Abstract
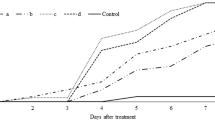
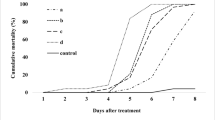
Entomopathogenic fungi often have wide host range and can be important biological control agents against the oriental fruit moth, Grapholita molesta (Busck) (Lepidoptera: Tortricidae), a significant pest of stone and pome fruits. The virulence of six entomopathogenic fungi, including three strains of Beauveria bassiana (Balsamo) Vuillemin (Hypocreales: Cordycipitaceae) (ARP14, GHA, A) and one strain each of Metarhizium robertsii Bischoff, Rehner, & Humber (Hypocreales: Clavicipitaceae), Metarhizium brunneum (Petch) (Hypocreales: Clavicipitaceae), and Isaria farinosa (Holmsk.) (Hypocreales: Cordycipitaceae) was assessed against first instars (< 5 h old) of G. molesta held in glass scintillation vials. Compared to commercialized strain of B. bassiana (GHA), B. bassiana ARP14, which was recovered from a cadaver of Riptortus pedestris (F.) (Hemiptera: Alydidae), killed first instar larvae 2.6 times faster at 12 h after exposure to 1 × 108 conidia/ml concentration at 95.9% RH and 25.4 °C. However, the mycosis rate after 14 days was similar to that of B. bassiana GHA in all treatments. Beauveria bassiana ARP14 also killed adult moths 2.7 (males) and 2.2 (females) times faster than did B. bassiana GHA, measured 168 h (7 days) after exposure. However, the mycosis rate after 14 days was similar from both fungi. In assay using three conidial concentrations (1 × 107, 1 × 108, and 1 × 109 conidia/ml) and three RHs levels (55, 75, and 95%). Mortality became higher as concentration and RH increase, but mycosis rate at 14 days wasn’t significantly different among treatments. These results suggest that B. bassiana ARP14 can be a potential biological control agent against G. molesta.






Similar content being viewed by others
References
Najar-Rodriguez A, Bellutti N, Dorn S (2013) Larval performance of the oriental fruit moth across fruits from primary and secondary hosts. Physiol Entomol 38:63–70
Rothschild GHL, Vickers RA (1991) Biology, ecology and control of the oriental fruit moth. In: van der Geest LPS, Evenhuis HH (eds) Tortricid pests: their biology. Natural Enemies and Control. Elsevier Publishers, New York, pp 389–412
Mazzi D, Dorn S (2012) Movement of insect pests in agricultural landscapes. Ann Appl Biol 160:97–113
Rice RE, Doyle J, Jones RA (1972) Pear as a host of the oriental fruit moth in California. J Econ Entomol 65:1212–1213
Zhao ZR, Wang YG, Yan GY (1989) A preliminary report on the oriental fruit moth in north Jiangsu. Insect Knowl 26:17–19
Kanga LHB, Pree DJ, van Lier JL, Walker GM (2003) Management of insecticide resistance in Oriental fruit moth (Grapholita molesta; Lepidoptera: Tortricidae) populations from Ontario. Pest Manag Sci 59:921–927
Yang CY, Han KS, Boo KS (2001) Occurrence of and damage by the oriental fruit moth, Grapholita molesta (Busck) (Lepidoptera: Tortricidae) in pear orchards. Korean J Appl Entomol 40:117–123
Pree DJ, Whitty KJ, Van Driel L, Walker GM (1998) Resistance to insecticides in Oriental fruit moth populations (Grapholita molesta) from the Niagara peninsula of Ontario. Can Entomol 130:245–256
Pree DJ, Whitty KJ, Pogoda MK, Bittner LA (2005) Status of resistance to insecticides in populations of the oriental fruit moth Grapholita molesta (Busck) (Lepidoptera: Tortricidae) in Southern Ontario. J Ent Soc Ont 136:53–70
Hung D-Z, Yang H-J, Li Y-F, Lin C-L, Chang S-Y, Sung F-C et al (2015) The long-term effects of organophosphates poisoning as a risk factor of CVDs: a nationwide population based cohort study. PLoS ONE 10(9):e0137632
Bing LA, Lewis LC (1991) Suppression of Ostrinia nubilalis (Hübner) (Lepidoptera: Pyralidae) by endophytic Beauveria bassiana (Balsamo) Vuillemin. Environ Entomol 20:1207–1211
Vandenberg JD, Shelton AM, Wilsey WT, Ramos M (1998) Assessment of Beauveria bassiana sprays for control of diamondback moth (Lepidoptera: Plutellidae) on crucifers. J Econ Entomol 91:624–630
Nguyen NTH, Borgemeister C, Poehling H, Zimmermann G (2007) Laboratory investigation on the potential of entomopathogenic fungi for biocontrol of Helicoverpa armigera (Lepidoptera: Noctuidae) larvae and pupae. Biocontrol Sci Technol 17:853–864
Duarte RT, Gonçalves KC, Espinosa DJJ, Moreira LF, De Bortoli SA, Humber RA, Polanczyk RA (2016) Potential of entomopathogenic fungi as biological control agents of diamondback moth (Lepidoptera: Plutellidae) and compatibility with chemical insecticides. J Econ Entomol 109:594–601
Wraight SP, Ramos ME, Avery PB, Jaronski ST, Vandenberg JD (2010) Comparative virulence of Beauveria bassiana isolates against lepidopteran pests of vegetable crops. J Invertebr Pathol 103:186–199
Leatherdale D (1970) The arthropod hosts of entomogenous fungi in Britain. Entomophaga 15:419–435
Cossentine JE, Judd GJR, Bissett JD, Lacey LA (2010) Susceptibility of apple clearwing moth larvae, Synanthedon myopaeformis (Lepidoptera: Sesiidae) to Beauveria bassiana and Metarhizium brunneum. Biocontrol Sci Technol 20:703–707
Dangi N, Lim UT (2018) Identification and evaluation of a new entomopathogenic fungal strain against Riptortus pedestris (Hemiptera: Alydidae) and its two parasitoids. PLoS ONE 13:e0195848
Du J, Li G, Xu X, Wu J (2015) Development and fecundity performance of oriental fruit moth (Lepidoptera: Tortricidae) reared on shoots and fruits of peach and pear in different seasons. Environ Entomol 44:1522–1530
Winston PW, Bates DH (1960) Saturated solutions for the control of humidity in biological research. Ecology 41:232–237
Abbott WS (1925) A method of computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Institute SAS (2012) SAS user’s guide: statistics. SAS Institute, Cary
Zar JH (2010) Biostatistical analysis, 5th edn. Prentice Hall, Upper Saddle River
Sosa- Gómez DRS, Boucias DG, Nation JL (1997) Attachment of Metarhizium anisoplise to the southern green stink bug Nezara viridula cuticle and fungistatic effect of cuticular lipids and aldehydes. J Invertebr Pathol 69:31–39
Sosa- Gómez DRS, Moscardi F (1998) Laboratory and field studies on the infection of stink bugs, Nezara viridula, Piezodorus guildinii, and Euschistus heros with Metarhizium anisoplise and Beauveria bassiana in Brazil. J Invertebr Pathol 71:115–120
Faria M, Wraight SP (2001) Biological control of Bemisia tabaci with fungi. Crop Prot 20:767–778
Feng MG, Chen B, Ying SH (2004) Trails of Beauveria bassiana, Paecilomyces fumosoroseus and imidacloprid for management of Trialeurodes vaporariorum (Homoptera: Aleyrodidae) on greenhouse grown lettuce. Biocontrol Sci Technol 14:513–544
Hatting JL, Wraight SP, Miller RM (2004) Efficacy of Beauveria bassiana (Hyphomycetes) for control of Russian wheat aphid (Homoptera: Aphididae) on resistant wheat under field conditions. Biocontrol Sci Technol 14:459–473
Pu XY, Feng MG, Shi CH (2005) Impact of three application methods on the field efficacy of a Beauveria bassiana-based mycoinsecticide against the false-eye leafhopper, Empoasca vitis (Homoptera: Cicadellidae) in tea canopy. Crop Prot 24:167–175
Sasaki F, Miyamoto T, Yamamoto A, Tamai Y, Yajima T (2012) Relationship between intraspecific variations and host insects of Ophiocordyceps nutans collected in Japan. Mycoscience 53:85–91
Kučera M, Samšiňáková A (1968) Toxins of the entomophagous fungus Beauveria bassiuna. J Invertebr Pathol 12:316–320
James RR, Buckner JS, Freeman TP (2003) Cuticular lipids and silver leaf whitefly stage affect conidial germination of Beauveria bassiana and Paecilomyces fumosoroseus. J Invertebr Pathol 84:67–74
Benz G (1987) Environment. In: Fuxa JR, Tanada Y (eds) Epizootiology of insect diseases. Wiley, NY, pp 177–214
Inglis DG, Goettel SM, Butt MT, Strasser H (2001) Use of hyphomycetes fungi for managing insect pests. In: Butt TM, Jackson C, Magan N (eds) Fungi as biocontrol agents. CABI Publishing, Wallingford, pp 23–27
Alves SB, Tamai MA, Rossi LS, Castiglioni E (2005) Beauveria bassiana pathogenicity to the citrus rust mite Phyllocoptruta olievora. Exp Appl Acarol 37:117–122
Seiedy M, Saboori A, Allahyari H, Hassanloui RT, Tork M (2010) Laboratory investigation on the virulence of two isolates of the entomopathogenic fungus Beauveria bassiana against the two spotted spider mite Tetranychus urticae (Acari: Tetranychidae). Int J Acarol 36:527–532
Ferron P, Fargues J, Riba G (1991) Fungi as microbial insecticides against pests. In: Arora DK, Ajello L, Mukerji KG (eds) Handbook of applied mycology Humans, Animals and Insects. Marcel Dekker Inc., New York, pp 665–705
Devi KU, Rao CUM (2006) Allee effect in the infection dynamics of the entomopathogenic fungus Beauveria bassiana (Bals) Vuill. on the beetle Mylabris pustulata. Mycopathologia 161:385–394
Sandhu SS, Sharma AK, Beniwal V, Goel G, Batra P, Kumar A, Jaglan S, Sharma AK, Malhotra S (2011) Myco-biocontrol of insect pests: factors involved mechanism, and regulation. J Pathol 2012:126819–126829
Hamill RL, Sullivan HR, Gorman M (1969) Determination of pyrrolnitrin and derivatives by gas-liquid chromatography. Appl Microbiol 18:310–312
Elsworth JF, Grove JF (1977) Cyclodepsipeptides from Beauveria bassiana Bals. Part 1. Beauverolides H and I. J Chem Soc Perkin Trans 13:270–273
Acknowledgements
We would like to thank Prof. Jae Su Kim at the Department of Agricultural Biology, Chonbuk National University. This work was carried out with the support of "Cooperative Research Program for Agriculture Science & Technology Development (Project No. PJ013362042020)" Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sarker, S., Woo, Y.H. & Lim, U.T. Laboratory Evaluation of Beauveria bassiana ARP14 Against Grapholita molesta (Lepidoptera: Tortricidae). Curr Microbiol 77, 2365–2373 (2020). https://doi.org/10.1007/s00284-020-02012-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-020-02012-4