Abstract
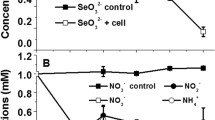
A silver nanoparticle (AgNP) producing, Gram stain-positive, aerobic, motile, and rod-shaped novel bacterial strain, designated as MAH-34 T was isolated from rhizospheric soil of magnolia tree. The colonies were creamy white, smooth, circular, and 0.9–2.0 mm in diameter when grown on R2A agar. Strain MAH-34 T was found to be able to grow at 10–37 °C, at pH 6.0–9.5, and at 0–1% NaCl. The strain showed activity for both catalase, and oxidase tests, and was able to rapid synthesis of AgNPs. The TEM image revealed the spherical shape of biosynthesized AgNPs, and the size was 5 to 15 nm. Based on 16S rRNA gene sequence comparisons, the isolate was shown to be a member of genus Paenibacillus, and the close type strains were Paenibacillus chondroitinus DSM 5051 T (98.3%), Paenibacillus aceris KUDC4121T (98.2%), Paenibacillus nebraskensis JJ-59 T (97.8%), Paenibacillus alginolyticus DSM 5050 T (97.6%), Paenibacillus ferrarius CY1T (97.4%), Paenibacillus frigoriresistens YIM 016 T (97.3%), and Paenibacillus pocheonensis Gsoil 1138 T (97.3%). Strain MAH-34 T had a genome size of 8,647,010 bp. The genomic G + C content was 46.0 mol %. The major isoprenoid quinone was determined as menaquinone-7 (MK-7). The major cellular fatty acids were determined as C15:0 anteiso, and C16:0 iso. Based on the DNA-DNA hybridization results, genotypic analysis, chemotaxonomic, and physiological data, strain MAH-34 T represents a novel species, for which the name Paenibacillus anseongense sp. nov. is proposed, with MAH-34 T as the type strain (= KACC 19974 T = CGMCC1.16610 T).


Similar content being viewed by others
References
Ash C, Priest FG, Collins MD (1993) Molecular identification of rRNA group 3 bacilli (Ash, Farrow, Wallbanks and Collins) using a PCR probe test. Proposal for the creation of a new genus Paenibacillus. Antonie Van Leeuwenhoek 64:253–260
Yao R, Wang R, Wang D, Su J, Zheng SX, Wang G (2014) Paenibacillus selenitireducens sp. nov., a selenite-reducing bacterium isolated from a selenium mineral soil. Int J Syst Evol Microbiol 64:805–811
Akter S, Huq MA (2018) Biological synthesis of ginsenoside Rd using Paenibacillus horti sp. nov. isolated from vegetable garden. Curr Microbiol 75:1566–1573
Hwang YJ, Ghim SY (2017) Paenibacillus aceris sp. nov., isolated from the rhizosphere of Acer okamotoanum, a plant native to Ulleungdo Island, Republic of Korea. Int J Syst Evol Microbiol 67:1039–1045
Kämpfer P, Busse HJ, McInroy JA, Hu CH, Kloepper JW, Glaeser SP (2017) Paenibacillus nebraskensis sp. nov., isolated from the root surface of field-grown maize. Int J Syst Evol Microbiol 67:4956–4961
Mel-A F, Kim YJ, Van An H, Sukweenadhi J, Singh P, Huq MA, Yang DC (2015) Burkholderia ginsengiterrae sp. nov. and Burkholderia panaciterrae sp. nov., antagonistic bacteria against root rot pathogen Cylindrocarpon destructans, isolated from ginseng soil. Arch Microbiol 197:439–447
Huq MA, Kim YJ, Min JW, Yang DC (2014) Use of Lactobacillus rossiae DC05 for bioconversion of the major ginsenosides Rb1 and Re into the pharmacologically active ginsenosides C-K and Rg2. Food Sci Biotechnol 23:1561–1567
Majdalawieh A, Kanan MC, El-Kadri O (2014) Recent advances in gold and silver nanoparticles: synthesis and applications. J Nanosci Nanotechnol 14:4757–4780
Singh P, Kim YJ, Singh H (2015) Biosynthesis, characterization, and antimicrobial applications of silver nanoparticles. Int J Nanomed 10:2567–2577
Fautz E, Reichenbach H (1980) A simple test for flexirubin-type pigments. FEMS Microbiol Lett 8:87–91
Huq MA (2017) Chryseobacterium chungangensis sp. nov., a bacterium isolated from soil of sweet gourd garden. Arch Microbiol. https://doi.org/10.1007/s00203-017-1469-8
Skerman VBD (1967) A Guide to the Identification of the Genera of Bacteria, 2nd edn. Williams and Wilkins, Baltimore
Christensen WB (1946) Urea decomposition as a means of differentiating proteus and paracolon cultures from each other and from salmonella and shigella types. J Bacteriol 52:461–466
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Kim OS, Cho YJ, Lee K, Yoon SH, Kim M, Na H, Park SC, Jeon YS, Lee JH, Yi H, Won S, Chun J (2012) Introducing EzTaxon-e: a prokaryotic 16S rRNA Gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser 41:95–98
Kimura M (1983) The Neutral Theory of Molecular Evolution. Cambridge University Press, Cambridge
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Bio Evol 4:406–425
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. MolBiolEvol 28:2731–2739
Felsenstein J (1985) Confidence limit on phylogenies: an approach using the bootstrap. Evolution / Evolution: Int J Org Evol 39:783–791
Yoon SH, Ha SM, Lim JM, Kwon SJ, Chun J (2017) A large-scale evaluation of algorithms to calculate average nucleotide identity. Antonie Van Leeuwenhoek 110:1281–1286
Ezaki T, Hashimoto Y, Yabuuchi E (1989) Fluorometric deoxyribonucleic acid-deoxyribonucleic acid hybridization in microdilution wells as an alternative to membrane filter hybridization in which radioisotopes are used to determine genetic relatedness among bacterial strains. Int J Syst Bacteriol 39:224–229
Stabili L, Gravili C, Tredici SM, Piraino S, Talà A, Boero F, Alifano P (2008) Epibiotic Vibrio luminous bacteria isolated from some hydrozoa and bryozoa species. Microb Ecol 56:625–636
Gillis M, De Ley J, De Cleene M (1970) The determination of molecular weight of bacterial genome DNA from renaturation rates. Eur J Biochem 12:143–153
McConaughy BL, Laird CD, McCarthy BJ (1969) Nucleic acid reassociation in formamide. Biochemistry 8:3289–3295
Sasser M (1990) Identification of Bacteria by Gas Chromatography of Cellular Fatty Acids. MIDI Technical Note 101. Newark, DE: MIDI Inc.
Collins MD (1985) Isoprenoid quinone analyses in bacterial classification and identification. In: Goodfellow M, Minnikin DE (eds) Chemical Methods in Bacterial Systematics. Academic Press, London, pp 267–287
Du J, Sing H, Yi TH (2017) Biosynthesis of silver nanoparticles by Novosphingobium sp. THG-C3 and their antimicrobial potential. Artif Cells Nanomed Biotechnol 45:211–217
Huq MA, Kim YJ, Hoang VA, Siddiqi MZ, Yang DC (2015) Paenibacillus ginsengiterrae sp. nov., a ginsenoside-hydrolyzing bacteria isolated from soil of ginseng field. Arch Microbiol 197:389–396
Stackebrandt E, Goebel BM (1994) Taxonomic note: A place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int J Syst Bacteriol 44:846–849
Huq MA (2018) Microvirga rosea sp. nov.: a nanoparticle producing bacterium isolated from soil of rose garden. Arch Microbiol 200:1439–1445
Huq MA (2020) Green synthesis of silver nanoparticles using Pseudoduganella eburnea MAHUQ-39 and their antimicrobial mechanisms investigation against drug resistant human pathogens. Int J Mol Sci 21(4):1510
Shida O, Takagi H, Kadowaki K, Nakamura LK, Komagata K (1997) Transfer of Bacillus alginolyticus, Bacillus chondroitinus, Bacillus curdlanolyticus, Bacillus glucanolyticus, Bacillus kobensis, and Bacillus thiaminolyticus to the genus Paenibacillus and emended description of the genus Paenibacillus. Int J Syst Bacteriol 47:289–298
Cao Y, Chen F, Li Y, Wei S, Wang G (2015) Paenibacillus ferrarius sp. nov., isolated from iron mineral soil. Int J Syst Evol Microbiol 65:165–170
Ming H, Nie GX, Jiang HC, Yu TT, Zhou EM, Feng HG, Tang SK, Li WJ (2012) Paenibacillus frigoriresistens sp. nov., a novel psychrotroph isolated from a peat bog in Heilongjiang. Northern China Antonie Van Leeuwenhoek 102:297–305
Baek SH, Yi TH, Lee ST, Im WT (2010) Paenibacillus pocheonensis sp. nov., a facultative anaerobe isolated from soil of a ginseng field. Int J Syst Evol Microbiol 60:1163–1167
Acknowledgements
This study was performed with the support of the National Research Foundation (NRF) of Korea Grant (Project No. NRF-2018R1C1B5041386, Recipient: Md. Amdadul Huq) funded by Korean Government, Republic of Korea. Special thanks to CGM 10 K project for analyzing the draft genome sequence of strain MAH-34 T (GCM60011526).
Author information
Authors and Affiliations
Contributions
Md. Amdadul Huq conceived the original screening and research plans, performed all of the experiments and wrote the article.
Corresponding author
Ethics declarations
Conflict of interest
The author declares that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
284_2020_2086_MOESM1_ESM.pdf
Supplementary Fig. S1. Transmission electron micrograph of strain MAH-34T after negative staining with uranyl acetate, Bar, 1.0 μm. (PDF 370 kb)
Rights and permissions
About this article
Cite this article
Huq, M.A. Paenibacillus anseongense sp. nov. a Silver Nanoparticle Producing Bacterium Isolated from Rhizospheric Soil. Curr Microbiol 77, 2023–2030 (2020). https://doi.org/10.1007/s00284-020-02086-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-020-02086-0