Abstract
Background
Previous phase III studies raised concern about the safety of the combination of capecitabine and irinotecan in patients with metastatic colorectal cancer (mCRC). We conducted a single arm phase II study to evaluate the safety and efficacy of bevacizumab in combination with dose-reduced capecitabine and irinotecan in patients with previously untreated mCRC.
Patients and methods
Patients with previously untreated mCRC were eligible. Capecitabine was given at 1,000 mg/m2 orally twice daily for 14 days and dose was reduced to 750 mg/m2 for patients over 65. Irinotecan was given at 200 mg/m2 and bevacizumab was given at 7.5 mg/kg intravenously on day 1. The treatment cycle was repeated every 21 days. The primary endpoint was progression-free survival (PFS); secondary endpoints included overall survival, response rate, and toxicity.
Results
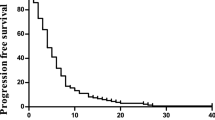
Fifty patients were enrolled, the median age was 58, and 54% were ECOG 0. The most common grade 3/4 adverse events included hand-foot syndrome (14%), neutropenia (12%), and diarrhea (10%). Response rate was 51% and disease control rate (response and stable disease) was 98%. Median PFS was 11.5 months (95% CI: 9.2–13.7), and 6 month PFS was 90% (95% CI: 77–98%).
Conclusion
With modest dose reductions, the combination of capecitabine, irinotecan, and bevacizumab was well tolerated and resulted in favorable outcomes for patients with previously untreated mCRC.


Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T et al (2008) Cancer statistics, 2008. CA Cancer J Clin 58(2):71–96
Ferlay J, Autier P, Boniol M, Heanue M, Colombet M, Boyle P (2007) Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol 18(3):581–592
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W et al (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350(23):2335–2342
Giantonio BJ, Levy DE, O’dwyer PJ, Meropol NJ, Catalano PJ, Benson AB III et al (2006) A phase II study of high-dose bevacizumab in combination with irinotecan, 5-fluorouracil, leucovorin, as initial therapy for advanced colorectal cancer: results from the Eastern Cooperative Oncology Group study E2200. Ann Oncol 17(9):1399–1403
Giantonio BJ, Catalano PJ, Meropol NJ, O’Dwyer PJ, Mitchell EP, Alberts SR et al (2007) Bevacizumab in combination with oxaliplatin, fluorouracil, and leucovorin (FOLFOX4) for previously treated metastatic colorectal cancer: results from the Eastern Cooperative Oncology Group Study E3200. J Clin Oncol 25(12):1539–1544
Saltz LB, Clarke S, Diaz-Rubio E, Scheithauer W, Figer A, Wong R et al (2008) Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol 26(12):2013–2019
Grothey A, Sugrue MM, Purdie DM, Dong W, Sargent D, Hedrick E et al (2008) Bevacizumab beyond first progression is associated with prolonged overall survival in metastatic colorectal cancer: results from a large observational cohort study (BRiTE). J Clin Oncol 26(33):5326–5334
Van Cutsem E, Rivera F, Berry S, Kretzschmar A, Michael M, Dibartolomeo M et al (2009) Safety and efficacy of first-line bevacizumab with FOLFOX, XELOX, FOLFIRI and fluoropyrimidines in metastatic colorectal cancer: the BEAT study. Ann Oncol 20(11):1842–1847
Colucci G, Gebbia V, Paoletti G, Giuliani F, Caruso M, Gebbia N et al (2005) Phase III randomized trial of FOLFIRI versus FOLFOX4 in the treatment of advanced colorectal cancer: a multicenter study of the Gruppo Oncologico Dell’Italia Meridionale. J Clin Oncol 23(22):4866–4875
de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J et al (2000) Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 18(16):2938–2947
Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P et al (2000) Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet 355(9209):1041–1047
Giacchetti S, Perpoint B, Zidani R, Le Bail N, Faggiuolo R, Focan C et al (2000) Phase III multicenter randomized trial of oxaliplatin added to chronomodulated fluorouracil-leucovorin as first-line treatment of metastatic colorectal cancer. J Clin Oncol 18(1):136–147
Kohne CH, van Cutsem E, Wils J, Bokemeyer C, El-Serafi M, Lutz MP et al (2005) Phase III study of weekly high-dose infusional fluorouracil plus folinic acid with or without irinotecan in patients with metastatic colorectal cancer: European Organisation for Research and Treatment of Cancer Gastrointestinal Group Study 40986. J Clin Oncol 23(22):4856–4865
Saltz LB, Cox JV, Blanke C, Rosen LS, Fehrenbacher L, Moore MJ et al (2000) Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. Irinotecan Study Group. N Engl J Med 343(13):905–914
Sorbye H, Glimelius B, Berglund A, Fokstuen T, Tveit KM, Braendengen M et al (2004) Multicenter phase II study of Nordic fluorouracil and folinic acid bolus schedule combined with oxaliplatin as first-line treatment of metastatic colorectal cancer. J Clin Oncol 22(1):31–38
Jonker DJ, O’Callaghan CJ, Karapetis CS, Zalcberg JR, Tu D, Au HJ et al (2007) Cetuximab for the treatment of colorectal cancer. N Engl J Med 357(20):2040–2048
Van Cutsem E, Peeters M, Siena S, Humblet Y, Hendlisz A, Neyns B et al (2007) Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J Clin Oncol 25(13):1658–1664
Cassidy J, Twelves C, Van Cutsem E, Hoff P, Bajetta E, Boyer M et al (2002) First-line oral capecitabine therapy in metastatic colorectal cancer: a favorable safety profile compared with intravenous 5-fluorouracil/leucovorin. Ann Oncol 13(4):566–575
Twelves C, Boyer M, Findlay M, Cassidy J, Weitzel C, Barker C et al (2001) Capecitabine (Xeloda) improves medical resource use compared with 5-fluorouracil plus leucovorin in a phase III trial conducted in patients with advanced colorectal carcinoma. Eur J Cancer 37(5):597–604
Van Cutsem E, Twelves C, Cassidy J, Allman D, Bajetta E, Boyer M et al (2001) Oral capecitabine compared with intravenous fluorouracil plus leucovorin in patients with metastatic colorectal cancer: results of a large phase III study. J Clin Oncol 19(21):4097–4106
Jansman FG, Postma MJ, van Hartskamp D, Willemse PH, Brouwers JR (2004) Cost-benefit analysis of capecitabine versus 5-fluorouracil/leucovorin in the treatment of colorectal cancer in the Netherlands. Clin Ther 26(4):579–589
Czejka M, Schueller J, Hauer K, Ostermann E (2005) Pharmacokinetics and metabolism of irinotecan combined with capecitabine in patients with advanced colorectal cancer. Anticancer Res 25(4):2985–2990
Rea DW, Nortier JW, Ten Bokkel Huinink WW, Falk S, Richel DJ, Maughan T et al (2005) A phase I/II and pharmacokinetic study of irinotecan in combination with capecitabine as first-line therapy for advanced colorectal cancer. Ann Oncol 16(7):1123–1132
Bajetta E, Di Bartolomeo M, Mariani L, Cassata A, Artale S, Frustaci S et al (2004) Randomized multicenter Phase II trial of two different schedules of irinotecan combined with capecitabine as first-line treatment in metastatic colorectal carcinoma. Cancer 100(2):279–287
Borner MM, Bernhard J, Dietrich D, Popescu R, Wernli M, Saletti P et al (2005) A randomized phase II trial of capecitabine and two different schedules of irinotecan in first-line treatment of metastatic colorectal cancer: efficacy, quality-of-life and toxicity. Ann Oncol 16(2):282–288
Cartwright T, Lopez T, Vukelja SJ, Encarnacion C, Boehm KA, Asmar L (2005) Results of a phase II open-label study of capecitabine in combination with irinotecan as first-line treatment for metastatic colorectal cancer. Clin Colorectal Cancer 5(1):50–56
Kim TW, Kang WK, Chang HM, Park JO, Ryoo BY, Ahn JS et al (2005) Multicenter phase II study of oral capecitabine plus irinotecan as first-line chemotherapy in advanced colorectal cancer: a Korean Cancer Study Group trial. Acta Oncol 44(3):230–235
Park SH, Bang SM, Cho EK, Baek JH, Oh JH, Im SA et al (2004) First-line chemotherapy with irinotecan plus capecitabine for advanced colorectal cancer. Oncology 66(5):353–357
Fuchs CS, Marshall J, Mitchell E, Wierzbicki R, Ganju V, Jeffery M et al (2007) Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in first-line treatment of metastatic colorectal cancer: results from the BICC-C study. J Clin Oncol 25(30):4779–4786
Kohne CH, De Greve J, Hartmann JT, Lang I, Vergauwe P, Becker K et al (2008) Irinotecan combined with infusional 5-fluorouracil/folinic acid or capecitabine plus celecoxib or placebo in the first-line treatment of patients with metastatic colorectal cancer. EORTC study 40015. Ann Oncol 19(5):920–926
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92(3):205–216
Garcia-Alfonso P, Munoz-Martin AJ, Alvarez-Suarez S, Jerez-Gilarranz Y, Riesco-Martinez M, Khosravi P et al (2010) Bevacizumab in combination with biweekly capecitabine and irinotecan, as first-line treatment for patients with metastatic colorectal cancer. Br J Cancer 103(10):1524–1528
Ducreux M, Adenis A, Mendiboure E et al (2009) Efficacy and safety of bevacizumab (BEV)—based combination regimens in patients with metastatic colorectal cancer (mCRC): randomized phase II study of BEV+FOLFIRI versus BEV+XELIRI (FNCLCC ACCORD 13/0503 study). J Clin Oncol 27:15s
Sobrero A, Ackland S, Clarke S, Perez-Carrion R, Chiara S, Gapski J et al (2009) Phase IV study of bevacizumab in combination with infusional fluorouracil, leucovorin and irinotecan (FOLFIRI) in first-line metastatic colorectal cancer. Oncology 77(2):113–119
Acknowledgments
Partially funding for this study was provided by Hoffmann-La-Roche Canada.
Conflicts of interest
D. Renouf received honoraria from Roche; S. Welch received honoraria and travel grants from Roche; R. Feld received research funding from Roche; M. Krzyzanowska received honoraria from Sanofi Aventis; E. Chen received research funding from Roche.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Renouf, D.J., Welch, S., Moore, M.J. et al. A phase II study of capecitabine, irinotecan, and bevacizumab in patients with previously untreated metastatic colorectal cancer. Cancer Chemother Pharmacol 69, 1339–1344 (2012). https://doi.org/10.1007/s00280-012-1843-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-012-1843-9