Abstract
Infectious diseases are among the leading causes of morbidity and mortality worldwide. Combating them becomes more complex when caused by the pathogens of the ESKAPE group, which are Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa and Enterobacter spp. The purpose of this study was to investigate the repositioning potential of the benzodiazepines clonazepam and diazepam individually and in combination with the antibacterial ciprofloxacin against ESKAPE. The minimum inhibitory concentration and minimum bactericidal concentration against seven American Type Culture Collection (ATCC) reference standard strains and 64 ESKAPE clinical isolates were determined. In addition, the interaction with ciprofloxacin was determined by the checkerboard method and fractional inhibitory concentration index (FICI) of clonazepam against 11 ESKAPE and diazepam against five ESKAPE. We also list the results found and their clinical significance. Benzodiazepines showed similar antibacterial activity against Gram-positive and Gram-negative. The checkerboard and FICI results showed a synergistic effect of these drugs when associated with ciprofloxacin against almost all tested isolates. Viewing the clinical cases studied, benzodiazepines have potential as treatment alternatives. The results allow us to conclude that clonazepam and diazepam, when in combination with ciprofloxacin, have promising activity against ESKAPE, therefore, assuming the position of candidates for repositioning.

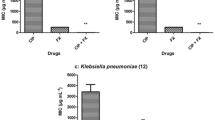
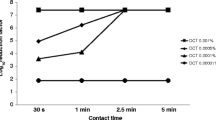
Source: prepared by the author, 2022

Source: prepared by the author, 2022
Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Rentschler S, Kaiser L, Deigner HP (2021) Emerging options for the diagnosis of bacterial infections and the characterization of antimicrobial resistance. Int J Mol Sci 22(1):456. https://doi.org/10.3390/ijms22010456. (PMID: 33466437)
Srivastava S, Singh PK, Vatsalya V et al (2018) Developments in the diagnostic techniques of infectious diseases: rural and urban prospective. Adv Infect Dis 8(3):121–138. https://doi.org/10.4236/aid.2018.83012. (PMID: 30197838)
Messacar K, Parker SK, Todd JK et al (2017) Implementation of rapid molecular infectious disease diagnostics: the role of diagnostic and antimicrobial stewardship. J Clin Microbiol 3:715–723. https://doi.org/10.1128/JCM.02264-16. (PMID: 28031432)
da Rosa TF, Foletto VS, Serafin MB et al (2020) Emerging strategies for ESKAPE treatment. Saúde 46:1. https://doi.org/10.5902/2236583443061
Foletto VS, da Rosa TF, Serafin MB et al (2021) Repositioning of non-antibiotic drugs as an alternative to microbial resistance: a systematic review. Int J Antimicrob Agents 58(3):106380. https://doi.org/10.1016/j.ijantimicag.2021.106380. (PMID: 34166776)
Mulani MS, Kamble EE, Kumkar SN et al (2019) Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: a review. Front Microbiol 10:539. https://doi.org/10.3389/fmicb.2019.00539. (PMID: 30988669)
Rice LB (2008) Federal funding for the study of antimicrobial resistance in nosocomial pathogens: no ESKAPE. J Infect Dis 197(8):1079–1081. https://doi.org/10.1086/533452. (PMID: 18419525)
da Rosa TF, Coelho SS, Foletto VS et al (2020) Alternatives for the treatment of infections caused by ESKAPE pathogens. J Clin Pharm Ther 45(4):863–873. https://doi.org/10.1111/jcpt.13149. (PMID: 32339305)
Bhatia P, Sharma A, George AJ et al (2021) Antibacterial activity of medicinal plants against ESKAPE: an update. Heliyon 7(2):e06310. https://doi.org/10.1016/j.heliyon.2021.e06310. (PMID: 33718642)
De Oliveira DMP, Forde BM, Kidd TJ et al (2020) Antimicrobial resistance in ESKAPE pathogens. Clin Microbiol Rev 33(3):e00181-e219. https://doi.org/10.1128/CMR.00181-19. (PMID: 32404435)
Patil A, Banerji R, Kanojiya P et al (2021) Bacteriophages for ESKAPE: role in pathogenicity and measures of control. Expert Rev Anti Infect Ther 19(7):845–865. https://doi.org/10.1080/14787210.2021.1858800. (PMID: 33261536)
Marturano JE, Lowery TJ (2019) ESKAPE pathogens in bloodstream infections are associated with higher cost and mortality but can be predicted using diagnoses upon admission. Open Forum Infect Dis. 2(12):ofz503. https://doi.org/10.1093/ofid/ofz503. (PMID: 31844639)
Foletto VS, Bottega A, Serafin MB et al (2019) Etiological and antimicrobial susceptibility profile of isolated blood cultures in a university hospital. Saúde (Santa Maria) 45(3):1–15. https://doi.org/10.5902/2236583440016
Miller WR, Murray BE, Rice LB et al (2020) Resistance in vancomycin resistant enterococci. Infect Dis Clin North Am 34(4):751–771. https://doi.org/10.1016/j.idc.2020.08.004. (PMID: 33131572)
Serafin MB, Foletto VS, da Rosa TF et al (2022) Repositioning of disulfiram in association with vancomycin against Enterococcus spp. MDR and XDR Curr Microbiol 79(5):137. https://doi.org/10.1007/s00284-022-02794-9. (PMID: 35303186)
Kumar P (2020) A review on quinoline derivatives as anti-methicillin resistant Staphylococcus aureus (MRSA) agents. BMC Chem 14(1):17. https://doi.org/10.1186/s13065-020-00669-3. (PMID: 32190843)
Diekema DJ, Hsueh PR, Mendes RE et al (2019) The microbiology of bloodstream infection: 20-year trends from the SENTRY antimicrobial surveillance program. Antimicrob Agents Chemother 63(7):e00355-e419. https://doi.org/10.1128/AAC.00355-19. (PMID: 31010862)
Perdigão Neto LV, Oliveira MS, Orsi TD et al (2020) Alternative drugs against multiresistant gram-negative bacteria. J Glob Antimicrob Resist 23:33–37. https://doi.org/10.1016/j.jgar.2020.07.025. (PMID: 32822906)
Jin S, Lee JY, Park JY et al (2020) Xpert Carba-R assay for detection of carbapenemase-producing organisms in patients admitted to emergency rooms. Medicine (Baltimore) 99(50):e23410. https://doi.org/10.1097/MD.0000000000023410. (PMID: 33327265)
Salomão MC, Guimarães T, Duailibi DF et al (2017) Carbapenem-resistant enterobacteriaceae in patients admitted to the emergency department: prevalence, risk factors, and acquisition rate. J Hosp Infect 97(3):241–246. https://doi.org/10.1016/j.jhin.2017.08.012. (PMID: 28826688)
Ashburn TT, Thor KB (2004) Drug repositioning: identifying and developing new uses for existing drugs. Nat Rev Drug Discov 3:673–683. https://doi.org/10.1038/nrd1468. (PMID: 15286734)
Luo H, Li M, Yang M et al (2021) Biomedical data and computational models for drug repositioning: a comprehensive review. Brief Bioinform 22(2):1604–1619. https://doi.org/10.1093/bib/bbz176. (PMID: 32043521)
Serafin MB, Hörner R (2018) Drug repositioning, a new alternative in infectious diseases. Braz J Infect Dis 22(3):252–256. https://doi.org/10.1016/j.bjid.2018.05.007. (PMID: 29963991)
da Rosa TF, Machado CS, Serafin MB et al (2021) Repurposing of escitalopram oxalate and clonazepam in combination with ciprofloxacin and sulfamethoxazole-trimethoprim for treatment of multidrug resistant microorganisms and evaluation of the cleavage capacity of plasmid DNA. Can J Microbiol 67(8):599–612. https://doi.org/10.1139/cjm-2020-0546. (PMID: 33481681)
Mouthon F, Charveriat M. (2014) Use of clonazepam in combination with antibiotic in the treatment of bacterially induced meningitis. WO Patent 2014–2803365A1, filled May 14, 2013, issued Nov 19, 2014.
Neves BJ, Braga RC, Bezerra JC et al (2015) In silico repositioning-chemogenomics strategy identifies new drugs with potential activity against multiple life stages of Schistosoma mansoni. PLoS Negl Trop Dis. 9(1):e3435. https://doi.org/10.1371/journal.pntd.0003435. (PMID: 25569258)
Holanda MA, da Silva CR, de A Neto JB et al (2021) Evaluation of the antifungal activity in vitro of midazolam against fluconazole-resistant Candida spp. isolates. Future Microbiol. 16(2):71–81. https://doi.org/10.2217/fmb-2020-0080. (PMID: 33459560)
Elmezayen AD, Al-Obaidi A, Şahin AT et al (2021) Drug repurposing for coronavirus (COVID-19): in silico screening of known drugs against coronavirus 3CL hydrolase and protease enzymes. J Biomol Struct Dyn 39(8):2980–2992. https://doi.org/10.1080/07391102.2020.1758791. (PMID: 32306862)
Hosomi K, Fujimoto M, Ushio K et al (2018) An integrative approach using real-world data to identify alternative therapeutic uses of existing drugs. PLoS ONE 13(10):e024648. https://doi.org/10.1371/journal.pone.0204648. (PMID: 30300381)
Karakida T, Onuma K, Saito MM et al (2019) Potential for drug repositioning of midazolam for dentin regeneration. Int J Mol Sci 20(3):670. https://doi.org/10.3390/ijms20030670. (PMID: 30720745)
Cordeiro L, Diniz-Neto H, Figueiredo P et al (2020) Potential of 2-Chloro-N-(4-fluoro-3-nitrophenyl)acetamide against Klebsiella pneumoniae and in vitro toxicity analysis. Mol 25(17):3959. https://doi.org/10.3390/molecules25173959. (PMID: 32877986)
Mandal A, Sinha C, Kumar JA et al (2010) An investigation on in vitro and in vivo antimicrobial properties of the antidepressant: amitriptyline hydrochloride. Braz J Microbiol 41(3):635–645. https://doi.org/10.1590/S1517-83822010000300014. (PMID: 24031539)
Naloto DCC, Lopes FC, Barberato-Filho S et al (2016) Prescription of benzodiazepines for adults and older adults from a mental health clinic. Cien Saude Colet 21:1267–1276. https://doi.org/10.1590/1413-81232015214.10292015
Zorzanelli RT, Giordani F, Guaraldo L et al (2019) Comsumption of the benzodiazepine clonazepam (Rivotril®) in Rio de Janeiro State, Brazil, 2009–2013: an ecological study. Ciênc Saúde Coletiva 24(8):3129–3140. https://doi.org/10.1590/1413-81232018248.23232017
Verrotti A, Milioni M, Zaccara G (2015) Safety and efficacy of diazepam autoinjector for the management of epilepsy. Expert Rev Neurother 2:127–133. https://doi.org/10.1586/14737175.2015.1003043. (PMID: 25614951)
Babu M, Pitchumani K, Ramesh P (2014) Synthesis of 5-benzyl-4-aryl-octahydro-1H-benzo[b][1,5]diazepin-2-ones as potent antidepressant and antimicrobial agentes. Med Chem Res 23(4):2070–2079. https://doi.org/10.1007/s00044-013-0694-1
Wang LZ, Li XQ, An YS (2015) 1,5-Benzodiazepine derivatives as potential antimicrobial agents: design, synthesis, biological evaluation, and structure-activity relationships. Org Biomol Chem 13(19):5497–5509. https://doi.org/10.1039/c5ob00655d
Hamed O, Fares O, Taleeb S et al (2020) New insights towards 1,4-benzodiazepines from curcumin design. Synth Antimicrob Act Med Chem 16(8):1112–1123. https://doi.org/10.2174/1573406415666190826160251
Johnston GAR (1996) GABA a receptor pharmacology. Pharmacol Ther 69(3):173–198
Tanaka E (1999) Clinically significant pharmacokinetic drug interactions with benzodiazepines. J Clin Pharm Ther 24(5):347–355. https://doi.org/10.1046/j.1365-2710.1999.00247.x
CLSI. Performance Standards for Antimicrobial Susceptibility Testing. (2016) 26th ed. CLSI supplement M100S. Wayne, PA: Clinical and Laboratory Standards Institute.
National Committee for Clinical Laboratory Standards (NCCLS) (1999) Methods for Determining Bactericidal Activity of antimicrobial agents; Aproved Guideline, NCCLS document M26-A. National Committee for Clinical Laboratory Standards.
Lorian V. (1986) Antibiotics in Laboratory Medicine. 1º ed. Williams & Wilkins, London. https://www.dilivros.com.br/livro-antibiotics-in-laboratory-medicine9781451176759,ams002.html. Access May 20, 2021.
Konaté K, Mavoungou JF, Lepengué AN et al (2012) Antibacterial activity against β- lactamase producing Methicillin and Ampicillin resistants Staphylococcus aureus: fractional inhibitory concentration index (FICI) determination. Ann Clin Microbiol Antimicrob 11:18. https://doi.org/10.1186/1476-0711-11-18. (PMID: 22716026)
Odds FC (2003) Synergy, antagonism, and what the chequerboard puts between them. J Antimicrob Chemother 52(1):1. https://doi.org/10.1093/jac/dkg301. (PMID: 12805255)
ANVISA. (2017). Management of Surveillance and Monitoring in Health Services - GVIMS and General Management of Technology in Health Services - GGTES. National Health Surveillance Agency.
ANVISA. (2021). TECHNICAL NOTE GVIMS/GGTES/ANVISA No 06/2021 Implementation of the Antimicrobial Use Management Program (PGA) by hospitals. National Health Surveillance Agency.
Zheng W, Sun W, Simeonov A (2018) Drug repurposing screens and synergistic drug combinations for infectious diseases. Br J Pharmacol 2:181–191. https://doi.org/10.1111/bph.13895. (PMID: 28685814)
Hirschtritt ME, Olfson M, Kroenke K (2021) Balancing the risks and benefits of benzodiazepines. JAMA 325(4):347–348. https://doi.org/10.1001/jama.2020.22106. (PMID: 33416846)
Ibezim A, Onah E, Dim EM et al (2021) A computational multi-targeting approach for drug repositioning for psoriasis treatment. BMC Complement Med Ther 21:193. https://doi.org/10.1186/s12906-021-03359-2. (PMID: 34225727)
Rodrigues D, Baldissera GS, Mathos D et al (2021) Amikacin for the treatment of carbapenem-resistant Klebsiella pneumoniae infections: clinical efficacy and toxicity. Braz J Microbiol 52(4):1913–1919. https://doi.org/10.1007/s42770-02100551-x. (PMID: 34191252)
Acknowledgements
We thank Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES) [Finance Code 001] for financial support.
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
The conception and design of the study: TFR, MBS, VSF, LNF, BRP, LF, LC and R.H; Acquisition of data: TFR, MBS, VSF, LNF, BRP, LF, LC and RH; Analysis and interpretation of the data: TFR, MBS, VSF, LNF, BRP, LF, LC and RH; Drafting and critical revision of the manuscript: TFR, MBS, VSF, LNF, BRP, LF, LC and RH; Final approval of the version to be submitted: TFR, MBS, VSF, LNF, BRP, LF, LC and RH.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical Approval
The research was approved by the Research Ethics Committee of the Federal University of Santa Maria, being registered under the number of the CAAE Ethical Appreciation Certificate 928.497. Registration in the National System of Genetic Heritage Management and Associated Traditional Knowledge (SisGen) is registered under number R4B27A6—Federal University of Santa Maria.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Rosa, T.F., Serafin, M.B., Foletto, V.S. et al. Repositioning of Benzodiazepine Drugs and Synergistic Effect with Ciprofloxacin Against ESKAPE Pathogens. Curr Microbiol 80, 160 (2023). https://doi.org/10.1007/s00284-023-03242-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03242-y