Abstract
Purpose
Our objective was to assess the efficacy and toxicity of concurrent chemoradiotherapy with cisplatin + weekly divided-dose docetaxel in patients with stage III non-small-cell lung cancer (NSCLC).
Methods
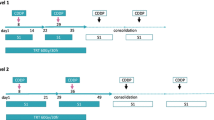
A total of 34 patients aged less than 75 years old with locally advanced stage III NSCLC were enrolled. The patients received intravenous infusions of cisplatin (80 mg/m2; day 1) and docetaxel (20 mg/m2; days 1, 8, 15), followed by a week’s drug-free interval. Standard concurrent thoracic radiotherapy was given for 6 weeks (2 Gy per fraction; total dose, 60 Gy).
Results
Over Grade 3 neutropenia, esophagitis and pulmonary toxicities were observed in 23.5, 17.6 and 11.8% of the cases, respectively. One complete response and 20 partial responses were obtained, with an objective response rate of 61.8%. The median survival time was 26.4 months (95% CI 16.9—not reached) and the 1- and 3-year survival rates were 76.5 and 41.2%, respectively.
Conclusion
Cisplatin + weekly docetaxel with concurrent radiotherapy is a feasible and effective regimen for locally advanced NSCLC.


Similar content being viewed by others
References
Dillman RO, Herndon J, Seagren SL et al (1996) Improved survival in stage III non-small-cell lung cancer: seven-year follow-up of cancer and leukemia group B (CALGB) 8433 trial. J Natl Cancer Inst 88:1210–1215
Sause W, Kolesar P, Taylor SIV et al (2000) Final results of phase III trial in regionally advanced unresectable non-small cell lung cancer: Radiation Therapy Oncology Group, Eastern Cooperative Oncology Group, and Southwest Oncology Group. Chest 117:358–364
Belani CP, Choy H, Bonomi P et al (2005) Combined chemoradiotherapy regimens of paclitaxel and carboplatin for locally advanced non-small-cell lung cancer: a randomized phase II locally advanced multi-modality protocol. J Clin Oncol 23:5883–5891
Furuse K, Fukuoka M, Kawahara M et al (1999) Phase III study of concurrent versus sequential thoracic radiotherapy in combination with mitomycin, vindesine, and cisplatin in unresectable stage III non-small-cell lung cancer. J Clin Oncol 17:2692–2699
Curran D, Scott C, Langer C et al (2003) Long-term benefit is observed in a phase III comparison of sequential versus concurrent chemo-radiotherapy for patients with unresected stage III NSCLC: RTOG 9410. Proc Am Soc Clin Oncol 22:621
Fournel P, Robinet G, Thomas P et al (2005) Randomized phase III trial of sequential chemoradiotherapy compared with concurrent chemoradiotherapy in locally advanced non-small-cell lung cancer: Groupe Lyon-Saint-Etienne d’Oncologie Thoracique-Groupe Français de Pneumo-Cancérologie NPC 95–01 Study. J Clin Oncol 23:5910–5917
Kunitoh H, Watanabe K, Furuse K et al (1996) Phase II trial of Decetaxel in previously untreated advanced non-small–cell lung cancer: a Japanese Cooperative study. J Clin Oncol 14:1649–1655
Schiller JH, Harrington D, Belani CP et al (2002) Eastern Cooperative Oncology Group. For the Eastern Cooperative Oncology Group. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 346:92–98
Kubota K, Watanabe K, Kunitoh H et al (2004) Phase III randomized trial of docetaxel plus cisplatin versus vindesine plus cisplatin in patients with stage IV non-small-cell lung cancer: the Japanese Taxotere Lung Cancer Study Group. J Clin Oncol 22:254–261
Choy H, Rodriguez S, Koester S et al (1993) Synergic effects of Taxol/Taxotere on radiation sensitivity on human tumor cell lines. Int J Radiat Oncol Biol Phys 24:274–275
Mason KA, Hunter NR, Milas M et al (1997) Docetaxel enhances tumor radioresponse in vivo. Clin Cancer Res 3(12 Pt 1):2431–2438
Karasawa K, Katsui K, Seki K et al (2003) Radiotherapy with concurrent docetaxel for advanced and recurrent breast cancer. Breast Cancer 10:268–274
Koukourakis MI, Kourousis C, Kamilaki M et al (1998) Weekly docetaxel and concomitant boost radiotherapy for non-small cell lung cancer. A phase I/II dose escalation. Eur J Cancer 34:838–844
Onishi H, Kuriyama K, Yamaguchi M et al (2003) Concurrent two-dimensional radiotherapy and weekly docetaxel in the treatment of stage III non-small cell lung cancer: a good local response but no gut survival due to radiation pneumonitis. Lung Cancer 40:79–84
Koizumi T, Tsunoda T, Fujimoto K et al (2001) Phase I trial of weekly docetaxel combined with cisplatin in patients with non-small-cell lung cancer. Lung Cancer 34:125–131
Tsunoda T, Koizumi T, Hayasaka M et al (2004) Phase II study of weekly docetaxel combined with cisplatin in patients with advanced non-small-cell lung cancer. Cancer Chemother Pharmacol 54:173–177
Alvarez MV, Cobreros G, Heras A et al (1978) Studies on cis-dichlorodiammineplatinum (II) as a radiosensitizer. Br J Cancer 3:68–72
Dewit L (1987) Combined treatment of radiation and cisdiamminedichloroplatinum (II): a review of experimental and clinical data. Int J Radiat Oncol Biol Phys 13:403–426
Oken MM, Creech RH, Tormey DC et al (1982) Toxicity and Response Criteria Of The Eastern Cooperative Oncology Group. Am J Clin Oncol 5:649–655
National Cancer Institute: Common Toxicity Criteria (CTC) v 2.0. http://ctep.cancer.gov/reporting/ctc.html
Miller AB, Hoogstraten B, Staquet M et al (1981) Reporting results of cancer treatment. Cancer 47:207–214
Yamamoto N, Nishimura Y, Nakagawa K et al (2006) Phase I/II study of weekly docetaxel dose escalation in combination with fixed weekly cisplatin and concurrent thoracic radiotherapy in locally advanced non-small cell lung cancer. Cancer Chemother Pharmacol 58:285–291
Wu HG, Bang YJ, Choi EK et al (2002) Phase I study of weekly docetaxel and cisplatin concurrent with thoracic radiotherapy in Stage III non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 52:75–80
Mudad R, Ramsey M, Kovitz K et al (2003) Concomitant weekly docetaxel, cisplatin and radiotherapy in locally advanced non-small cell lung cancer: a dose finding study. Lung Cancer 39:173–177
Kiura K, Ueoka H, Segawa Y et al (2003) Phase I/II study of docetaxel and cisplatin with concurrent thoracic radiation therapy for locally advanced non-small-cell lung cancer. Br J Cancer 89:795–802
Iwasaki Y, Ohsugi S, Natsuhara A et al (2006) Phase I/II trial of biweekly docetaxel and cisplatin with concurrent thoracic radiation for stage III non-small-cell lung cancer. Cancer Chemother Pharmacol 58:735–741
Albain KS, Crowley JJ, Turrisi AT 3rd et al (2002) A Concurrent cisplatin, etoposide, and chest radiotherapy in pathologic stage IIIB non-small-cell lung cancer: a Southwest Oncology Group phase II study, SWOG 9019. J Clin Oncol 20:3454–3460
Robert F, Spencer SA, Childs HA 3rd et al (2002) Concurrent chemoradiation therapy with cisplatin and paclitaxel for locally advanced non-small cell lung cancer: long-term follow-up of a phase I trial. Lung Cancer 37:189–199
Hainsworth JD, Burris HA 3rd, Erland JB et al (1998) Phase I trial of docetaxel administered by weekly infusion in patients with advanced refractory cancer. J Clin Oncol 16:2164–2168
Ohe Y, Niho S, Kakinuma R et al (2001) Phase I studies of cisplatin and docetaxel administration by three consecutive weekly infusions for advanced non-small cell lung cancer in elderly and non-elderly patients. Jpn J Clin Oncol 31:100–106
Socinski MA, Zhang C, Herndon JE 2nd et al (2004) Combined modality trials of the Cancer and Leukemia Group B in stage III non-small-cell lung cancer: analysis of factors influencing survival and toxicity. Ann Oncol 15:1033–1041
Hirose T, Mizutani Y, Ohmori T et al (2006) The combination of cisplatin and vinorelbine with concurrent thoracic radiation therapy for locally advanced stage IIIA or IIIB non-small-cell lung cancer. Cancer Chemother Pharmacol 58:361–367
Sakai H, Yoneda S, Kobayashi K et al (2004) Phase II study of bi-weekly docetaxel and carboplatin with concurrent thoracic radiation therapy followed by consolidation chemotherapy with docetaxel plus carboplatin for stage III unresectable non-small cell lung cancer. Lung Cancer 43:195–201
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakamura, M., Koizumi, T., Hayasaka, M. et al. Cisplatin and weekly docetaxel with concurrent thoracic radiotherapy for locally advanced stage III non-small-cell lung cancer. Cancer Chemother Pharmacol 63, 1091–1096 (2009). https://doi.org/10.1007/s00280-008-0837-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-008-0837-0