Abstract
Purpose
The ability of predicting severe adverse reactions caused by regorafenib is important. We evaluated regorafenib concentrations for adverse reaction risks and assessed the relevance of laboratory values and gene polymorphisms.
Methods
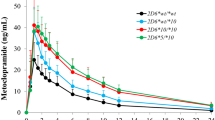
A total of 28 Japanese cancer patients who were treated with regorafenib were evaluated for the steady state of serum regorafenib concentrations and adverse reactions for 28 days. In addition, we determined the association of regorafenib concentrations with ABCG2 and OATP1B1 polymorphisms, which are regorafenib transporters.
Results
Regorafenib concentrations were significantly higher in the group with Grade 2 or higher total bilirubin elevation and thrombocytopenia compared with the group with grades 0 or 1 [3.45 (2.18–7.31) vs. 1.76 (0.26–2.77) µg/mL, P = 0.01 and 3.45 (2.12–7.31) vs. 1.76 (0.26–2.77) µg/mL, P = 0.02, respectively]. A strong association was noted between serum regorafenib concentrations and total bilirubin levels, but the physical and genetic factors predicting regorafenib pharmacokinetics could not be clarified.
Conclusions
Regorafenib concentrations were associated with total bilirubin elevation and thrombocytopenia. Total serum bilirubin could be a useful marker when estimating regorafenib pharmacokinetics.



Similar content being viewed by others
References
Grothey A, Van Cutsem E, Sobrero A et al (2013) Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381:303–312
Mross K, Frost A, Steinbild S et al (2012) A phase I dose-escalation study of regorafenib (BAY 73-4506), an inhibitor of oncogenic, angiogenic, and stromal kinases, in patients with advanced solid tumors. Clin Cancer Res 18:2658–2667
Osawa H (2017) Response to regorafenib at an initial dose of 120 mg as salvage therapy for metastatic colorectal cancer. Mol Clin Oncol 6:365–372
Kort A, Durmus S, Sparidans RW et al (2015) Brain and testis accumulation of regorafenib is restricted by breast cancer resistance protein (BCRP/ABCG2) and P-glycoprotein (P-GP/ABCB1). Pharm Res 32:2205–2216
Ohya H, Shibayama Y, Ogura J et al (2015) Regorafenib is transported by the organic anion transporter 1B1 and the multidrug resistance protein 2. Biol Pharm Bull 38:582–586
Maeda A, Ando H, Ura T et al (2017) Association between ABCG2 and SLCO1B1 polymorphisms and adverse drug reactions to regorafenib: a preliminary study. Int J Clin Pharmacol Ther 55:409–415
Allard M, Khoudour N, Rousseau B et al (2017) Simultaneous analysis of regorafenib and sorafenib and three of their metabolites in human plasma using LC-MS/MS. J Pharm Biomed Anal 142:42–48
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl 48:452–458
Sunakawa Y, Furuse J, Okusaka T et al (2014) Regorafenib in Japanese patients with solid tumors: phase I study of safety, efficacy, and pharmacokinetics. Investig New Drugs 32:104–112
Strumberg D, Scheulen ME, Schultheis B et al (2012) Regorafenib (BAY 73-4506) in advanced colorectal cancer: a phase I study. Br J Cancer 106:1722–1727
Cui Y, Konig J, Leier I et al (2001) Hepatic uptake of bilirubin and its conjugates by the human organic anion transporter SLC21A6. J Biol Chem 276:9626–9630
Briz O, Serrano MA, MacIas RI et al (2003) Role of organic anion-transporting polypeptides, OATP-A, OATP-C and OATP-8, in the human placenta-maternal liver tandem excretory pathway for foetal bilirubin. Biochem J 371:897–905
Campbell SD, de Morais SM, Xu JJ (2004) Inhibition of human organic anion transporting polypeptide OATP 1B1 as a mechanism of drug-induced hyperbilirubinemia. Chem Biol Interact 150:179–187
Zhao B, Zhao H (2017) Incidence and risk of regorafenib-induced hepatotoxicity. Oncotarget 8:84102–84111
Keppler D (2014) The roles of MRP2, MRP3, OATP1B1, and OATP1B3 in conjugated hyperbilirubinemia. Drug Metab Dispos 42:561–565
Schultheis B, Folprecht G, Kuhlmann J et al (2013) Regorafenib in combination with FOLFOX or FOLFIRI as first- or second-line treatment of colorectal cancer: results of a multicenter, phase Ib study. Ann Oncol 24:1560–1567
Teft WA, Welch S, Lenehan J et al (2015) OATP1B1 and tumour OATP1B3 modulate exposure, toxicity, and survival after irinotecan-based chemotherapy. Br J Cancer 112:857–865
Nozawa T, Minami H, Sugiura S et al (2005) Role of organic anion transporter OATP1B1 (OATP-C) in hepatic uptake of irinotecan and its active metabolite, 7-ethyl-10-hydroxycamptothecin: in vitro evidence and effect of single nucleotide polymorphisms. Drug Metab Dispos 33:434–439
Pasanen MK, Fredrikson H, Neuvonen PJ et al (2007) Different effects of SLCO1B1 polymorphism on the pharmacokinetics of atorvastatin and rosuvastatin. Clin Pharmacol Ther 82:726–733
Nishizato Y, Ieiri I, Suzuki H et al (2003) Polymorphisms of OATP-C (SLC21A6) and OAT3 (SLC22A8) genes: consequences for pravastatin pharmacokinetics. Clin Pharmacol Ther 73:554–565
Ieiri I, Higuchi S, Sugiyama Y (2009) Genetic polymorphisms of uptake (OATP1B1, 1B3) and efflux (MRP2, BCRP) transporters: implications for inter-individual differences in the pharmacokinetics and pharmacodynamics of statins and other clinically relevant drugs. Expert Opin Drug Metab Toxicol 5:703–729
Deng JW, Song IS, Shin HJ et al (2008) The effect of SLCO1B1*15 on the disposition of pravastatin and pitavastatin is substrate dependent: the contribution of transporting activity changes by SLCO1B1*15. Pharmacogenet Genom 18:424–433
Choi JH, Lee MG, Cho JY et al (2008) Influence of OATP1B1 genotype on the pharmacokinetics of rosuvastatin in Koreans. Clin Pharmacol Ther 83:251–257
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Maeda, A., Irie, K., Ando, H. et al. Associations among regorafenib concentrations, severe adverse reactions, and ABCG2 and OATP1B1 polymorphisms. Cancer Chemother Pharmacol 83, 107–113 (2019). https://doi.org/10.1007/s00280-018-3710-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3710-9