Abstract
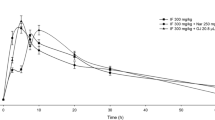
The flavonol monohydroxyethylrutoside (monoHER) has demonstrated protection against doxorubicin-induced cardiotoxicity in in vitro and in vivo studies without affecting the antitumor effect. In the present phase I study, the possible side effects and the pharmacokinetics of monoHER were evaluated in healthy volunteers with the aim to develop a safe and feasible dose to be evaluated in cancer patients treated with doxorubicin. The study was performed as a single blind, randomized trial in healthy volunteers (age between 19 and 56 years). At each dose level, six subjects received monoHER and three placebo. MonoHER was solubilized in 100 ml dextrose 5% and administered as an i.v. infusion in 10 min. The placebo consisted of 100 ml dextrose 5%. The starting dose of monoHER was 100 mg/m2. Dose escalation by 100% of the preceding dose took place after finishing each dose level until the protecting pharmacokinetic values for C max and AUC∞ (as observed in mice after 500 mg/kg monoHER i.p.) were reached and/or serious side effects were observed. The dose was escalated up to 1,500 mg/m2. The mean values of C max and AUC∞ were 360±69.3 μM and 6.8±2.1 μmol min/ml, respectively. These values were comparable to the C max and AUC∞ observed under the protecting conditions in mice. No serious side effects occurred during the entire study. Thus, 1,500 mg/m2 is a feasible and safe dose to be evaluated in a phase II study to investigate the protective properties of monoHER against doxorubicin-induced cardiotoxicity in cancer patients.





Similar content being viewed by others
References
Abou El Hassan MA, Kedde MA, Bast A et al (2001) High-performance liquid chromatography with electrochemical detection for the determination of 7-monohydroxyethylrutoside in plasma. J Chromatogr B Biomed Sci Appl 752:115–121
Abou El Hassan MA, Touw DJ, Wilhelm AJ et al (2000) Stability of monoHER in an aqueous formulation for i.v. administration. Int J Pharm 211:51–56
Abou El Hassan M, Kedde MA, Bast A et al (2003) Bioavailability and pharmacokinetics of the cardioprotecting flavonoid monohydroxyethylrutoside in mice. Cancer Chemother Pharmacol 52:371–376
Anonymous (1995) Dexrazoxane for cardiac protection against doxorubicin. Med Lett 37:110–110
Balant LP, Wermeille M, Griffiths LA (1984) Metabolism and pharmacokinetics of hydroxyethylated rutosides in animals and man. Rev Drug Metab Drug Interact 5:1–24
Barrow A, Griffiths LA (1974) Metabolism of the hydroxyethylrutosides II. Excretion and metabolism of 3’, 4’, 7-tri-o-(β−hydroxyethyl)rutoside and related compounds in laboratory animals after parenteral administration. Xenobiotica 4:1–16
Bielack SS, Erttmann R, Winkler K et al (1989) Doxorubicin: effect of different schedules on toxicity and anti-tumor efficacy. Eur J Cancer Clin Oncol 25:873–882
Bravo L (1998) Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutr Rev 56:317–333
Blum RH, Carter SK (1974) Adriamycin, a new anticancer drug with significant clinical activity. Ann Intern Med 80:249–259
Busby MG, Jeffcoat AR, Bloedon LT et al (2002) Clinical characteristics and pharmacokinetics of purified soy isoflavones: single-dose administration to healthy man. Am J Clin Nutr 75:126–136
De Forni M, Chabot GG, Armand J-P et al (1995) Phase I and pharmacology study of flavone acetic acid administered two or three times weekly without alkalinization. Cancer Chemother Pharmacol 35:219–224
Ferry DR, Smith A, Malkhandi J et al (1996) Phase I clinical trial of the flavonoid quercetin: pharmacokinetics and evidence for in vivo tyrosine kinase inhibition. Clin Cancer Res 2:659–668
Graefe E, Derendorf H, Velt M (1999) Pharmacokinetics and bioavailability of the flavonol quercetin in humans. Int J Clin Pharmacol Ther 37:219–233
Griffiths LA, Hackett AM (1978) Hepatic clearance and disposition of hydroxyethylrutosides. Arch Toxicol (suppl 1):243–246
Gugler R, Leschik M, Dengler HJ (1975) Disposition of quercetin in man after single oral and intravenous doses. Eur J Clin Pharmacol 9:229–234
Hackett AM, Griffiths L (1977) The disposition and metabolism of 3’,4’,7-tri-o-(β−hydroxyethyl)rutoside and 7-mono-o-(β-hydroxyethyl)rutoside in the mouse. Xenobiotica 7:641–651
Hackett AM, Griffiths LA (1979) The metabolism and excretion of 7-mono-O-(β-hydroxyethyl)rutoside in the dog. Eur J Drug Metab Pharmacokinet 4:207–212
Havlin KA, Kuhn JG, Craig JB et al (1991) Phase I clinical and pharmacokinetic trial of flavone acetic acid. J Natl Cancer Inst 83:124–128
Hensley ML, Schuchter LM, Lindley C et al (1999) American society of clinical practice guidelines for the use of chemotherapy and radiotherapy protectants. J Clin Oncol 17:3333–3355
Horenstein MS, Vander Heide RS, L’Ecuyer TJ (2000) Molecular basis of anthracycline-induced cardiotoxicity and its prevention. Mol Genet Metab 71:436–444
Hortobágyi GN (1997) Anthracyclines in the treatment of cancer. Drugs 54:17
Hrdina R, Geršl V, Klimtová I et al (2000) Anthracycline-induced cardiotoxicity. Acta Med 43:75–82
Iarussi D, Indolfi P, Galderisi M et al (2000) Cardiac toxicity after anthracycline chemotherapy in childhood. Herz 25:676–688
Koeller JM, Earhart RH, Davis HL (1981) Phase I trial of ICRF-187 by 48-hour continuous infusion. Cancer Treat Rep 65:459–463
Mross K, Van der Vijgh WJF, Gall H et al (1988) Pharmacokinetics and metabolism of epidoxorubicin and doxorubicin in humans. J Clin Oncol 6:517–526
Neumann HAM, Carlsson K, Brom GHM (1992) Uptake and localisation of O-(B-hydroxyethyl)-rutosides in the venous wall, measured by laser scanning microscopy. Eur J Clin Pharmacol 43:423–426
Pai VB, Nahata MC (2000) Cardiotoxicity of chemotherapeutic agents: incidence, treatment and prevention. Drug Saf 22:263–302
Scalbert A, Williamson G (2000) Dietary intake and bioavailability of polyphenols. J Nutr 130:2073–2085
Setchell KDR, Brown NM, Desai P et al (2001) Bioavailability of pure isoflavones in healthy humans and analysis of commercial soy isoflavone supplements. J Nutr 131:1362S–1375S
Van Acker FAA, Van Acker SABE, Kramer K et al (2000) 7-Monohydroxyethylrutoside protects against chronic doxorubicin-induced cardiotoxicity when administered only once per week. Clin Cancer Res 6:1337–1341
Van Acker SABE, Boven E, Kuiper K et al (1997) Monohydroxyethylrutoside, a dose-dependant cardioprotective agent, does not affect the antitumor activity of doxorubicin. Clin Cancer Res 3:1747–1754
Van Acker SABE, Husken BCP, Toward R et al (1993) The protective effect of Venoruton and constituents on acute doxorubicin-induced cardiotoxicity. Phlebology (suppl 1):31–32
Van Acker SABE, Kramer K, Grimbergen JA et al (1995) Monohydroxyethylrutoside as protector against chronic doxorubicin-induced cardiotoxicity. Br J Pharmacol 115:1260–1264
Van Acker SABE, Voest EE, Beems RB et al (1993) Cardioprotective properties of O-(-hydroxyethyl) rutosides in doxorubicin-pretreated BALB/c mice. Cancer Res 53:4603–4607
Weijl NI, Cleton FJ, Osanto S (1997) Free radicals and antioxidants in chemotherapy-induced toxicity. Cancer Treat Rev 23:209–240
Young RC, Ozols RF, Myers CE (1981) The anthracycline antineoplastic drugs. N Engl J Med 305:139–153
Acknowledgments
We wish to thank the staff of the Department of Hospital Pharmacy, Vrije Universiteit Medical Center, Amsterdam, The Netherlands, for their assistance. This work was supported by a grant-in-aid from the Koningin Wilhelmina Foundation, Amsterdam, The Netherlands.
Author information
Authors and Affiliations
Additional information
Preliminary results of this study have been presented as a poster at AACR (Proc. AACR 43 (2002) 2751).
Rights and permissions
About this article
Cite this article
Willems, A.M., Bruynzeel, A.M., Kedde, M.A. et al. A phase I study of monohydroxyethylrutoside in healthy volunteers. Cancer Chemother Pharmacol 57, 678–684 (2006). https://doi.org/10.1007/s00280-005-0083-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-005-0083-7