Abstract
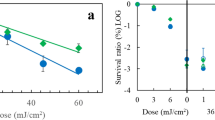
Three bacterial strains—Escherichia coli, Acinetobacter calcoaceticus, and the A. calcoaceticus RecA(−) mutant—underwent photosensitization by a low-concentration (0.73 μmol/L) tetramethyl pyridyl porphine (a cationic hydrophylic photosensitizer) and a 4-J/cm2 dose of 407 to 420 nm blue light. The viability of the first two strains decreased by approximately 60%. and that of the RecA(−) strain decreased by 90%. Increasing the amount of photosensitizer to 14.6 μmol/L at the same dose of blue light resulted in a 95% to 98% decrease in viability of the three strains. Very little damage to the bacterial DNA was observed after this treatment. Increasing the concentration photosensitizer under the same illumination conditions also resulted in very little damage to the DNA. Western blotting demonstrated that the low photosensitization procedures enhance RecA production for mending the damaged chromosomal DNA. RecA production as a result of low-dose photosensitization was confirmed and demonstrated by immunofluorescent staining and gold immunolabeling. Although DNA is not the primary target for photosensitization, this process of RecA production may provide a certain degree of DNA mending and may also affect the survival of bacterial cells on low-intensity photosensitization.




Similar content being viewed by others
Literature Cited
Malik Z, Gozhansky S, Nitzan Y (1982) Effect of photoactivated hematoporphyrin derivative on bacteria and antibiotic resistance. Microbios Lett 21:103–112
Nitzan Y, Balzam-Sudakevitz A, Ashkenazi H (1998) Eradication of Acinetobacter baumannii by photosensitized agents in vitro. J Photochem Photobiol B 42:211–218
Bertoloni G, Salvato B, Dall, Acqua M, Vazzoler M, Jori G (1984) Hematoporphyrin-sensitized photoinactivation of Streptococcus faecalis. Photochem Photobiol 39:811–816
Nitzan Y, Gutterman M, Malik Z, Ehrenberg B (1992) Inactivation of Gram negative bacteria by photosensitized porphyrins. Photochem Photobiol 55:89–96
Nitzan Y, Shainberg B, Malik Z (1987) Photodynamic effects of deuteroporphyrin on Gram positive bacteria. Curr Microbiol 15:251–258
Nitzan Y, Gozhansky S, Malik Z (1983) Effect of photoactivated hematoporphyrin derivative on the viability of Staphylococcus aureus. Curr Microbiol 8:279–284
Merchat M, Bertolini G, Giacomini P, Villanueva A, Jori G (1996) Meso-substituted cationic porphyrins as efficient photosensitizers of gram-positive and gram-negative bacteria. J Photochem Photobiol B 32:153–157
Merchat M, Spikes JD, Bertoloni G, Jori G (1996) Studies on the mechanism of bacteria photosensitization by meso-substituted cationic porphyrins. J Photochem Photobiol B 35:149–157
Minnock A, Vernon DI, Schofield J, Griffiths J, Parish JH, Brown ST (1996) Photoinactivation of bacteria. Use of a cationic water-soluble zinc phthalocyanine to photoinactivate both gram-negative and gram-positive bacteria. J Photochem Photobiol B 32:159–164
Nitzan Y, Ashkenazi H (1999) Photoinactivation of Deinococcus radiodurans: An unusual Gram-positive microorganism. Photochem Photobiol 69:505–510
Valduga G, Breda B, Giacometti GM, Jori G, Reddi E (1999) Photosensitization of wild and mutant strains of Escherichia coli by meso-tetra (N-methyl-4-pyridyl)porphine. Biochem Biophys Res Commun 256:84–88
Nitzan Y, Ashkenazi H (2001) Photoinactivation of Acinetobacter baumannii and Escherichia coli B by a cationic hydrophilic porphyrin at various light wavelengths. Curr Microbiol 42:408–414
Yu X, Egelman EH (1993) The LexA repressor binds within the deep helical groove of the activated RecA filament. J Mol Biol 231:29–40
Kurumizaka H, Aihara H, Ikawa S, Kashima T, Bazemore LR, Kawasaki K, et al. (1996) A possible role of the C-terminal domain of the RecA protein. A gateway model for double-stranded DNA binding. J Biol Chem 271:33515–33524
Williams RC, Spengler SJ (1986) Fibers of RecA protein and complexes of RecA protein and single-stranded phi X174 DNA as visualized by negative-stain electron microscopy. J Mol Biol 187:109–118
Bryant FR, Taylor AR, Lehman IR (1985) Interaction of the recA protein of Escherichia coli with single-stranded DNA. J Biol Chem 260:1196–1202
Levin-Zaidman S, Frenkiel-Krispin D, Shimoni E, Sabanay I, Wolf SG, Minsky A (2000) Ordered intracellular RecA-DNA assemblies: a potential site of in vivo RecA-mediated activities. Proc Natl Acad Sci U S A 97:6791–6796
Yu X, Egelman EH (1992) Structural data suggest that the active and inactive forms of the RecA filament are not simply interconvertible. J Mol Biol 227:334–346
Ogawa T, Yu X, Shinohara A, Egelman EH (1993) Similarity of the yeast RAD51 filament to the bacterial RecA filament. Science 259:1896–1899
Egelman EH, Yu X (1989) The location of DNA in RecA-DNA helical filaments. Science 245:404–407
Palmen R, Vosman B, Kok R, van der Zee JR, Hellingwerf KJ (1992) Characterization of transformation-deficient mutants of Acinetobacter calcoaceticus. Mol Microbiol 6:1747–1754
Wilson K. (1998) Preparation of genomic DNA from bacteria. In: Ausubel F, Brent R, Kingston R, Moore P, Seidman F, Smith J, et al. (eds) Current protocols in molecular biology, vol. 1. New York, NY: Wiley, pp 241–245
Sambrook J, Russell D, Sambrook J (2001) Molecular cloning: A laboratory manual. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press
Harlow E, Lane D (1998) Antibodies: A laboratory manual. New York, NY: Cold Spring Harbor Laboratory Press
Ashkenazi H, Malik Z, Harth Y, Nitzan Y (2003) Eradication of Propionibacterium acnes by its endogenic porphyrins after illumination with high intensity blue light. FEMS Immunol Med Microbiol 35:17–24
Nitzan Y, Shainberg B, Malik Z (1989) The mechanism of photodynamic inactivation of S. aureus by deuteroporphyrin. Curr Microbiol 19:265–269
Nir U, Ladan H, Malik Z, Nitzan Y (1991) In vivo effects of porphyrins on bacterial DNA. J Photochem Photobiol B 11:295–306
Ashkenazi H, Nitzan Y, Gal D (2003) Photodynamic effects of antioxidant substituted porphyrin photosensitizers on gram-positive and -negative bacterial. Photochem Photobiol 77:186–191
Salmon-Divon M, Nitzan Y, Malik Z (2004) Mechanistic aspects of Escherichia coli photodynamic inactivation by cationic tetra-meso(N-methylpyridyl)porphine. Photochem Photobiol Sci 3:423–429
Courcelle J, Khodursky A, Peter B, Brown PO, Hanawalt PC (2001) Comparative gene expression profiles following UV exposure in wild-type and SOS-deficient Escherichia coli. Genetics 158:41–64
Levin-Zaidman S, Englander J, Shimoni E, Sharma SK, Minton KW, Minsky A (2003) Ringlike structure of Deinoccus radiodurans genome: A key to radioresistance? Science 299:254–256
Acknowledgments
This work was supported in part by a grant from the Health Sciences Research Center Funds (to Y.N.) and in part by the Rappaport Foundation for Medical Microbiology (to Y.N.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ashkenazi, H., Pechatnikov, I. & Nitzan, Y. Low-Intensity Photosensitization May Enhance RecA Production. Curr Microbiol 52, 317–323 (2006). https://doi.org/10.1007/s00284-005-0323-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-005-0323-4