Abstract
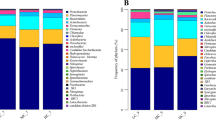
The dynamic microflora associated within, and in the surrounding aquatic environment, has been found to be responsible for the functional properties of many aquatic plants. The aim of the current work was to evaluate the effectiveness of Lemnaceae-based wastewater treatment system under tropical conditions and investigate the changes in the aquatic microflora upon plant growth. A biological wastewater treatment system was designed and investigated using mixed Lemnaceae culture comprising Lemna minor and Spirodela polyrhiza in a batch mode. A significant reduction in total solids (31.8%), biochemical oxygen demand (93.5%), and chemical oxygen demand (73.2%) was observed after seven days of duckweed growth using a low inoculum. A preliminary study on the change in the microbial population diversity and functionality, in the wastewater before and after treatment, revealed an increase in the denitrifying microflora in wastewater post-Lemnaceae treatment. Dominance of 10 bacterial phyla, contributing for 98.3% of the total bacterial communities, was recorded, and ~ 50.6% loss of diversity post-treatment of wastewater was revealed by the Shannon Index. Among 16 bacterial families showing relative abundance of ≥ 1% in untreated wastewater, Methylobacteriaceae, Pseudomonadaceae, Brucellaceae, Rhodobacteraceae, and Acetobacteraceae prevailed in the water post-treatment by duckweeds. This is a novel work done on the dynamics of aquatic microflora associated with Lemnaceae under tropical Indian conditions. It confirms the application of Lemnaceae-based wastewater treatment system as effective biofilter and calls for further studies on the active involvement of the endophytic and aquatic microflora in the functions of these plant.




Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
Code Availability
Not applicable.
References
Ziegler P, Sree KS, Appenroth KJ (2016) Duckweeds for water remediation and toxicity testing. Toxicol Environ Chem 98(10):1127–1154. https://doi.org/10.1080/02772248.2015.1094701
Appenroth KJ, Sree KS, Böhm V, Hammann S, Vetter W, Leiterer M, Jahreis G (2017) Nutritional value of duckweeds (Lemnaceae) as human food. Food Chem 217:266–273. https://doi.org/10.1016/j.foodchem.2016.08.116
Rale V, Singh P, Shetty K (2019) Lemnaceae: source food supplements to functional foods. In: Shetty K, Sarkar D (eds) Functional foods and biotechnology. CRC Press, USA, pp 133–141
US EPA. United States Environmental Protection Agency. 2002. Wastewater technology fact sheet. The living machine. Office of Water. EPA 832-F-02–025. https://www3.epa.gov/npdes/pubs/living_machine.pdf. Assessed 3 Aug 2021
Singh P, Nilisha S, Patil Y (2021) Biomanagement of chromium: current trends and promising future strategies. J Environ Manag 279:111547. https://doi.org/10.1016/j.jenvman.2020.111547
Amare E, Kebede F, Mulat W (2018) Wastewater treatment by Lemna minor and Azolla filiculoides in tropical semi-arid regions of Ethiopia. Ecol Eng 120:464–473. https://doi.org/10.1016/j.ecoleng.2018.07.005
O’Brien AM, Laurich J, Lash E, Frederickson ME (2020) Mutualistic outcomes across plant populations, microbes, and environments in the duckweed Lemna minor. Microbiol Ecol 80:384–397. https://doi.org/10.1007/s00248-019-01452-1
Tanaka Y, Tamaki H, Tanaka K, Tozawa E, Matsuzawa H, Toyama T, Mori K (2018) Duckweed-microbe co-cultivation method for isolating a wide variety of microbes including taxonomically novel microbes. Microbes Environ 33(4):402–406. https://doi.org/10.1264/jsme2.ME18067
Matsuzawa H, Tanaka Y, Tamaki H, Kamagata Y, Mori K (2010) Culture-dependent and independent analyses of the microbial communities inhabiting the giant duckweed (Spirodela polyrrhiza) rhizoplane and isolation of a variety of rarely cultivated organisms within the phylum Verrucomicrobia. Microbes Environ 25:302–308. https://doi.org/10.1264/jsme2.ME10144
Lu X, Kruatrachue M, Pokethitiyook P, Homyok K (2004) Removal of cadmium and zinc by water hyacinth, Eichhornia crassipes. Sci Asia 30:93–103
Holt JG, Krieg NR, Sneath PHA, Staley JT, Williams ST (1994) Bergey’s manual of determinative bacteriology, 9th edn. Williams & Wilkins, Baltimore
Jani K, Dhotre D, Bandal J, Shouche Y, Suryavanshi M, Rale V, Sharma A (2018) World’s largest mass bathing event influences the bacterial communities of Godavari, a holy river of India. Microbial Ecol 76(3):706–718. https://doi.org/10.1007/s00248-018-1169-1
Sharma A, Jani K, Thite V, Dhar SK, Shouche Y (2019) Geochemistry shapes bacterial communities and their metabolic potentials in tertiary coalbed. Geomicrobiol J 36(2):179–187. https://doi.org/10.1080/01490451.2018.1526987
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Lozupone CA, Turnbaugh PJ, Fierer N, Knight R (2011) Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc Nat Acad Sci USA 108(Suppl 1):4516–4522. https://doi.org/10.1073/pnas.1000080107
Jani K, Ghattargi V, Pawar S, Inamdar M, Shouche Y, Sharma A (2018) Anthropogenic activities induce depletion in microbial communities at urban sites of the river Ganges. Current Microbiol 75(1):79–83. https://doi.org/10.1007/s00284-017-1352-5
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336. https://doi.org/10.1038/nmeth.f.303
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26(19):2460–2461. https://doi.org/10.1093/bioinformatics/btq461
Douglas GM, Maffei VJ, Zaneveld J, Yurgel SN, Brown JR, Taylor CM, Huttenhower C, Langille MG (2020) PICRUSt2 for prediction of metagenome functions. Nature Biotechnol 38:1–5. https://doi.org/10.1038/s41587-020-0548-6
Peter H, Sommaruga R (2016) Shifts in diversity and function of lake bacterial communities upon glacier retreat. ISME J 10:1545–1554. https://doi.org/10.1038/ismej.2015.245
Chen G, Fang Y, Huang J, Zhao Y, Li Q, Lai F, Xu Y, Tian X, He K, Jin Y, Tan L, Zhao X (2018) Duckweed systems for eutrophic water purification through converting wastewater nutrients to high-starch biomass: comparative evaluation of three different genera (Spirodela polyrhiza, Lemna minor and Landoltia punctata) in monoculture or polyculture. RSC Adv 8(32):17927–17937. https://doi.org/10.1039/C8RA01856A
Xiao Y, Fang Y, Jin YL, Zhang GH, Zhao H (2013) Culturing duckweed in the field for starch accumulation. Indust Crops Prod 48:183–190. https://doi.org/10.1016/j.indcrop.2013.04.017
Korner S, Vermaat JE (1998) The relative importance of Lemna gibba L., bacteria and algae for nitrogen and phosphorous removal in duckweed-covered domestic wastewater. Water Res 33(12):3651–3661. https://doi.org/10.1016/S0043-1354(98)00166-3
Selvarani AJ, Padmavathy P, Srinivasan A, Jawahar P (2015) Performance of duckweed (Lemna minor) on different types of wastewater treatment. Internat J Fishery Aq Stud 2:208–212
Shafai SA, El-Gohary FA, Nasr FA, Van Der Steen NP, Gijzen HJ (2007) Nutrient recovery from domestic wastewater using a UASB-duckweed ponds system. Biores Technol 98(4):798–807. https://doi.org/10.1016/j.biortech.2006.03.011
Nasr FA, Doma HS, Nassar HF (2009) Treatment of domestic wastewater using an anaerobic baffled reactor followed by a duckweed pond for agricultural purposes. Environmentalist 29(3):270–279. https://doi.org/10.1007/s10669-008-9188-y
McLay CL (1976) The effect of pH on the population growth of three species of duckweed: Spirodela oligorrhiza, Lemna minor and Wolffia arrhiza. Freshwater Biol 6(2):125–136. https://doi.org/10.1111/j.1365-2427.1976.tb01596.x
Oron G (1994) Duckweed culture for wastewater renovation and biomass production. Agricul Water Manag 26(1–2):27–40. https://doi.org/10.1016/0378-3774(94)90022-1
Ardiansyah A, Fotedar R (2016) The abundance and diversity of heterotrophic bacteria as a function of harvesting frequency of duckweed (Lemna minor L.) in recirculating aquaculture systems. Lett Appl Microbiol 63(1):53–59. https://doi.org/10.1111/lam.12585
Oron G, Porath D, Jansen H (1987) Performance of duckweed species Lemna gibba on municipal wastewater for effluent renovation and protein production. Biotechnol Bioeng 29:258–268. https://doi.org/10.1002/bit.260290217
Mandi L (1994) Marrakesh wastewater purification experiment using vascular aquatic plants Eichhornia crassipes and Lemna gibba. Water Sci Technol 29:283–287. https://doi.org/10.2166/wst.1994.0210
Van der Steen P, Brenner A, Oron G (1998) An integrated duckweed and algae pond system for nitrogen removal and renovation. Water Sci Technol 38(1):335. https://doi.org/10.1016/S0273-1223(98)00419-3
Zimmo OR, Al-Sa’ed RM, Van der Steen NP, Gijzen HJ (2002) Process performance assessment of algae-based and duckweed-based wastewater treatment systems. Water Sci Technol 45(1):91–101. https://doi.org/10.2166/wst.2002.0013
Azeez NM, Sabbar AA (2012) Efficiency of duckweed (Lemna minor L.) in phyto-treatment of wastewater pollutants from Basrah oil refinery. J Appl Phytotechnol Environ Sanita 1(4):163–172
Daud MK, Ali S, Abbas Z, Zaheer IE, Riaz MA, Malik A, Zhu SJ (2018) Potential of duckweed (Lemna minor) for the phytoremediation of landfill leachate. J Chem. https://doi.org/10.1155/2018/3951540
Priya A, Avishek K, Pathak G (2012) Assessing the potentials of Lemna minor in the treatment of domestic wastewater at pilot scale. Environ Mon Assess 184(7):4301–4307. https://doi.org/10.1007/s10661-011-2265-6
Chen G, Huang J, Fang Y, Zhao Y, Tian X, Jin Y, Zhao H (2019) Microbial community succession and pollutants removal of a novel carriers enhanced duckweed treatment system for rural wastewater in Dianchi Lake basin. Biores Technol 276:8–17. https://doi.org/10.1016/j.biortech.2018.12.102
Steen NP, Ferrer AVM, Samarasinghe KG, Gijzen HJ (2003) Quantification and comparison of methane emissions from algae and duckweed based wastewater treatment ponds. In: Memorias del evento: Agua 2003, Universidad del Valle, CINARA, International Water Association, Cartagena de Indias, pp 1–7, Ilus
Msaddak A, Rejili M, Durán D, Rey L, Imperial J, Palacios JM, Mars M (2017) Members of Microvirga and Bradyrhizobium genera are native endosymbiotic bacteria nodulating Lupinus luteus in Northern Tunisian soils. FEMS Microbiol Ecol. https://doi.org/10.1093/femsec/fix068
Kundu P, Pramanik A, Dasgupta A, Mukherjee J, Mukherjee S (2014) Simultaneous heterotrophic nitrification and aerobic denitrification by Chryseobacterium sp. R31 isolated from abattoir wastewater. BioMed Res Internat 2014:1–12. https://doi.org/10.1155/2014/436056
Kasana RC, Pandey CB (2018) Exiguobacterium: an overview of a versatile genus with potential in industry and agriculture. Crit Rev Biotechnol 38(1):141–156. https://doi.org/10.1080/07388551.2017.1312273
Lalucat J, Bennasar A, Bosch R, Garcıa-Valdes E, Palleroni NJ (2006) Biology of Pseudomonas stutzeri. Microbiol Mol Biol Rev 70:510–547. https://doi.org/10.1128/MMBR.00047-05
Stout LM, Dodova EN, Tyson JF, Nüsslein K (2010) Phytoprotective influence of bacteria on growth and cadmium accumulation in the aquatic plant Lemna minor. Water Res 44(17):4970–4979. https://doi.org/10.1016/j.watres.2010.07.073
Toyama T, Kuroda M, Ogata Y, Hachiya Y, Quach A, Tokura K, Ike M (2017) Enhanced biomass production of duckweeds by inoculating a plant growth-promoting bacterium, Acinetobacter calcoaceticus P23, in sterile medium and non-sterile environmental waters. Water Sci Technol 76(6):1418–1428. https://doi.org/10.2166/wst.2017.296
Ishizawa H, Kuroda M, Morikawa M, Ike M (2017) Evaluation of environmental bacterial communities as a factor affecting the growth of duckweed Lemna minor. Biotechnol Biofuels 10:1–10. https://doi.org/10.1186/s13068-017-0746-8
Acknowledgements
The authors wish to acknowledge Ms. Pooja Tendulkar from Lemnion Green Solutions Pvt. Ltd., Pune, for supplying the duckweed cultures. We also wish to acknowledge Symbiosis School of Biological Sciences and NCCS, Pune for sharing infrastructure for the collaborative research. The authors are also thankful to Symbiosis International (Deemed University) for providing every support for research and academics.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Final data compilation, result analysis, and the first draft of the manuscript were prepared by PS. Material preparation, data collection, and analysis were performed by KJ, SS, and SP. VR, YS, and PJ helped with the resources and research infrastructure availability. YP and MD were involved in overall supervision and investigation of the work. All authors contributed to the study conception and design. All authors were involved in review and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical Approval
Not Applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not Applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, P., Jani, K., Sharma, S. et al. Microbial Population Dynamics in Lemnaceae (Duckweed)-Based Wastewater Treatment System. Curr Microbiol 80, 56 (2023). https://doi.org/10.1007/s00284-022-03149-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-022-03149-0