Abstract
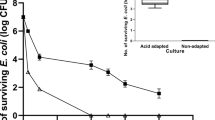
Escherichia coli Nissle 1917 (EcN) is one of the probiotics that has drawn more attention from researchers in recent days as it extends many host beneficial effects. EcN is being used as a treatment regimen especially for gastrointestinal disorders for more than 100 years. Apart from its clinical applications in its original form, EcN is being genetically engineered to meet the therapeutic requirements which ultimately led to the gradual transformation of EcN from being a mere food supplement to a complex therapeutic agent. However, comprehensive analysis of physiological characterization of EcN is inadequate. In this study, we have systematically studied various physiological parameters and found that EcN grows very well at the normal as well as at stressful conditions such as temperature (30, 37 and 42 °C), nutritional (minimal and LB), pH (ranging from 3 to 7) and osmotic stress (0.4 M NaCl, 0.4 M KCl, 0.4 M Sucrose and salt conditions). However, EcN shows nearly onefold reduction in viability at extreme acidic conditions (pH 3 and 4). It produces biofilm and curlin very efficiently compared to the laboratory strain MG1655. Through genetic analysis we have also shown that EcN exhibits high level of transformation efficiency and greater ability to retain heterogenous plasmid. Very interestingly, we have found that EcN is highly resistant to P1 phage infection. Since, EcN is being exploited largely for its clinical and therapeutic applications, the results that we have reported here would add more value and further expand its scope in clinical and biotechnological research.





Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this published article (and its supplementary information files).
Abbreviations
- EcN:
-
E. coli Nissle 1917
- IBD:
-
Inflammatory Bowel Disease
- LB media:
-
Luria Bertani media
- PBS:
-
Phosphate Buffered Saline
- CFU:
-
Colony Forming Units
References
Behnsen J, Deriu E, Sassone-Corsi M, Raffatellu M (2013) Probiotics: properties, examples, and specific applications. Cold Spring Harbor Perspect Med. 3(3):a010074. https://doi.org/10.1101/cshperspect.a010074
Sonnenborn U (2016) Escherichia coli strain Nissle 1917-from bench to bedside and back: history of a special Escherichia coli strain with probiotic properties. FEMS Microbiol Lett 363(19):fnw212. https://doi.org/10.1093/femsle/fnw212
Frank DN, Pace NR (2008) Gastrointestinal microbiology enters the metagenomics era. Curr Opin Gastroenterol 24(1):4–10. https://doi.org/10.1097/MOG.0b013e3282f2b0e8
Valdes AM, Walter J, Segal E, Spector TD (2018) Role of the gut microbiota in nutrition and health. BMJ (Clin Res ed.) 361:k2179. https://doi.org/10.1136/bmj.k2179
Sartor RB (2004) Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: antibiotics, probiotics, and prebiotics. Gastroenterology 126(6):1620–1633. https://doi.org/10.1053/j.gastro.2004.03.024
Kechagia M, Basoulis D, Konstantopoulou S, Dimitriadi D, Gyftopoulou K, Skarmoutsou N, Fakiri EM (2013) Health benefits of probiotics: a review. ISRN Nutr. https://doi.org/10.5402/2013/481651
Kaur IP, Chopra K, Saini A (2002) Probiotics: potential pharmaceutical applications. Eur J Pharm Sci 15(1):1–9. https://doi.org/10.1016/s0928-0987(01)00209-3
Khalighi A, Behdani R, Kouhestani S (2016) Probiotics: a comprehensive review of their classification, mode of action and role in human nutrition. Probiotics Prebiotics Human Nutr Health 10:63646. https://doi.org/10.5772/63646
Sonnenborn U, Schulze J (2009) The non-pathogenic Escherichia coli strain Nissle 1917–features of a versatile probiotic. Microb Ecol Health Dis 21(3–4):122–158. https://doi.org/10.3109/08910600903444267
Miller JH (1992) A short course in bacterial genetics: a laboratory manual and handbook for Escherichia coli and related bacteria. Plainview
Sambrook J, Russel DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor, New York
Demerec M, Adelberg EA, Clark AJ, Hartman PE (1966) A proposal for a uniform nomenclature in bacterial genetics. Genetics 54(1):61–76. https://doi.org/10.1093/genetics/54.1.61
Berlyn MK (1998) Linkage map of Escherichia coli K-12, edition 10: the traditional map. Microbiol Mol Biol Rev 62(3):814–984. https://doi.org/10.1128/MMBR.62.3.814-984.1998
O’Toole GA (2011) Microtiter dish biofilm formation assay. J Visual Exp 47:2437. https://doi.org/10.3791/2437
Blum-Oehler G, Oswald S, Eiteljörge K, Sonnenborn U, Schulze J, Kruis W, Hacker J (2003) Development of strain-specific PCR reactions for the detection of the probiotic Escherichia coli strain Nissle 1917 in fecal samples. Res Microbiol 154(1):59–66. https://doi.org/10.1016/s0923-2508(02)00007-4
Pradhan S, Weiss AA (2020) Probiotic properties of Escherichia coli Nissle in human intestinal organoids. mBio 11(4):e01470-20. https://doi.org/10.1128/mBio.01470-20
Grozdanov L, Raasch C, Schulze J, Sonnenborn U, Gottschalk G, Hacker J, Dobrindt U (2004) Analysis of the genome structure of the nonpathogenic probiotic Escherichia coli strain Nissle 1917. J Bacteriol 186(16):5432–5441. https://doi.org/10.1128/JB.186.16.5432-5441.2004
Gill J, Lau K, Lee T, Lucas H (2019) Escherichia coli Nissle 1917 forms biofilm and outgrows Escherichia coli K12 in a temperature-dependent manner. UJEMI 24:1–8
Kim D, Kim Y, Yoon SH (2021) Development of a genome-scale metabolic model and phenome analysis of the probiotic Escherichia coli strain Nissle 1917. Int J Mol Sci 22(4):2122. https://doi.org/10.3390/ijms22042122
Crook N, Ferreiro A, Gasparrini AJ, Pesesky MW, Gibson MK, Wang B, Sun X, Condiotte Z, Dobrowolski S, Peterson D, Dantas G (2019) Adaptive strategies of the candidate probiotic E. coli Nissle in the mammalian gut. Cell Host Microbe 25(4):499–512. https://doi.org/10.1016/j.chom.2019.02.005
Revelles O, Millard P, Nougayrède JP, Dobrindt U, Oswald E, Létisse F, Portais JC (2013) The carbon storage regulator (Csr) system exerts a nutrient-specific control over central metabolism in Escherichia coli strain Nissle 1917. PloS One 8(6):e66386. https://doi.org/10.1371/journal.pone.0066386
Li F, Xiong XS, Yang YY, Wang JJ, Wang MM, Tang JW, Liu QH, Wang L, Gu B (2021) Effects of NaCl concentrations on growth patterns, phenotypes associated with virulence, and energy metabolism in Escherichia coli BW25113. Front Microbiol 12:705326. https://doi.org/10.3389/fmicb.2021.705326
Bidlas E, Lambert RJ (2008) Comparing the antimicrobial effectiveness of NaCl and KCl with a view to salt/sodium replacement. Int J Food Microbiol 124(1):98–102. https://doi.org/10.1016/j.ijfoodmicro.2008.02.031
Gandhi A, Cui Y, Zhou M, Shah NP (2014) Effect of KCl substitution on bacterial viability of Escherichia coli (ATCC 25922) and selected probiotics. J Dairy Sci 97(10):5939–5951. https://doi.org/10.3168/jds.2013-7681
Yim J, Cho SW, Kim B, Park S, Han YH, Seo SW (2020) Transcriptional profiling of the probiotic Escherichia coli Nissle 1917 strain under simulated microgravity. Int J Mol Sci 21(8):2666. https://doi.org/10.3390/ijms21082666
Prilassnig M, Wenisch C, Daxboeck F, Feierl G (2007) Are probiotics detectable in human feces after oral uptake by healthy volunteers? Wien Klin Wochenschr 119(15–16):456–462. https://doi.org/10.1007/s00508-007-0808-1
Lasaro MA, Salinger N, Zhang J, Wang Y, Zhong Z, Goulian M, Zhu J (2009) F1C fimbriae play an important role in biofilm formation and intestinal colonization by the Escherichia coli commensal strain Nissle 1917. Appl Environ Microbiol 75(1):246–251. https://doi.org/10.1128/AEM.01144-08
Coldewey SM, Hartmann M, Schmidt DS, Engelking U, Ukena SN, Gunzer F (2007) Impact of the rpoS genotype for acid resistance patterns of pathogenic and probiotic Escherichia coli. BMC Microbiol 7:21. https://doi.org/10.1186/1471-2180-7-21
Ramachandran C, Ramakrishnan R, Usha A (2016) Evaluation of safety, antimicrobial activity and probiotic properties of Escherichia coli Nissle 1917 isolated from Idli batter. Res J Biotech 11(7):42–49
Ou B, Yang Y, Tham WL, Chen L, Guo J, Zhu G (2016) Genetic engineering of probiotic Escherichia coli Nissle 1917 for clinical application. Appl Microbiol Biotechnol 100(20):8693–8699. https://doi.org/10.1007/s00253-016-7829-5
Nzakizwanayo J, Kumar S, Ogilvie LA, Patel BA, Dedi C, Macfarlane WM, Jones BV (2015) Disruption of Escherichia coli Nissle 1917 K5 capsule biosynthesis, through loss of distinct kfi genes, modulates interaction with intestinal epithelial cells and impact on cell health. PloS One 10(3):e0120430. https://doi.org/10.1371/journal.pone.0120430
Bury S, Soundararajan M, Bharti R, von Bünau R, Förstner KU, Oelschlaeger TA (2018) The probiotic Escherichia coli Strain Nissle 1917 combats lambdoid bacteriophages stx and λ. Front Microbiol 9:929. https://doi.org/10.3389/fmicb.2018.00929
Acknowledgements
We would like to thank Prof. M. Hussain Munavar for his continuous support and concern. We thank Prof. Ramachandran Murugesan and Prof. Sripathi Kandula for their help and support. We thank Ardeypharm, Germany for providing EcN1917 strain, Dr. Harinarayanan, for providing pCA24N plasmid and Dr. Sutharsan for gifting pHerd30T-sfCherry plasmid. We thank the management of CHRI, CARE for providing the infrastructure facilities. We thank DBT, Govt of India for the extramural research funding. We would like to thank the anonymous reviewers for the comments that have helped in improving the quality of this manuscript.
Funding
This work was supported by the extramural research fund received from DBT, Government of India (BT/PR40731/COT/142/22/2021).
Author information
Authors and Affiliations
Contributions
SM analyzed the data, wrote/reviewed the manuscript and provided resources; SR and RS performed the experiments and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramadevi, S., Shelin, R. & Shanmugaraja, M. Comprehensive Analysis of the Physiological Characterization of Escherichia coli Nissle 1917. Curr Microbiol 80, 150 (2023). https://doi.org/10.1007/s00284-023-03253-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03253-9