Abstract
Purpose
To assess the efficacy and toxicity of an oral anticancer fluoropyrimidine derivative, S-1, for previously treated patients with advanced non-small cell lung cancer (NSCLC).
Patients and methods
Patients with advanced (clinical stage IIIB-IV) NSCLC who had previously received one platinum-based chemotherapy were enrolled. S-1 was administered orally at the dosage decided by using the nomogram based on patient BSA b.i.d. for 28 consecutive days, repeated every 6 weeks.
Results
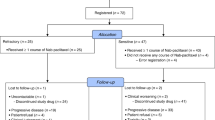
Between August 2005 and July 2007, 50 patients were entered into this study. Six patients achieved partial response (PR), and the overall response rate of eligible patients was 12.5% (6/48) (95% confidence interval (95%CI), 3.1–21.9%). Disease control rate was 39.6% (19/48) (95%CI, 25.7–53.4%). Median progression-free survival was 2.5 months. Median survival time was 8.2 months, and 1-year survival rate was 29.6%. No grade 4 toxicities were encountered. Grade 3 hematological toxicities comprised neutropenia in one patient (2.1%) and anemia in one patient (2.1%). Grade 3 non-hematological toxicities were observed in only five patients (10.4%). Treatment-related death did not occur.
Conclusion
S-1 is an active and well-tolerated monotherapy for second-line treatment of advanced NSCLC.

Similar content being viewed by others
References
Greenlee RT, Murray T, Bolden S, Wingo PA (2000) Cancer statistics, 2000. CA Cancer J Clin 50:7–33
Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, Zhu J, Johnson DH, Eastern Cooperative Oncology Group (2002) Comparison of four chemotherapy regimens for advanced non-small cell lung cancer. N Engl J Med 346(2):92–98
Smit EF, van Meerbeeck JP, Lianes P, Debruyne C, Legrand C, Schramel F, Smit H, Gaafar R, Biesma B, Manegold C, Neymark N, Giaccone G, European Organization for Research, Treatment of Cancer Lung Cancer Group (2003) Three-arm randomized study of two cisplatin-based regimens and paclitaxel plus gemcitabine in advanced non-small cell lung cancer: a phase III trial of the European Organization for Research and Treatment of Cancer Lung Cancer Group, EORTC 08975. J Clin Oncol 21(21):3909–3917
Shepherd FA, Dancey J, Ramlau R, Mattson K, Gralla R, O’Rourke M, Levitan N, Gressot L, Vincent M, Burkes R, Coughlin S, Kim Y, Berille J (2000) Prospective randomized trial of docetaxel versus best supportive care in patients with non-small cell lung cancer previously treated with platinum-based chemotherapy. J Clin Oncol 18(10):2095–2103
Fossella FV, DeVore R, Kerr RN, Crawford J, Natale RR, Dunphy F, Kalman L, Miller V, Lee JS, Moore M, Gandara D, Karp D, Vokes E, Kris M, Kim Y, Gamza F, Hammershaimb L (2000) Randomized phase III trial of docetaxel versus vinorelbine or ifosfamide in patients with advanced non-small cell lung cancer previously treated with platinum-containing chemotherapy regimens: The TAX 320 Non-Small Cell Lung Cancer Study Group. J Clin Oncol 18(12):2354–2362
Hanna N, Shepherd FA, Fossella FV, Pereira JR, De Marinis F, von Pawel J, Gatzemeier U, Tsao TC, Pless M, Muller T, Lim HL, Desch C, Szondy K, Gervais R, Manegold C, Shaharyar, Paul S, Paoletti P, Einhorn L, Bunn PA Jr (2004) Randomized phase III trial of pemetrexed versus docetaxel in patients with non-small cell lung cancer previously treated with chemotherapy. J Clin Oncol 22(9):1589–1597
Shepherd FA, Rodrigues Pereira J, Ciuleanu T, Tan EH, Hirsh V, Thongprasert S, Campos D, Maoleekoonpiroj S, Smylie M, Martins R, van Kooten M, Dediu M, Findlay B, Tu D, Johnston D, Bezjak A, Clark G, Santabárbara P, Seymour L, National Cancer Institute of Canada Clinical Trials Group (2005) Erlotinib in previously treated non-small cell lung cancer. N Engl J Med 353(2):123–132
Shirasaka T, Shimamato Y, Ohshimo H, Yamaguchi M, Kato T, Yonekura K, Fukushima M (1996) Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 7(5):548–557
Shirasaka T, Shimamoto Y, Fukushima M (1993) Inhibition by oxonic acid of gastrointestinal toxicity of 5-fluorouracil without loss of its antitumor activity in rats. Cancer Res 53(17):4004–4009
Kawahara M, Furuse K, Segawa Y, Yoshimori K, Matsui K, Kudoh S, Hasegawa K, Niitani H (2001) Phase II study of S-1, a novel oral fluorouracil, in advanced non-small cell lung cancer. Br J Cancer 85(7):939–943
Rossi D, Catalano V, Alessandroni P, Fedeli A, Fedeli SL, Giordani P, Baldelli AM, Casadei V, Ceccolini M, Ugolini M, Dennetta D, Catalano G (2007) A phase II study of single-agent oral vinorelbine in patients with pretreated advanced non-small cell lung cancer. Clin Lung Cancer 8(6):382–385
Furuse K, Kawahara M, Hasegawa K, Kudoh S, Takada M, Sugiura T, Ichinose Y, Fukuoka M, Ohashi Y, Niitani H (2001) Early phase II study of S-1, a new oral fluoropyrimidine, for advanced non-small cell lung cancer. Int J Clin Oncol 6(5):236–241
Mukohara T, Takeda K, Miyazaki M, Takifuji N, Terakawa K, Negoro S (2001) Japanese experience with second-line chemotherapy with low-dose (60 mg/m2) docetaxel in patients with advanced non-small cell lung cancer. Cancer Chemother Pharmacol 48(5):356–360
Ichinose Y, Yoshimori K, Sakai H, Nakai Y, Sugiura T, Kawahara M, Niitani H (2004) S-1 plus cisplatin combination chemotherapy in patients with advanced non-small cell lung cancer: a multi-institutional phase II trial. Clin Cancer Res 10(23):7860–7864
Ramlau R, Gervais R, Krzakowski M, von Pawel J, Kaukel E, Abratt RP, Dharan B, Grotzinger KM, Ross G, Dane G, Shepherd FA (2006) Phase III study comparing oral topotecan to intravenous docetaxel in patients with pretreated advanced non-small cell lung cancer. J Clin Oncol 24(18):2800–2807 Epub 2006 May 8
Baas P, Szczesna A, Albert I, Milanowski J, Juhász E, Sztancsik Z, von Pawel J, Oyama R, Burgers S (2008) Phase I/II study of a 3 weekly oral taxane (DJ-927) in patients with recurrent, advanced non-small cell lung cancer. J Thorac Oncol 3(7):745–750
Conflict of interest statement
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Totani, Y., Saito, Y., Hayashi, M. et al. A phase II study of S-1 monotherapy as second-line treatment for advanced non-small cell lung cancer. Cancer Chemother Pharmacol 64, 1181–1185 (2009). https://doi.org/10.1007/s00280-009-0981-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-009-0981-1