Abstract
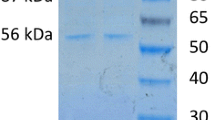
The first cloning and characterization of the gene katA, encoding the major catalase (KatA), from Xanthomonas is reported. A reverse genetic approach using a synthesized katA-specific DNA probe to screen a X. campestris pv. phaseoli genomic library was employed. A positively hybridizing clone designated pKat29 that contained a full-length katA was isolated. Analysis of the nucleotide sequence revealed an open reading frame of 1,521 bp encoding a 507-amino acid protein with a theoretical molecular mass of 56 kDa. The deduced amino acid sequence of KatA revealed 84% and 78% identity to CatF of Pseudomonas syringae and KatB of P. aeruginosa, respectively. Phylogenetic analysis places Xanthomonas katA in the clade I group of bacterial catalases. Unexpectedly, expression of katA in a heterologous Escherichia coli host resulted in a temperature-sensitive expression. The KatA enzyme was purified from an overproducing mutant of X. campestris and was characterized. It has apparent K m and V max values of 75 mM [H2O2] and 2.55 × 105 μmol H2O2 μmol heme−1 s−1, respectively. The enzyme is highly sensitive to 3-amino-1,2,4-triazole and NaN3, has a narrower optimal pH range than other catalases, and is more sensitive to heat inactivation.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 4 March 2002 / Accepted: 8 April 2002
Rights and permissions
About this article
Cite this article
Chauvatcharin, N., Vattanaviboon, P., Switala, J. et al. Cloning and Characterization of katA, Encoding the Major Monofunctional Catalase from Xanthomonas campestris pv. phaseoli and Characterization of the Encoded Catalase KatA. Curr Microbiol 46, 0083–0087 (2003). https://doi.org/10.1007/s00284-002-3812-8
Issue Date:
DOI: https://doi.org/10.1007/s00284-002-3812-8