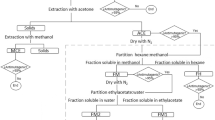
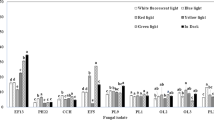
Abstract
Phenolic (free, conjugated and bound) and carotenoid extracts from microalgae Nannochloropsis sp. and Spirulina sp. were investigated regarding their potential to mitigate contamination by Fusarium complex fungal pathogens. Free phenolic acid extracts from both microalgae were the most efficient, promoting the lowest mycelial growth rates of 0.51 cm day− 1 (Spirulina sp.) and 0.78 cm day− 1 (Nannochloropsis sp.). An experiment involving natural free phenolic acid extracts and synthetic solutions was carried out based on the natural phenolic acid profile. The results revealed that the synthetic mixtures of phenolic acids from both microalgae were less efficient than the natural extracts at inhibiting fungal growth, indicating that no purification is required. The half-maximal effective concentration (EC50) values of 49.6 μg mL− 1 and 33.9 μg mL− 1 were determined for the Nannochloropsis and Spirulina phenolic acid extracts, respectively. The use of phenolic extracts represents a new perspective regarding the application of compounds produced by marine biotechnology to prevent Fusarium species contamination.




Similar content being viewed by others
References
Atanasova-Penichon V, Barreauand C, Richard-Forget F (2016) Antioxidant secondary metabolites in cereals: potential involvement in resistance to Fusarium and mycotoxin accumulation. Front Microbiol 22:7–566. https://doi.org/10.3389/fmicb.2016.00566
Baskaran R, Pullencheri D, Somasundaram R (2016) Characterization of free, esterified and bound phenolics in custard apple (Annona squamosa L) fruit pulp by UPLC–ESI–MS/MS. Food Res Int 82:121–127. https://doi.org/10.1016/j.foodres.2016.02.001
Borges L, Morón-Villarreyes JA, D’Oca MGM, Abreu PC (2011) Effects of flocculants on lipid extraction and fatty acid composition of the microalgae Nannochloropsis oculata and Thalassiosira weissflogii. Biomass Bioenergy 35:4449–4454. https://doi.org/10.1016/j.biombioe.2011.09.003
Cipolatti EP, Bulsing BA, Sá CS, Burkert CAV, Badiale-Furlong E, Burkert JFM (2015) Carotenoids from Phaffia rhodozyma: antioxidant activity and stability of extracts. Afr J Biotechnol 14(23):1982–1988. https://doi.org/10.5897/AJB2015.14682
Del Ponte E, Spolti P, Ward T, Gomes LB, Nicolli CP, Kuhnem PR, Silva CN, Tessmann DJ (2015) Regional and field-specific factors affect the composition of Fusarium head blight pathogens in subtropical no-till wheat agroecosystem of Brazil. Phytopathology 105:246–254. https://doi.org/10.1094/PHYTO-04-14-0102-R
Dors GC, Caldas SS, Fagundes CAA, Primel EG, Badiale-Furlong E (2013) Fungicides and the effects on mycotoxins on milling fractions of irrigated rice. J Agric Food Chem 61:1985–1999. https://doi.org/10.1021/jf305144t
Freitas RF, Schrack EC, He Q, Silliman BR, Furlong EB, Telles AC, Costa CSB (2016) Consumer control of the establishment of marsh foundation plants in intertidal mudflats. Mar Ecol Prog Ser 547:79–89. https://doi.org/10.3354/meps11624
Gomes LB, Ward TJ, Furlong EB, Del Ponte EM (2015) Species composition, toxigenic potential and pathogenicity of Fusarium graminearum species complex isolates from southern Brazilian rice. Plant Pathol 64:980–987. https://doi.org/10.1111/ppa.12332
Gutarowska B, Zakowska Z (2009) Mathematical models of mycelium growth and ergosterol synthesis in stationary mould culture. Lett Appl Microbiol 48:605–610. https://doi.org/10.1111/j.1472-765X.2009.02577.x
Heidtmann-Bemvenuti R, Tralamazza SM, Ferreira CFJ, Corrêa B, Badiale-Furlong E (2016) Effect of natural compounds on Fusarium graminearum complex. J Sci Food Agric 96(12):3998–4008. https://doi.org/10.1002/jsfa.7591
Leslie JF, Summerell BA (2006) The Fusarium laboratory manual. Blackwell Publishing, Ames
Millao S, Uquiche E (2016) Antioxidant activity of supercritical extracts from Nannochloropsis gaditana: correlation with its content of carotenoids and tocopherols. J Supercrit Fluids 111:143–150. https://doi.org/10.1016/j.supflu.2016.02.002
Morais MG, Radmann EM, Andrade MR, Teixeira GG, Brusch LRF, Costa JAV (2009) Pilot scale semicontinuous production of Spirulina biomass in southern Brazil. Aquaculture 294:60–64. https://doi.org/10.1016/j.aquaculture.2009.05.009
Nguefack J, Leth V, Amvam Zollo PH, Mathur SB (2004) Evaluation of five essential oils from aromatic plants of Cameroon for controlling food spoilage and mycotoxin producing fungi. Int J Food Microbiol 94:329–334. https://doi.org/10.1016/j.ijfoodmicro.2004.02.017
Pagnussatt FA, Del Ponte EM, Garda-Buffon J, Badiale-Furlong E (2014) Inhibition of Fusarium graminearum growth and mycotoxin production by phenolic extract from Spirulina sp. Pestic Biochem Physiol 108:21–26. https://doi.org/10.1016/j.pestbp.2013.11.002
Pagnussatt FA, de Lima VR, Dora CL, Costa JAV, Putaux JL, Badiale-Furlong E (2016) Assessment of the encapsulation effect of phenolic compounds from Spirulina sp. LEB-18 on their antifusarium activities. Food Chem 211:616–623. https://doi.org/10.1016/j.foodchem.2016.05.098
Pagnussatt FA, Kupski L, Darley FT, Filoda PF, Del Ponte EM, Garda-Buffon J, Badiale-Furlong E (2013) Fusarium graminearum growth inhibition mechanism using phenolic compounds from Spirulina sp. Ciênc Tecnol Aliment 33:75–80. https://doi.org/10.1590/S0101-20612013000500012
Paliwal C, Ghosh T, George B, Pancha I, Maurya R, Chokshi K, Ghosh A, Mishra S (2016) Microalgal carotenoids: potential nutraceutical compounds with chemotaxonomic importance. Algal Res 15:24–31. https://doi.org/10.1016/j.algal.2016.01.017
Quideau S, Deffieux D, Douat-Casassus C, Pouyégu L (2011) Plant polyphenols: chemical properties, biological activities, and synthesis. Angew Chem Int Ed (Engl) 50:586–621. https://doi.org/10.1002/anie.201000044
Rodriguez-Amaya DB (2001) A guide to carotenoid analysis in food. Human Nutrition Institute—International Life Sciences Institute, Washington, DC
Scaglioni PT, Badiale-Furlong E (2017) Can microalgae act as source of preservatives in food chain? J Food Sci Eng 7:283–296. https://doi.org/10.17265/2159-5828/2017.06.001
Scaglioni PT, Blandino M, Scarpino V, Giordano D, Testa G, Furlong EB (2018) Application of fungicides and microalgal phenolic extracts for the direct control of Fumonisin contamination in maize. J Agric Food Chem 66:4835–4841. https://doi.org/10.1021/acs.jafc.8b00540
Scaglioni PT, de Garcia SO, Badiale-Furlong E (2018) Inhibition of in vitro trichothecenes production by microalgae phenolic extracts. Food Res Int. https://doi.org/10.1016/j.foodres.2018.07.008
Scaglioni PT, Quadros L, Paula M, Furlong VB, Abreu PC, Furlong EB (2018) Inhibition of enzymatic and oxidative process by phenolic extracts from Spirulina sp. and Nannochloropsis sp. Food Technol Biotechnol 56:16. https://doi.org/10.17113/ftb.56.03.18.5495
Shao Y, Pan J, Zhang C, Jiang L, He Y (2015) Detection in situ of carotenoid in microalgae by transmission spectroscopy. Comput Electron Agric 112:121–127. https://doi.org/10.1016/j.compag.2014.10.008
Shukla R, Singh P, Prakash B, Dubey NK (2012) Antifungal, aflatoxin inhibition and antioxidant activity of Callistemon lanceolatus (Sm.) sweet essential oil and its major component 1,8-cineole against fungal isolates from chickpea seeds. Food Control 25:27–33. https://doi.org/10.1016/j.foodcont.2011.10.010
Singh A, Bajpai V, Kumar S, Kumar B, Srivastava M, Rameshkumar KB (2016) Comparative profiling of phenolic compounds from different plant parts of six Terminalia species by liquid chromatography–tandem mass spectrometry with chemometric analysis. Ind Crops Prod 87:236–246. https://doi.org/10.1016/j.indcrop.2016.04.048
Skroza D, Mekinic IG, Svilovic S, Simat V, Katalinic V (2015) Investigation of the potential synergistic effect of resveratrol with other phenolic compounds: a case of binary phenolic mixtures. J Food Compos Anal 38:13–18. https://doi.org/10.1016/j.jfca.2014.06.013
Souza MM, Prietto L, Ribeiro AC, Souza TD, Badiale-Furlong E (2011) Assessment of the antifungal activity of Spirulina platensis phenolic extract against Aspergillus flavus. Ciênc Agrotecnol 35(6):1050–1058. https://doi.org/10.1590/S1413-70542011000600003
Stanisz E, Zgola-Grzeskowiak A, Waskiewicz A, Stepien L, Beszterda M (2015) Can ergosterol be an indicator of Fusarium fungi and mycotoxins in cereal products? J Braz Chem Soc 26:705–712. https://doi.org/10.5935/0103-5053.20150030
Acknowledgements
The authors thank the Programa de Pós-Graduação em Engenharia e Ciência de Alimentos (FURG) and CNPq-Conselho Nacional de Desenvolvimento Científico e Tecnológico for supported this study by the Project 472633/2012.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Scaglioni, P.T., Pagnussatt, F.A., Lemos, A.C. et al. Nannochloropsis sp. and Spirulina sp. as a Source of Antifungal Compounds to Mitigate Contamination by Fusarium graminearum Species Complex. Curr Microbiol 76, 930–938 (2019). https://doi.org/10.1007/s00284-019-01663-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-019-01663-2