Abstract
Purpose
Doxorubicin, a highly effective and widely used anthracycline antibiotic in multiple chemotherapy regimens, has been limited by its cardiotoxicity. The aim of this study is to investigate the effect of nitric oxide donor prodrug JS-K on proliferation and apoptosis in renal carcinoma cells and cardiac myocytes toxicity induced by Doxorubicin and to explore possible p53-related mechanism in renal carcinoma cells.
Methods
The effect of JS-K on anti-cancer activity of Doxorubicin was investigated in renal carcinoma cells via detecting cell proliferation, cytotoxicity, cell death and apoptosis and expressions of apoptotic-related proteins. Effect of p53 on the combination of JS-K and Doxorubicin was determined using p53 inhibitor Pifithrin-α and p53 activator III. Furthermore, the effect of JS-K on cardiac myocytes toxicity of Doxorubicin was investigated in H9c2 (2-1) cardiac myocytes via measuring cell growth, cell death and apoptosis, expressions of proteins involved in apoptosis and intracellular reactive oxygen species.
Results
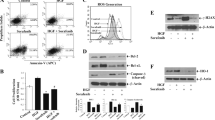
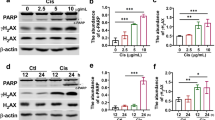
We demonstrated that JS-K could increase Doxorubicin-induced renal carcinoma cell growth suppression and apoptosis and could increase expressions of proteins that are involved in apoptosis. Additionally, Pifithrin-α reversed the promoting effect of JS-K on Doxorubicin-induced renal carcinoma cell apoptosis; conversely, the p53 activator III exacerbated the promoting effect of JS-K on Doxorubicin-induced renal carcinoma cell apoptosis. Furthermore, JS-K protected H9c2 (2-1) cardiac myocytes against Doxorubicin-induced toxicity and decreased Doxorubicin-induced reactive oxygen species production.
Conclusions
JS-K enhances the anti-cancer activity of Doxorubicin in renal carcinoma cells by upregulating p53 expression and prevents cardiac myocytes toxicity of Doxorubicin by decreasing oxidative stress.






Similar content being viewed by others
References
Smolensky D, Rathore K, Cekanova M (2015) Phosphatidylinositol- 3-kinase inhibitor induces chemosensitivity to a novel derivative of Doxorubicin, AD198 chemotherapy in human bladder cancer cells in vitro. BMC Cancer 15:927. doi:10.1186/s12885-015-1930-5
Zhu H, Luo P, Fu Y, Wang J, Dai J, Shao J, Yang X, Chang L, Weng Q, Yang B, He Q (2015) Dihydromyricetin prevents cardiotoxicity and enhances anticancer activity induced by adriamycin. Oncotarget 6(5):3254–3267. doi:10.18632/oncotarget.2410
Becker PS, Gooley TA, Green DJ, Burwick N, Kim TY, Kojouri K, Inoue Y, Moore DJ, Nelli E, Dennie T, Bensinger WI (2016) A phase 2 study of bortezomib, cyclophosphamide, pegylated liposomal Doxorubicin and dexamethasone for newly diagnosed multiple myeloma. Blood Cancer J 6:e422. doi:10.1038/bcj.2016.31
Ahmadi F, Mojarrab M, Ghazi-Khansari M, Hosseinzadeh L (2015) A semipolar fraction of petroleum ether extract of Artemisia aucheri induces apoptosis and enhances the apoptotic response to Doxorubicin in human neuroblastoma SKNMC cell line. Res Pharm Sci 10(4):335–344
Muggia FM, Speyer JL (1999) Doxorubicin-induced cardiomyopathy. N Engl J Med 340(8):654–655
Takemura G, Fujiwara H (2007) Doxorubicin-induced cardiomyopathy from the cardiotoxic mechanisms to management. Prog Cardiovasc Dis 49(5):330–352. doi:10.1016/j.pcad.2006.10.002
Mitry MA, Edwards JG (2016) Doxorubicin induced heart failure: phenotype and molecular mechanisms. Int J Cardiol Heart Vasc 10:17–24. doi:10.1016/j.ijcha.2015.11.004
Wang S, Konorev EA, Kotamraju S, Joseph J, Kalivendi S, Kalyanaraman B (2004) Doxorubicin induces apoptosis in normal and tumor cells via distinctly different mechanisms. intermediacy of H(2)O(2)- and p53-dependent pathways. J Biol Chem 279(24):25535–25543. doi:10.1074/jbc.M400944200
Hovorka O, Subr V, Vetvicka D, Kovar L, Strohalm J, Strohalm M, Benda A, Hof M, Ulbrich K, Rihova B (2010) Spectral analysis of Doxorubicin accumulation and the indirect quantification of its DNA intercalation. Eur J Pharm Biopharm 76(3):514–524. doi:10.1016/j.ejpb.2010.07.008
Kim KH, Oudit GY, Backx PH (2008) Erythropoietin protects against Doxorubicin-induced cardiomyopathy via a phosphatidylinositol 3-kinase-dependent pathway. J Pharm Exp Ther 324(1):160–169. doi:10.1124/jpet.107.125773
Venkatesan B, Prabhu SD, Venkatachalam K, Mummidi S, Valente AJ, Clark RA, Delafontaine P, Chandrasekar B (2010) WNT1-inducible signaling pathway protein-1 activates diverse cell survival pathways and blocks Doxorubicin-induced cardiomyocyte death. Cell Signal 22(5):809–820. doi:10.1016/j.cellsig.2010.01.005
Kiziltepe T, Hideshima T, Ishitsuka K, Ocio EM, Raje N, Catley L, Li CQ, Trudel LJ, Yasui H, Vallet S, Kutok JL, Chauhan D, Mitsiades CS, Saavedra JE, Wogan GN, Keefer LK, Shami PJ, Anderson KC (2007) JS-K, a GST-activated nitric oxide generator, induces DNA double-strand breaks, activates DNA damage response pathways, and induces apoptosis in vitro and in vivo in human multiple myeloma cells. Blood 110(2):709–718. doi:10.1182/blood-2006-10-052845
McMurtry V, Saavedra JE, Nieves-Alicea R, Simeone AM, Keefer LK, Tari AM (2011) JS-K, a nitric oxide-releasing prodrug, induces breast cancer cell death while sparing normal mammary epithelial cells. Int J Oncol 38(4):963–971. doi:10.3892/ijo.2011.925
Laschak M, Spindler KD, Schrader AJ, Hessenauer A, Streicher W, Schrader M, Cronauer MV (2012) JS-K, a glutathione/glutathione S-transferase-activated nitric oxide releasing prodrug inhibits androgen receptor and WNT-signaling in prostate cancer cells. BMC Cancer 12:130. doi:10.1186/1471-2407-12-130
Qiu M, Chen L, Tan G, Ke L, Zhang S, Chen H, Liu J (2015) A reactive oxygen species activation mechanism contributes to JS-K-induced apoptosis in human bladder cancer cells. Sci Rep 5:15104. doi:10.1038/srep15104
Liu L, Wang D, Wang J, Wang S (2016) The Nitric oxide prodrug JS-K induces Ca(2+)-mediated apoptosis in human hepatocellular carcinoma HepG2 Cells. J Biochem Mol Toxicol 30(4):192–199. doi:10.1002/jbt.21778
Gunzle J, Osterberg N, Saavedra JE, Weyerbrock A (2016) Nitric oxide released from JS-K induces cell death by mitotic catastrophe as part of necrosis in glioblastoma multiforme. Cell Death Dis 7(9):e2349. doi:10.1038/cddis.2016.254
Issaeva N, Bozko P, Enge M, Protopopova M, Verhoef LG, Masucci M, Pramanik A, Selivanova G (2004) Small molecule RITA binds to p53, blocks p53-HDM-2 interaction and activates p53 function in tumors. Nat Med 10(12):1321–1328. doi:10.1038/nm1146
Theodoulou M, Hudis C (2004) Cardiac profiles of liposomal anthracyclines: greater cardiac safety versus conventional Doxorubicin? Cancer 100(10):2052–2063. doi:10.1002/cncr.20207
Wang G, Zhang J, Liu L, Sharma S, Dong Q (2012) Quercetin potentiates Doxorubicin mediated antitumor effects against liver cancer through p53/Bcl-xl. PLoS ONE 7(12):e51764. doi:10.1371/journal.pone.0051764
Green DR (2000) Apoptotic pathways: paper wraps stone blunts scissors. Cell 102(1):1–4
Johnstone RW, Ruefli AA, Lowe SW (2002) Apoptosis: a link between cancer genetics and chemotherapy. Cell 108(2):153–164
Roberts AM, Watson IR, Evans AJ, Foster DA, Irwin MS, Ohh M (2009) Suppression of hypoxia-inducible factor 2alpha restores p53 activity via Hdm2 and reverses chemoresistance of renal carcinoma cells. Can Res 69(23):9056–9064. doi:10.1158/0008-5472.CAN-09-1770
Liu T, Xiong J, Yi S, Zhang H, Zhou S, Gu L, Zhou M (2016) FKBP12 enhances sensitivity to chemotherapy-induced cancer cell apoptosis by inhibiting MDM2. Oncogene. doi:10.1038/onc.2016.331
Walia V, Kakar S, Elble R (2011) Micromanagement of the mitochondrial apoptotic pathway by p53. Front Biosci (Landmark Ed) 16:749–758
Chipuk JE, Green DR (2009) PUMA cooperates with direct activator proteins to promote mitochondrial outer membrane permeabilization and apoptosis. Cell Cycle 8(17):2692–2696. doi:10.4161/cc.8.17.9412
Bulucu F, Ocal R, Karadurmus N, Sahin M, Kenar L, Aydin A, Oktenli C, Koc B, Inal V, Yamanel L, Yaman H (2009) Effects of N-acetylcysteine, deferoxamine and selenium on Doxorubicin-induced hepatotoxicity. Biol Trace Elem Res 132(1–3):184–196. doi:10.1007/s12011-009-8377-y
Liu B, Tan X, Liang J, Wu S, Liu J, Zhang Q, Zhu R (2014) A reduction in reactive oxygen species contributes to dihydromyricetin-induced apoptosis in human hepatocellular carcinoma cells. Sci Rep 4:7041. doi:10.1038/srep07041
Maciag AE, Chakrapani H, Saavedra JE, Morris NL, Holland RJ, Kosak KM, Shami PJ, Anderson LM, Keefer LK (2011) The nitric oxide prodrug JS-K is effective against non-small-cell lung cancer cells in vitro and in vivo: involvement of reactive oxygen species. J Pharm Exp Ther 336(2):313–320. doi:10.1124/jpet.110.174904
Shami PJ, Saavedra JE, Wang LY, Bonifant CL, Diwan BA, Singh SV, Gu Y, Fox SD, Buzard GS, Citro ML, Waterhouse DJ, Davies KM, Ji X, Keefer LK (2003) JS-K, a glutathione/glutathione S-transferase-activated nitric oxide donor of the diazeniumdiolate class with potent antineoplastic activity. Mol Cancer Ther 2(4):409–417
Primeau AJ, Rendon A, Hedley D, Lilge L, Tannock IF (2005) The distribution of the anticancer drug Doxorubicin in relation to blood vessels in solid tumors. Clin Cancer Res 11(24 Pt 1):8782–8788. doi:10.1158/1078-0432.CCR-05-1664
Peng ZH, Kopecek J (2015) Enhancing accumulation and penetration of HPMA copolymer-Doxorubicin conjugates in 2D and 3D prostate cancer cells via iRGD conjugation with an MMP-2 cleavable spacer. J Am Chem Soc 137(21):6726–6729. doi:10.1021/jacs.5b00922
Esparza-Lopez J, Ramos-Elias PA, Castro-Sanchez A, Rocha-Zavaleta L, Escobar-Arriaga E, Zentella-Dehesa A, Leon-Rodriguez E, Medina-Franco H, Ibarra-Sanchez Mde J (2016) Primary breast cancer cell culture yields intra-tumor heterogeneous subpopulations expressing exclusive patterns of receptor tyrosine kinases. BMC Cancer 16(1):740. doi:10.1186/s12885-016-2769-0
Acknowledgements
We sincerely thank other colleagues in our laboratory for their active help in this study. This work was supported by The National Natural Science Funds (No. 81272833) of China.
Funding
This study was funded by The National Natural Science Funds of China (Grant Number 81272833).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Qiu, M., Ke, L., Zhang, S. et al. JS-K, a GST-activated nitric oxide donor prodrug, enhances chemo-sensitivity in renal carcinoma cells and prevents cardiac myocytes toxicity induced by Doxorubicin. Cancer Chemother Pharmacol 80, 275–286 (2017). https://doi.org/10.1007/s00280-017-3359-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-017-3359-9