation, solubilization of inorganic phosphate, and production of organic acids by individual and co-inoculated microorganisms. Geomicrobiol J 40:111–121. https://doi.org/10.1080/01490451.2022.2124329
Maheshwari R, Bhutani N, Suneja P (2019) Screening and characterization of siderophore producing endophytic bacteria from Cicer arietinum and Pisum sativum plants. J Appl Biol Biotechnol 7:7–4. https://doi.org/10.7324/jabb.2019.70502
Wittenwiler M (2007) Mechanisms of iron mobilization by siderophores. Master Studies in Environmental Sciences Master, ETH Zürich
Siddiqui ZA (2005) PGPR: prospective biocontrol agents of plant pathogens. In: PGPR: biocontrol and biofertilization. Springer, Dordrecht, pp 111–142. https://doi.org/10.1007/1-4020-4152-7_4
LeTourneau MK, Marshall MJ, Grant M, Freeze PM, Strawn DG, Lai B, Thomashow LS (2019) Phenazine-1-carboxylic acid-producing bacteria enhance the reactivity of iron minerals in dryland and irrigated wheat rhizospheres. Environ Sci Technol 53(24):14273–14284. https://doi.org/10.1021/acs.est.9b03962
Hossain GM, Ghazali AH, Islam T, Mia MA (2022) Enhanced nutrient accumulation in non-leguminous crop plants by the application of endophytic bacteria Bacillus species. In: Islam MT, Rahman M, Pandey P (eds) Bacilli in agrobiotechnology. Bacilli in climate resilient agriculture and bioprospecting. Springer, Cham, pp 349–364. https://doi.org/10.1007/978-3-030-85465-2_16
Francis JK (2003) Calotropis procera. US Department of Agriculture, Forest Service, International Institute of Tropical Forestry, Puerto Rico
Hassan LM, Galal TM, Farahat EA, El-Midany MM (2015) The biology of Calotropis procera (Aiton) WT. Trees 29:311–320. https://doi.org/10.1007/s00468-015-1158-7
Shokry AM, Al-Karim S, Ramadan A, Gadallah N, Al Attas SG, Sabir JS, Bahieldin A (2014) Detection of a Usp-like gene in Calotropis procera plant from the de novo assembled genome contigs of the high-throughput sequencing dataset. C R Biol 337:86–94. https://doi.org/10.1016/j.crvi.2013.12.008
Joshi S, Singh AV, Prasad B (2018) Enzymatic activity and plant growth promoting potential of endophytic bacteria isolated from Ocimum sanctum and Aloe vera. Int J Curr Microbiol App Sci 7:2314–2326. https://doi.org/10.20546/ijcmas.2018.706.27
Gordon SA, Weber RP (1951) Colorimetric estimation of indoleacetic acid. Plant Physiol 26(1):192
Jasim B, John Jimtha C, Jyothis M, Radhakrishnan EK (2013) Plant growth promoting potential of endophytic bacteria isolated from Piper nigrum. Plant Growth Regul 71:1–11. https://doi.org/10.1007/s10725-013-9802-y
Pirhadi M, Enayatizamir N, Motamedi H, Sorkheh K (2018) Impact of soil salinity on diversity and community of sugarcane endophytic plant growth promoting bacteria (Saccharum officinarum L. Var. CP48). Appl Ecol Environ 16:725–739. https://doi.org/10.15666/aeer/1601_725739
Pandey D, Kehri HK, Zoomi I, Chaturvedi S, Chaudhary KL (2022) Seasonal effect on the diversity of soil fungi and screening for arsenic tolerance and their remediation. J Appl Biol Biotechnol 10:4–6. https://doi.org/10.7324/JABB.2022.10s106
Murphy JAMES, Riley JP (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36. https://doi.org/10.1016/S0003-2670(00)88444-5
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophore. Ann Biochem 160:47–56. https://doi.org/10.1016/0003-2697(87)90612-9
White TJ, Bruns T, Lee SJWT, Taylor JL (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc 18:315–322
Christakis CA, Daskalogiannis G, Chatzaki A, Markakis EA, Mermigka G, Sagia A, Sarris PF (2021) Endophytic bacterial isolates from halophytes demonstrate phytopathogen biocontrol and plant growth promotion under high salinity. Front Microbiol 12:681567. https://doi.org/10.3389/fmicb.2021.681567
Khan AL, Lee IJ (2013) Endophytic Penicillium funiculosum LHL06 secretes gibberellin that reprograms Glycine max L. growth during copper stress. BMC Plant Biol 13:1–14. https://doi.org/10.1186/1471-2229-13-86
Chen C, Xin K, Liu H et al (2017) Pantoea alhagi, a novel endophytic bacterium with ability to improve growth and drought tolerance in wheat. Sci Rep 7:41564. https://doi.org/10.1038/srep41564
Duca D, Lorv J, Patten CL, Rose D, Glick BR (2014) Indole-3-acetic acid in plant–microbe interactions. Antonie Van Leeuwenhoek 106:85–125. https://doi.org/10.1007/s10482-013-0095-y
Kour D, Yadav AN (2022) Bacterial mitigation of drought stress in plants: current perspectives and future challenges. Curr Microbiol 79:1–19. https://doi.org/10.1007/s00284-022-02939-w
Chandra D, Srivastava R, Sharma AK (2018) Influence of IAA and ACC deaminase producing fluorescent pseudomonads in alleviating drought stress in wheat (Triticum aestivum). Agric Res 7:290–299. https://doi.org/10.1007/s40003-018-0305-y
Fouda A, Eid AM, Elsaied A, El-Belely EF, Barghoth MG, Azab E, Hassan SED (2021) Plant growth-promoting endophytic bacterial community inhabiting the leaves of Pulicaria incisa (Lam.) DC inherent to arid regions. Plants 10(1):76. https://doi.org/10.3390/plants10010076
Jilani G, Zhang D, Chaudhry AN, Iqbal Z, Ikram M, Bashir M (2021) Role of phosphate-solubilising microorganisms in agricultural development. In: Mohamed HI, El-Beltagi HEDS, Abd-Elsalam KA (eds) Plant growth-promoting microbes for sustainable biotic and abiotic stress management. Springer, Cham. https://doi.org/10.1007/978-3-030-66587-6_17
Suebrasri T, Harada H, Jogloy S, Ekprasert J, Boonlue S (2020) Auxin-producing fungal endophytes promote growth of sunchoke. Rhizosphere 16:100271. https://doi.org/10.1016/j.rhisph.2020.100271
Oren A (2008) Microbial life at high salt concentrations: phylogenetic and metabolic diversity. Saline Syst 4:1–13. https://doi.org/10.1186/1746-1448-4-2
Ali B, Wang X, Saleem MH, Sumaira Hafeez A, Afridi MS, Khan S, Zaib-Un-Nisa Ullah I, Amaral Júnior ATd, Alatawi A, Ali S (2022) PGPR-mediated salt tolerance in maize by modulating plant physiology, antioxidant defense, compatible solutes accumulation and bio-surfactant producing genes. Plants 11:345. https://doi.org/10.3390/plants11030345
Agha MS, Abbas MA, Sofy MR, Haroun SA, Mowafy AM (2021) Dual inoculation of Bradyrhizobium and Enterobacter alleviates the adverse effect of salinity on Glycine max seedling. Notulae Botanicae. Hortic Agrobotanici Cluj-Napoca 49(3):12461. https://doi.org/10.15835/nbha49312461
Dhakar K, Sharma A, Pandey A (2014) Cold, pH and salt tolerant Penicillium spp. inhabit the high-altitude soils in Himalaya, India. World J Microbiol Biotechnol 30:1315–1324. https://doi.org/10.1007/s11274-013-1545-4
Hamayun M, Khan N, Khan MN, Qadir M, Hussain A, Iqbal A, Lee IJ (2021) Antimicrobial and plant growth-promoting activities of bacterial endophytes isolated from Calotropis procera (Ait.) WT Aiton. Biocell 45(2):363. https://doi.org/10.32604/biocell.2021.013907
Ullah I, Al-Ghamdi KM, Anwar Y, Mahyoub A, J, (2020) Exploring the Insect Control and Plant Growth Promotion Potentials of Endophytes Isolated From Calotropis procera Present in Jeddah KSA. Nat Prod Commun 15:3
Khan SA, Humayun M, Yoon H et al (2008) Plant growth promotion and Penicillium citrinum. BMC Microbiol 8:231. https://doi.org/10.1186/1471-2180-8-231
Kedar A, Rathod D, Yadav A, Agarka G, Rai M (2014) Endophytic Phoma sp. isolated from medicinal plants promote the growth of Zea mays. Nusantara Biosci 6:2. https://doi.org/10.13057/nusbiosci/n060205
Funding
Sonali Jaiswal has received Junior Research Fellowship from University Grants Commission (UGC) India [UGC Reference no: 960/ (CSIR-UGC NET JUNE 2017)] to carry out the present research work.
Author information
Authors and Affiliations
Contributions
SJ made substantial contributions to the conception or design of the work, made the analysis, and interpretation of data in the work. AO has drafted the work or revised it critically for important intellectual content. SKM has approved the version to be published, agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved, made proofreading of manuscript, and gave suggestions to improve it. All authors read and approved the final manuscript. This is a part of Sonali Jaiswal’s Ph.D. program.
Corresponding author
Ethics declarations
Conflict of interest
There was no conflict of interest among the authors.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jaiswal, S., Ojha, A. & Mishra, S.K. Assessment of Plant Growth-Promoting Parameters of Endophytes Isolated from Calotropis procera and Their Performance Under Irrigated and Non-irrigated Conditions. Curr Microbiol 81, 49 (2024). https://doi.org/10.1007/s00284-023-03570-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03570-z