Abstract
Purpose
To determine the oral bioavailability of R-XK469, a water-soluble investigational anticancer agent undergoing phase I clinical trials as an intravenous product.
Methods
R-XK469 was administered to two groups of catheterized Sprague-Dawley rats via the oral and IV routes at a dose of 10 mg/kg and blood samples were collected at predetermined times. XK469 in plasma samples was quantified using a HPLC method. The pharmacokinetic parameters were computed using WinNonlin 4.0.1 software.
Results
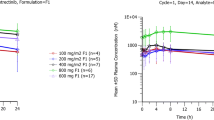
The pharmacokinetic parameters of XK469 following oral and IV administrations, respectively, were (mean±SD): Cmax 138±64 and 404±355 μg/ml; AUC0–∞ 2381±773 and 2854±1924 μg h/ml; and elimination half-life (T1/2) 12.9±5.8 and 13.5±7.8 h Tmax was 2.92±1.92 h following oral dosing. Oral R-XK469 was 83% bioavailable.
Conclusion
Together with the antitumor efficacy of oral XK469 shown in preclinical models and its schedule dependency, these results indicate the promise of developing an oral dosage form of R-XK469 for clinical development.

Similar content being viewed by others
References
Chan KK, Jiang C, Zheng HH, Chiu MH, Joseph M (1999) Proceedings of the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics, abstract 546. Washington, DC
Chan KK, Zheng HH, Zhang Y, Swaan P, Joseph M (2002) Proceedings of the Annual Meeting of AACR, Abstract 2127. San Francisco
Corbett TH, LoRusso P, Demchick L, Simpson C, Pugh S, White K, Kushner J, Polin L, Meyer J, Czarnecki J, Heilbrun L, Horwitz JP, Gross JL, Behrens CH, Harrison BA, McRipley RJ, Trainor G (1998) Preclinical antitumor efficacy of analogs of XK469: sodium-(2-[4-(7-chloro-2-quinoxalinyloxy)phenoxy]propionate. Invest New Drugs 16:129–139
LoRusso PM, Parchment R, Demchik L, Knight J, Polin L, Dzubow J, Behrens C, Harrison B, Trainor G, Corbett TH (1998) Preclinical antitumor activity of XK469 (NSC 656889). Invest New Drugs 16:287–296
Parchment R, Wiegand RA, Kassab JT, Boinpally RR (2003) Proc ASCO 22:136 (abstract 543)
Polin L, White K, Kushner J, Paluch J, Simpson C, Pugh S, Edelstein MK, Hazeldine S, Fontana J, LoRusso P, Horwitz JP, Corbett TH (2002) Preclinical efficacy evaluations of XK-469: dose schedule, route and cross-resistance behavior in tumor bearing mice. Invest New Drugs 20:13–22
Undevia SD, Innocenti F, Sprague E, Schilsky RL, Skoog L, Kindler HL, Fleming GF, Ramirez J, Ratain MJ (2003) Proc ASCO 22:136 (abstract 546)
Wiegand RA, Doyle TD, Grieshaber CK, LoRusso PM, Kassab J, Jasti BR, Parchment RE (1999) In Proceedings of the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics, abstract 542. Washington, DC
Zheng H, Jiang C, Chiu MH, Covey JM, Chan KK (2002) Chiral pharmacokinetics and inversion of enantiomers of a new quinoxaline topoisomerase IIbeta poison in the rat. Drug Metab Dispos 30:344–348
Acknowledgements
This study was supported by NIH grant UO1-CA62487 and the Pharmacology Core, Meyer L. Prentis Comprehensive Cancer Center of Metropolitan Detroit. The authors wish to thank the Pharmaceutical Resources Branch of the NCI for supplying XK469, Drs. Joseph Tomaszewski and Joe Covey of the Toxicology and Pharmacology Branch, and Dr. Vishnuvajjala Rao of the Pharmaceutical Resources Branch of the NCI for helpful advice and fruitful discussions. We thank Dr. Elizabeth Dawe and Debra Wilson of DLAR for taking care of the animals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boinpally, R.R., Zhou, SL., LoRusso, P.M. et al. Bioavailability and pharmacokinetics of the investigational anticancer agent XK469 (NSC 698215) in rats following oral and intravenous administration. Cancer Chemother Pharmacol 55, 404–407 (2005). https://doi.org/10.1007/s00280-004-0862-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-004-0862-6