Abstract
Purpose
The phase II study was conducted to evaluate the efficacy and safety of irinotecan as salvage single-agent chemotherapy in patients with advanced pancreatic cancer.
Methods
Patients with measurable metastatic pancreatic cancer, progressive after previous gemcitabine-based chemotherapy were treated with irinotecan 150 mg/m2 every 2 weeks. Treatment was repeated until disease progression or unacceptable toxicity.
Results
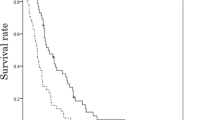
Between March 2004 to February 2007, 33 patients were registered and treated with irinotecan monotherapy. The patients’ median age was 59 years (range 36–70) and two had an ECOG performance status of 2. A total of 167 chemotherapy cycles were delivered (median, 4; range 2–12). In an intent-to-treat analysis, three (9%) confirmed partial response and 13 patients with stable disease were observed for a disease control rate of 48%. The median progression-free and overall survivals were 2.0 months (95% CI, 0.7–3.3) and 6.6 months (95% CI, 5.8–7.4), respectively. Toxic effects were mainly gastrointestinal (nausea in 64% of patients, diarrhea in 36%), Toxicity profiles were generally predictable and manageable, and there was no treatment-related death.
Conclusions
Second-line chemotherapy with single-agent irinotecan is marginally effective and well tolerated regimen for gemcitabine-pretreated patients with advanced pancreatic cancer.

Similar content being viewed by others
References
Bae JM, Won YJ, Jung KW, Park JG (2002) Annual report of the Korean central cancer registry program 2000. Cancer Res Treat 34:77–83
Bissery MC, Vrignaud P, Lavelle F, Chabot GG (1996) Experimental antitumor activity and pharmacokinetics of the camptothecin analog irinotecan (CPT-11) in mice. Anticancer Drugs 7:437–460
Boeck S, Heinemann V (2008) Second-line therapy in gemcitabine-pretreated patients with advanced pancreatic cancer. J Clin Oncol 26:1178–1179 author reply 1179
Burris HA III, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, Cripps MC, Portenoy RK, Storniolo AM, Tarassoff P, Nelson R, Dorr FA, Stephens CD, Von Hoff DD (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15:2403–2413
Creemers GJ, Lund B, Verweij J (1994) Topoisomerase I inhibitors: topotecan and irenotecan. Cancer Treat Rev 20:73–96
Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J, Alakl M, Gruia G, Awad L, Rougier P (2000) Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet 355:1041–1047
Ducreux M, Boige V, Malka D (2007) Treatment of advanced pancreatic cancer. Semin Oncol 34:S25–S30
Fleming TR (1982) One-sample multiple testing procedure for phase II clinical trials. Biometrics 38:143–151
Kang SP, Saif MW (2008) Optimal second line treatment options for gemcitabine refractory advanced pancreatic cancer patients. Can we establish standard of care with available data? JOP 9:83–90
Kelly DM, Benjamin IS (1995) Pancreatic carcinoma. Ann Oncol 6:19–28
Klapdor R, Fenner C (2000) Irinotecan(Campto R): efficacy as third/forth line therapy in advanced pancreatic cancer. Anticancer Res 20:5209–5212
Rocha Lima CM, Green MR, Rotche R, Miller WH Jr, Jeffrey GM, Cisar LA, Morganti A, Orlando N, Gruia G, Miller LL (2004) Irinotecan plus gemcitabine results in no survival advantage compared with gemcitabine monotherapy in patients with locally advanced or metastatic pancreatic cancer despite increased tumor response rate. J Clin Oncol 22:3776–3783
Saltz LB, Cox JV, Blanke C, Rosen LS, Fehrenbacher L, Moore MJ, Maroun JA, Ackland SP, Locker PK, Pirotta N, Elfring GL, Miller LL (2000) Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. Irinotecan Study Group. N Engl J Med 343:905–914
Stathopoulos GP, Syrigos K, Aravantinos G, Polyzos A, Papakotoulas P, Fountzilas G, Potamianou A, Ziras N, Boukovinas J, Varthalitis J, Androulakis N, Kotsakis A, Samonis G, Georgoulias V (2006) A multicenter phase III trial comparing irinotecan-gemcitabine (IG) with gemcitabine (G) monotherapy as first-line treatment in patients with locally advanced or metastatic pancreatic cancer. Br J Cancer 95:587–592
Taieb J, Lecomte T, Aparicio T, Asnacios A, Mansourbakht T, Artru P, Fallik D, Spano JP, Landi B, Lledo G, Desrame J (2007) FOLFIRI.3, a new regimen combining 5-fluorouracil, folinic acid and irinotecan, for advanced pancreatic cancer: results of an Association des Gastro-Enterologues Oncologues (Gastroenterologist Oncologist Association) multicenter phase II study. Ann Oncol 18:498–503
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Ueno H, Okusaka T, Funakoshi A, Ishii H, Yamao K, Ishikawa O, Ohkawa S, Saitoh S (2007) A phase II study of weekly irinotecan as first-line therapy for patients with metastatic pancreatic cancer. Cancer Chemother Pharmacol 59:447–454
Ulrich-Pur H, Raderer M, Verena Kornek G, Schull B, Schmid K, Haider K, Kwasny W, Depisch D, Schneeweiss B, Lang F, Scheithauer W (2003) Irinotecan plus raltitrexed vs. raltitrexed alone in patients with gemcitabine-pretreated advanced pancreatic adenocarcinoma. Br J Cancer 88:1180–1184
Wagener DJ, Verdonk HE, Dirix LY, Catimel G, Siegenthaler P, Buitenhuis M, Mathieu-Boue A, Verweij J (1995) Phase II trial of CPT-11 in patients with advanced pancreatic cancer, an EORTC early clinical trials group study. Ann Oncol 6:129–132
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yi, S.Y., Park, Y.S., Kim, H.S. et al. Irinotecan monotherapy as second-line treatment in advanced pancreatic cancer. Cancer Chemother Pharmacol 63, 1141–1145 (2009). https://doi.org/10.1007/s00280-008-0839-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-008-0839-y