Abstract
Purpose
This was an open label, phase I (3 + 3 design), multi-centre study evaluating panobinostat in pediatric patients with refractory solid tumors.
Methods
Primary endpoints were to establish MTD, define and describe associated toxicities, including dose limiting toxicities (DLT) and to characterize its pharmacokinetics (PK). Secondary endpoints included assessing the anti-tumour activity of panobinostat, and its biologic activity, by measuring acetylation of histones in peripheral blood mononuclear cells.
Results
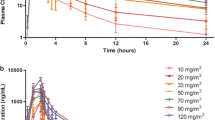
Nine patients were enrolled and treated with intravenous panobinostat at a dosing level of 15 mg/m2 which was tolerated. Six were evaluable for adverse events. Two (33%) patients experienced Grade 3–4 thrombocytopenia, 1 (17%) experienced Grade 3 anemia, and 2 (33%) experienced Grade 3 neutropenia. Grade 4 drug related pain occurred in 2 (33%) of the patients studied. Two (33%) patients experienced a Grade 2 QTcF change (0.478 ± 0.006 ms). One cardiac DLT (T wave changes) was reported. PK values for 15 mg/m2 (n = 9) dosing were: Tmax 0.8 h, Cmax 235.2 ng/mL, AUC0–t 346.8 h ng/mL and t1/2 7.3 h. Panobinostat significantly induced acetylation of histone H3 and H4 at all time points measured when compared to pre-treatment samples (p < 0.05). Pooled quantitative Western blot data confirmed that panobinostat significantly induced acetylation of histone H4 at 6 h (p < 0.01), 24 h (p < 0.01) and 28–70 h (p < 0.01) post dose.
Conclusion
A significant biological effect of panobinostat, measured by acetylation status of histone H3 and H4, was achieved at a dose of 15 mg/m2. PK data and drug tolerability at 15 mg/m2 was similar to that previously published.



Similar content being viewed by others
References
Wolffe AP (1994) Transcriptional activation. Switched-on chromatin. Curr Biol 4(6):525–528
Yoshida M, Furumai R, Nishiyama M et al (2001) Histone deacetylase as a new target for cancer chemotherapy. Cancer Chemother Pharmacol 48(Suppl 1):S20–S26
Marks P, Rifkind R, Richon V et al (2001) Histone deacetylases and cancer: causes and therapies. Nat Rev 1:194–202
Weidle UH, Grossmann A (2000) Inhibition of histone deacetylases: a new strategy to target epigenetic modifications for anticancer treatment. Anticancer Res 20(3A):1471–1485
Wade PA, Wolffe AP (1997) Histone acetyltransferases in control. Curr Biol 7(2):R82–R84
Deckert J, Struhl K (2001) Histone acetylation at promoters is differentially affected by specific activators and repressors. Mol Cell Biol 21(8):2726–2735
Gray SG, Ekstrom TJ (2001) The human histone deacetylase family. Exp Cell Res 262(2):75–83
Giles RH, Peters DJ, Breuning MH (1998) Conjunction dysfunction: CBP/p300 in human disease. Trends Genet 14(5):178–183
Gayther SA, Batley SJ, Linger L et al (2000) Mutations truncating the EP300 acetylase in human cancers. Nat Genet 24(3):300–303
Murata T, Kurokawa R, Krones A et al (2001) Defect of histone acetyltransferase activity of the nuclear transcriptional coactivator CBP in Rubinstein–Taybi syndrome. Hum Mol Genet 10(10):1071–1076
Sakai K, Nagahara H, Abe K et al (1992) Loss of heterozygosity on chromosome 16 in hepatocellular carcinoma. J Gastroenterol Hepatol 7(3):288–292
Rowley JD, Reshmi S, Sobulo O et al (1997) All patients with the T(11;16)(q23;p13.3) that involves MLL and CBP have treatment-related hematologic disorders. Blood 90(2):535–541
Irminger-Finger I, Siegel BD, Leung WC (1999) The functions of breast cancer susceptibility gene 1 (BRCA1) product and its associated proteins. J Biol Chem 380(2):117–128
Kutko MC, Glick RD, Butler LM et al (2003) Histone deacetylase inhibitors induce growth suppression and cell death in human rhabdomyosarcoma in vitro. Clin Cancer Res 9(15):5749–5755
Feinberg AP, Cui H, Ohlsson R (2002) DNA methylation and genomic imprinting: insights from cancer into epigenetic mechanisms. Semin Cancer Biol 12(5):389–398
Waldeck K, Cullinane C, Ardley K et al (2016) Long term, continuous exposure to panobinostat induces terminal differentiation and long term survival in the TH-MYCN neuroblastoma mouse model. Int J Cancer 139(1):194–204
Muscat A, Popovski D, Jayasekara WS et al (2016) Low-dose histone deacetylase inhibitor treatment leads to tumour growth arrest and multi-lineage differentiation of malignant rhabdoid tumours. Clin Cancer Res 22:3560–3570. https://doi.org/10.1158/1078-0432.CCR-15-2260
Cain JE, McCaw A, Jayasekara WS et al (2013) Sustained low-dose treatment with the histone deacetylase inhibitor LBH589 induces terminal differentiation of osteosarcoma cells. Sarcoma 2013:608964
Coffey DC, Kutko MC, Glick RD et al (2001) The histone deacetylase inhibitor, CBHA, inhibits growth of human neuroblastoma xenografts in vivo, alone and synergistically with all-trans retinoic acid. Cancer Res 7(8):3591–3594
Coffey DC, Kutko MC, Glick RD et al (2000) Histone deacetylase inhibitors and retinoic acids inhibit growth of human neuroblastoma in vitro. Med Pediatr Oncol 35(6):577–581
Glick RD, Swendeman SL, Coffey DC et al (1999) Hybrid polar histone deacetylase inhibitor induces apoptosis and CD95/CD95 ligand expression in human neuroblastoma. Cancer Res 59(17):4392–4399
Biegel JA, Kalpana G, Knudsen ES et al (2002) The role of INI1 and the SWI/SNF complex in the development of rhabdoid tumours: meeting summary from the workshop on childhood atypical teratoid/rhabdoid tumours. Cancer Res 62(1):323–328
Prince HM, George D, Patnaik A et al (2007) Phase I study of oral LBH589, a novel deacetylase (DAC) inhibitor in advanced solid tumours and non-Hodgkins lymphoma. J Clin Oncol 25(18S):3500
Children’s Oncology Group, Fouladi M, Furman WL et al (2006) Phase I study of depsipeptide in pediatric patients with refractory solid tumors: a Children’s Oncology Group report. J Clin Oncol 24(22):3678–3685
Fouladi M, Park JR, Stewart CF et al (2010) Pediatric phase I trial and pharmacokinetic study of vorinostat: a Children’s Oncology Group phase I consortium report. J Clin Oncol 28(22):3623–3629
Chan K, Fang D, Gan H et al (2013) The histone H3.3K27M mutation in pediatric glioma reprograms H3K27 methylation and gene expression. Genes Dev 27:985–990
Grasso CS, Tang Y, Truffaux N et al (2015) Functionally defined therapeutic targets in diffuse intrinsic pontine glioma. Nat Med 21(6):555–559
Rigby L, Muscat A, Ashley D et al (2012) Methods for the analysis of histone H3 and H4 acetylation in blood. Epigenetics 7(8):875–882
Ronzoni S, Faretta M, Ballarini M et al (2005) New method to detect histone acetylation levels by flow cytometry. Cytometry 66A:52–61
Sharma S, Beck J, Mita M et al (2013) A phase I dose-escalation study of intravenous panobinostat in patients with lymphoma and solid tumors. Invest New Drugs 31(4):974–985
Anne M, Sammartino D, Barginear M et al (2013) Profile of panobinostat and its potential for treatment in solid tumors: an update. Onco Targets Ther 6:1613–1624
Giles F, Fischer T, Cortes J et al (2006) A phase I study of intravenous LBH589, a novel cinnamic hydroxamic acid analogue histone deacetylase inhibitor, in patients with refractory hematologic malignancies. Clin Cancer Res 12:4628–4635
Graham C, Tucker C, Creech J et al (2006) Evaluation of the antitumor efficacy, pharmacokinetics, and pharmacodynamics of the histone deacetylase inhibitor depsipeptide in childhood cancer models in vivo. Clin Cancer Res 12(1):223–234
Marquard L, Peterson K, Persson M et al (2008) Monitoring the effect of belinostat in solid tumors by H4 acetylation. APMIS 116(5):382–392
Prince HM, Bishton M (2009) Panobinostat (LBH589): a novel pan-deacetylase inhibitor with activity in T cell lymphoma. Hematol Meet Rep 3(1):33–38
Acknowledgements
This research would not have been possible without the generous support of the You Are My Sunshine (YAMS) Foundation and the Kathleen Tinsley Fellowship.
Funding
This trial was sponsored by the Australian and New Zealand Children’s Haematology and Oncology Group (ANZCHOG), 27 - 31 Wright Street, Clayton, VIC, 3168, Australia. This trial was supported by unrestricted funding and drug supply from Novartis Pharmaceuticals, Australia PTY Ltd., 54 Waterloo Road, Macquarie Park, NSW, 2113, Australia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Human and animal rights statement
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Patient and/or their legal guardians must have signed a written informed consent form.
Rights and permissions
About this article
Cite this article
Wood, P.J., Strong, R., McArthur, G.A. et al. A phase I study of panobinostat in pediatric patients with refractory solid tumors, including CNS tumors. Cancer Chemother Pharmacol 82, 493–503 (2018). https://doi.org/10.1007/s00280-018-3634-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3634-4