Abstract
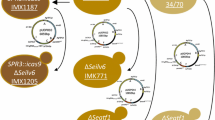
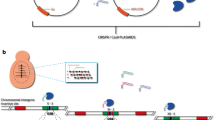
The yeast S. cerevisiae serves as a model organism for many decades. Numerous molecular tools have been developed throughout the years to engineer its genome. Specifically, homologous recombination protocols allowed gene deletion, replacement and tagging of almost every S. cerevisiae gene, thus enabling mechanistic understanding of various cellular processes. Recently, CRISPR/Cas9-based approaches have been adapted to the yeast system, simplifying the protocols to manipulate this organism. In CRISPR/Cas9 systems, guide-RNA directs a site-specific double-strand DNA cleavage by the Cas9 nuclease. The directed cleavage enhances homologous recombination events, thereby facilitating changes to desired genomic loci. The use of a single vector to express both guide-RNA and Cas9 enzyme may simplify genomic manipulations and was used to introduce double-strand breaks at artificial sites (Anand et al. in Nature 544(7650):377–380, 2017. https://doi.org/10.1038/nature22046) or within selection markers (Ryan et al. in Cold Spring Harbor Protoc, 2014. https://doi.org/10.1101/pdb.prot086827). Here, we generalize this approach to demonstrate its utility in modifying natural genomic loci. We devise vectors to perform common genetic manipulations in S. cerevisiae, including gene deletion, single-base mutations, introduction of site-specific polymorphism and tag insertion. Notably, a vector that efficiently cleaves within GFP was generated, allowing replacing a GFP tag with other sequences. This vector may be of utility for replacing any gene tagged with GFP by a sequence of choice. Importantly, we demonstrate the efficiency of chemically synthesized 80-mer homologous DNA as a substrate for recombination, alleviating the need for PCR steps in the procedure. In all presented applications, high efficiency of the expected gene alteration and no other change in the genomic loci were obtained. Overall, this work expands the repertoire of single-plasmid CRISPR/cas9 approaches and provides a facile alternative to manipulate the yeast genome.





Similar content being viewed by others
References
Anand R, Beach A, Li K, Haber J (2017) Rad51-mediated double-strand break repair and mismatch correction of divergent substrates. Nature 544(7650):377–380. https://doi.org/10.1038/nature22046
Anand R, Memisoglu G, Haber J (2017) Cas9-mediated gene editing in Saccharomyces cerevisiae. Protoc Exch. https://doi.org/10.1038/protex.2017.021a
Bailey TL, Boden M, Buske FA, Frith M, Grant CE, Clementi L, Ren J, Li WW, Noble WS (2009) MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res 37(Web Server issue):W202–W208. https://doi.org/10.1093/nar/gkp335
Barrangou R, Fremaux C, Deveau H, Richards M, Boyaval P, Moineau S, Romero DA, Horvath P (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315(5819):1709–1712. https://doi.org/10.1126/science.1138140
Botstein D, Fink GR (2011) Yeast: an experimental organism for 21st century biology. Genetics 189(3):695–704. https://doi.org/10.1534/genetics.111.130765
Chen Y, Wang Z, Ni H, Xu Y, Chen Q, Jiang L (2017) CRISPR/Cas9-mediated base-editing system efficiently generates gain-of-function mutations in Arabidopsis. Science China Life Sci 60(5):520–523. https://doi.org/10.1007/s11427-017-9021-5
Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA, Zhang F (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339(6121):819–823. https://doi.org/10.1126/science.1231143
Degreif D, Kremenovic M, Geiger T, Bertl A (2018) Preloading budding yeast with all-in-one CRISPR/Cas9 vectors for easy and high-efficient genome editing. J Biol Methods 5(3):98. 10.14440/jbm.2018.254.
DiCarlo JE, Norville JE, Mali P, Rios X, Aach J, Church GM (2013) Genome engineering in Saccharomyces cerevisiae using CRISPR-Cas systems. Nucleic Acids Res 41(7):4336–4343. https://doi.org/10.1093/nar/gkt135
Generoso WC, Gottardi M, Oreb M, Boles E (2016) Simplified CRISPR-Cas genome editing for Saccharomyces cerevisiae. J Microbiol Methods 127:203–205. https://doi.org/10.1016/J.MIMET.2016.06.020
Ghaemmaghami S, Huh W-K, Bower K, Howson RW, Belle A, Dephoure N, O'Shea EK, Weissman JS (2003) Global analysis of protein expression in yeast. Nature 425(6959):737–741. https://doi.org/10.1038/nature02046
Giaever G, Dow S, Lucau-danila A, Anderson K, Arkin AP, Astromoff A et al (2002) Functional profiling of the Saccharomyces cerevisiae genome. Nature 418:387–391. https://doi.org/10.1038/nature00935
Gietz RD, Schiestl RH, Willems AR, Woods RA (1995) Studies on the transformation of intact yeast cells by the LiAc/SS-DNA/PEG procedure. Yeast 11(4):355–360. https://doi.org/10.1002/yea.320110408
Horwitz AA, Walter JM, Schubert MG, Kung SH, Hawkins K, Platt DM et al (2015) Efficient multiplexed integration of synergistic alleles and metabolic pathways in yeasts via CRISPR-Cas. Cell Syst. https://doi.org/10.1016/j.cels.2015.02.001
Hu G, Luo S, Rao H, Cheng H, Gan X (2018) A simple PCR-based strategy for the introduction of point mutations in the yeast Saccharomyces cerevisiae via CRISPR/Cas9. Biochem Mol Biol J. https://doi.org/10.21767/2471-8084.100058
Huh W-K, Falvo JV, Gerke LC, Carroll AS, Howson RW, Weissman JS, O’Shea EK (2003) Global analysis of protein localization in budding yeast. Nature 425(6959):686–691. https://doi.org/10.1038/nature02026
Jakočinas T, Bonde I, Herrgård M, Harrison SJ, Kristensen M, Pedersen LE, Jensen MK, Keasling JD (2015) Multiplex metabolic pathway engineering using CRISPR/Cas9 in Saccharomyces cerevisiae. Metab Eng. https://doi.org/10.1016/j.ymben.2015.01.008
Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337(6096):816–821. https://doi.org/10.1126/science.1225829
Levi O, Arava Y (2019) mRNA association by aminoacyl tRNA synthetase occurs at a putative anticodon mimic and autoregulates translation in response to tRNA levels. PLoS Biol 17(5):e3000274. https://doi.org/10.1371/journal.pbio.3000274
Li J, Sun Y, Du J, Zhao Y, Xia L (2017) Generation of targeted point mutations in rice by a modified CRISPR/Cas9 system. Mol Plant 10(3):526–529. https://doi.org/10.1016/J.MOLP.2016.12.001
Mitsui R, Yamada R, Ogino H (2019) CRISPR system in the yeast Saccharomyces cerevisiae and its application in the bioproduction of useful chemicals. World J Microbiol Biotechnol 35(7):111. https://doi.org/10.1007/s11274-019-2688-8
Paquet D, Kwart D, Chen A, Sproul A, Jacob S, Teo S, Olsen KM, Gregg A, Noggle S, Tessier-Lavigne M (2016) Efficient introduction of specific homozygous and heterozygous mutations using CRISPR/Cas9. Nature 533(7601):125–129. https://doi.org/10.1038/nature17664
Reider Apel A, d’Espaux L, Wehrs M, Sachs D, Li RA, Tong GJ, Garber M, Nnadi O, Zhuang W, Hillson NJ, Keasling JD, Mukhopadhyay A (2017) A Cas9-based toolkit to program gene expression in Saccharomyces cerevisiae. Nucleic Acids Res 45(1):496–508. https://doi.org/10.1093/nar/gkw1023
Ryan OW, Poddar S, Cate JHD (2016) CRISPR–Cas9 genome engineering in Saccharomyces cerevisiae cells. Cold Spring Harbor Protoc. https://doi.org/10.1101/pdb.prot086827
Ryan OW, Skerker JM, Maurer MJ, Li X, Tsai JC, Poddar S, Lee ME, DeLoache W, Dueber JE, Arkin AP, Cate JH (2014) Selection of chromosomal DNA libraries using a multiplex CRISPR system. ELife. https://doi.org/10.7554/eLife.03703
Smith JD, Suresh S, Schlecht U, Wu M, Wagih O, Peltz G, Davis RW, Steinmetz LM, Parts L, St Onge RP (2016) Quantitative CRISPR interference screens in yeast identify chemical–genetic interactions and new rules for guide RNA design. Genome Biol 17(1):45. https://doi.org/10.1186/s13059-016-0900-9
Soreanu I, Hendler A, Dahan D, Dovrat D, Aharoni A (2018) Marker-free genetic manipulations in yeast using CRISPR/CAS9 system. Curr Genet 64(5):1129–1139. https://doi.org/10.1007/s00294-018-0831-y
Storici F, Durham CL, Gordenin DA, Resnick MA (2003) Chromosomal site-specific double-strand breaks are efficiently targeted for repair by oligonucleotides in yeast. Proc Natl Acad Sci USA 100(25):14994–14999. https://doi.org/10.1073/pnas.2036296100
Stovicek V, Borodina I, Forster J (2015) CRISPR-Cas system enables fast and simple genome editing of industrial Saccharomyces cerevisiae strains. Metab Eng Commun 2:13–22. https://doi.org/10.1016/j.meteno.2015.03.001
Toksoy Öner E (2006) Optimization of ethanol production from starch by an amylolytic nuclear petite Saccharomyces cerevisiae strain. Yeast 23(12):849–856. https://doi.org/10.1002/yea.1399
Toulmay A, Schneiter R (2006) A two-step method for the introduction of single or multiple defined point mutations into the genome of Saccharomyces cerevisiae. Yeast 23(11):825–831. https://doi.org/10.1002/yea.1397
Vyas VK, Bushkin GG, Bernstein DA, Getz MA, Sewastianik M, Barrasa MI, Bartel DP, Fink GR (2018) New CRISPR mutagenesis strategies reveal variation in repair mechanisms among fungi. mSphere. https://doi.org/10.1128/mSphere.00154-18
Wendland J (2003) PCR-based methods facilitate targeted gene manipulations and cloning procedures. Curr Genet 44(3):115–123. https://doi.org/10.1007/s00294-003-0436-x
Zhang Y, Wang J, Wang Z, Zhang Y, Shi S, Nielsen J, Liu Z (2019) A gRNA-tRNA array for CRISPR-Cas9 based rapid multiplexed genome editing in Saccharomyces cerevisiae. Nat Commun 10(1):1053. https://doi.org/10.1038/s41467-019-09005-3
Acknowledgements
We thank Prof. Schraga Shcwartz and Prof. Maya Schuldiner from Weizmann Institute of Science for plasmids and strains, and Dr. Avigail Atir-Lande for advice and assistance. This work was funded by Israel Science Foundation Grant 258/18.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Levi, O., Arava, Y. Expanding the CRISPR/Cas9 Toolbox for Gene Engineering in S. cerevisiae. Curr Microbiol 77, 468–478 (2020). https://doi.org/10.1007/s00284-019-01851-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-019-01851-0