Abstract
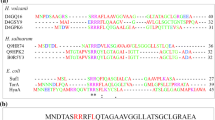
The twin-arginine translocation (Tat) system, which is used for folded protein secretion, is rare in lactic acid bacteria (LAB). Previously, a Tat system composed of TatAS and TatCS subunits (the subscript S denotes a Streptococcus thermophilus origin) was identified in S. thermophilus CGMCC 7.179. In the present study, the tatA S and tatC S genes were cloned and functionally analyzed in Escherichia coli DE3 tat-deficient mutants. The E. coli tatABCDE-deficient mutant complemented with tatC S A S exhibited shortened cellular chains, but its ability to grow in the presence of sodium dodecyl sulfate (SDS) was not restored, suggesting that the S. thermophilus Tat system could partially replace that of E. coli. Surprisingly, the E. coli tatABE-deficient mutant complemented with tatA S and the E. coli tatC-deficient mutant complemented with tatC S displayed relatively normal cellular morphology and enhanced tolerance to SDS. These results suggest that TatAS and TatCS could serve as active protein translocases in E. coli DE3 tat-deficient mutants. Moreover, TatAS acted as a bifunctional subunit to fulfill the roles of both TatA and TatB of E. coli DE3. Thus, this minimal Tat system would be a promising candidate to translocate recombinant proteins in LAB.



Similar content being viewed by others
References
Barnett JP, Eijlander RT, Kuipers OP, Robinson CA (2008) Minimal Tat system from a gram-positive organism: a bifunctional TatA subunit participates in discrete TatAC and TatA complexes. J Biol Chem 283(5):2534–2542
Berks BC (1996) A common export pathway for proteins binding complex redox cofactors. Mol Microbiol 22:393–404
Bernhardt TG, de Boer PA (2003) The Escherichia coli amidase AmiC is a periplasmic septal ring component exported via the twin-arginine transport pathway. Mol Microbiol 47(5):1171
Biswas L, Biswas R, Nerz C, Ohlsen K, Schlag M, Schafer T et al (2009) Role of the Twin-Arginine translocation pathway in Staphylococcus. J Bacteriol 191(19):5921–5929
Blaudeck N, Kreutzenbeck P, Muller M, Sprenger GA, Freudl R (2005) Isolation and characterization of bifunctional Escherichia coli TatA mutant proteins that allow efficient Tat-dependent protein translocation in the absence of TatB. J Biol Chem 280(5):3426–3432
Cherepanov PP, Wackernagel W (1995) Gene disruption in Escherichia coli: Tc R and Km R cassettes with the option of Flp-catalyzed excision of the antibiotic-resistance determinant. Gene 158(1):9–14
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci USA 97(12):6640–6645
DeLisa MP, Tullman D, Georgiou G (2003) Folding quality control in the export of proteins by the bacterial twin-arginine translocation pathway. Proc Natl Acad Sci USA 100(10):6115–6120
Goosens VJ, Monteferrante CG, van Dijl JM (2014) The Tat system of Gram-positive bacteria. Biochim Biophys Acta 1843(8):1698–1706
Hols P, Hancy F, Fontaine L, Grossiord B, Prozzi D, Leblond-Bourget N et al (2005) New insights in the molecular biology and physiology of Streptococcus thermophilus revealed by comparative genomics. FEMS Microbiol Rev 29(3):435–463
Hu Y, Zhao E, Li H, Xia B, Jin C (2010) Solution NMR structure of the TatA component of the twin-arginine protein transport system from gram-positive bacterium Bacillus subtilis. J Am Chem Soc 132(45):15942
Ize B, Stanley NR, Buchanan G, Palmer T (2003) Role of the Escherichia coli Tat pathway in outer membrane integrity. Mol Microbiol 48(5):1183–1193
Jongbloed JD, Grieger U, Antelmann H, Hecker M, Nijland R, Bron S et al (2004) Two minimal Tat translocases Bacillus. Mol Microbiol 54(5):1319–1325
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H et al (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23(21):2947–2948
Le Loir Y, Gruss A, Ehrlich SD, Langella PA (1998) Nine-residue synthetic propeptide enhances secretion efficiency of heterologous proteins in Lactococcus lactis. J Bacterio 180(7):1895–1903
Matos CF, Di Cola A, Robinson C (2009) TatD is a central component of a Tat translocon-initiated quality control system for exported FeS proteins in Escherichia coli. EMBO Rep 10(5):474–479
Mori H, Cline K (2001) Post-translational protein translocation into thylakoids by the Sec and ∆pH-dependent pathways. Biochim Biophys Acta 1541(1):80–90
Palmer T, Berks BC (2012) The twin-arginine translocation (Tat) protein export pathway. Nat Rev Microbiol 10(7):483
Robert X, Gouet P (2014) Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res 42:320–324
Rodriguez F, Rouse SL, Tait CE, Harmer J, De Riso A, Timmel CR et al (2013) Structural model for the protein-translocating element of the twin-arginine transport system. Proc Natl Acad Sci USA 110(12):101
Rollauer SE, Tarry MJ, Graham JE, Jääskeläinen M, Jager F, Johnson S et al (2012) Structure of the TatC core of the twin-arginine protein transport system. Nature 492(7428):210
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. BioScience 3:2344
Sauve V, Bruno S, Berks BC, Hemmings AM (2007) The soxYZ complex carries sulfur cycle intermediates on a peptide swinging arm. J Biol Chem 282(32):23194–23204
Tamime AY, Deeth HC (1980) Yoghurt: Technology and biochemistry. J Food Prot 43(12):939–977
Tiwari KB, Birlingmair J, Wilkinson BJ, Jayaswal RK (2015) Role of the twin-arginine translocase (tat) system in iron uptake in Listeria monocytogenes. Microbiology 161(2):264–271
Weiner JH, Bilous PT, Shaw GM, Lubitz SP, Frost L, Thomas GH et al (1998) A novel and ubiquitous system for membrane targeting and secretion of cofactor-containing proteins. Cell 93(1):93–101
Zhang C, Xin Y, Wang Y, Guo T, Lu S, Kong J (2015) Identification of a novel dye-decolorizing peroxidase, EfeB, translocated by a twin-arginine translocation system in Streptococcus thermophilus CGMCC 7.179. Appl Environ Microbiol 81(18):6108–6119
Zhang Y, Wang L, Hu Y, Jin C (2014) Solution structure of the TatB component of the twin-arginine translocation system. Biochim Biophys Acta 1838(7):1881–1888
Zoufaly S, Frobel J, Rose P, Flecken T, Maurer C, Moser M et al (2012) Mapping precursor-binding site on Tatc subunit of twin arginine-specific protein translocase by site-specific photo cross-linking. J Biol Chem 287(16):13430–13441
Acknowledgements
We would like to thank N. Galleron for kindly providing the plasmid pSec:leiss:Nuc. This work was supported by National Natural Science Foundation of China (No. 31471715), Public Service Sectors (Agriculture) Special and Scientific Research Projects (No. 201503134) and National Natural Science Foundation of China (No. 31571855).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, C., Guo, T., Xin, Y. et al. Functional Analysis of the Minimal Twin-Arginine Translocation System Components from Streptococcus thermophilus CGMCC 7.179 in Escherichia coli DE3. Curr Microbiol 74, 678–684 (2017). https://doi.org/10.1007/s00284-017-1234-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-017-1234-x