Abstract
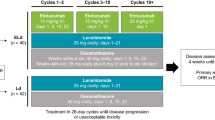
Elotuzumab-based regimens are sometimes selected for multiple myeloma treatment after daratumumab-based regimens. However, there has been insufficient discussion on the efficacy of elotuzumab after daratumumab. We used Kansai Myeloma Forum registration data in a multicenter retrospective evaluation of the efficacy of elotuzumab after daratumumab. Overall survival (OS) rate and time to next treatment (TTNT) were significantly worse in the cohort given elotuzumab after daratumumab (Dara cohort, n = 47) than in the cohort with no history of daratumumab administration before elotuzumab (No-Dara cohort, n = 80, OS: P = 0.03; TTNT: P = 0.02; best response: P < 0.01). In the Dara cohort, OS and TTNT rates were worse with sequential elotuzumab use after daratumumab than with non-sequential (OS: P = 0.02; TTNT: P = 0.03). In patients given elotuzumab < 180 days after daratumumab, OS (P = 0.08) and best response (P = 0.21) tended to be worse, and TTNT was significantly worse (P = 0.01), than in those given elotuzumab after ≥ 180 days. These findings were confirmed by subgroup analyses and multivariate analyses. Monoclonal-antibody-free treatment might be preferable after daratumumab-based regimens. If possible, elotuzumab-based regimens should be considered only ≥ 180 days after daratumumab use.




Similar content being viewed by others
Data availability
The data of this study are available from the first and corresponding author, NN, upon reasonable request.
References
F E Davies. C Pawlyn, S Z Usmani, et al. (2022) Perspective on the Risk-Stratified Treatment of Multiple Myeloma. Blood Cancer Discov. OF1-OF12. https://doi.org/10.1158/2643-3230.BCD-21-0205.
Anderson KC (2016) Progress and Paradigms in Multiple Myeloma. Clin Cancer Res 22(22):5419–5427
Hagen P, Zhang J, Barton K (2022) High-risk disease in newly diagnosed multiple myeloma: beyond the R-ISS and IMWG definitions. Blood Cancer J 12(5):83
Hosoya H, Sidana S (2021) Antibody-Based Treatment Approaches in Multiple Myeloma. Curr Hematol Malig Rep 16(2):183–191
Einsele H, Schreder M (2016) Treatment of multiple myeloma with the immunostimulatory SLAMF7 antibody elotuzumab. Ther Adv Hematol 7(5):288–301
Abramson HN (2021) Immunotherapy of Multiple Myeloma: Promise and Challenges. Immunotargets Ther 10:343–371
Panaampon J, Kariya R, Okada S (2022) Elotuzumab, a potential therapeutic humanized anti-SLAMF7 monoclonal antibody, enhances natural killer cell-mediated killing of primary effusion lymphoma cells. Cancer Immunol Immunother. https://doi.org/10.1007/s00262-022-03177-6
Pazina T, James AM, Colby KB et al (2019) Enhanced SLAMF7 Homotypic Interactions by Elotuzumab Improves NK Cell Killing of Multiple Myeloma. Cancer Immunol Res 7(10):1633–1646
Campbell KS, Cohen AD, Pazina T (2018) Mechanisms of NK Cell Activation and Clinical Activity of the Therapeutic SLAMF7 Antibody. Elotuzumab in Multiple Myeloma Front Immunol 9:25551. https://doi.org/10.3389/fimmu.2018.02551
Pazina T, James AM, MacFarlane AW 4th et al (2017) The anti-SLAMF7 antibody elotuzumab mediates NK cell activation through both CD16-dependent and independent mechanisms. Oncoimmunology 6(9):e1339853. https://doi.org/10.1080/2162402X.2017.1339853
Lonial S, Dimopoulos M, Palumbo A et al (2015) Elotuzumab Therapy for Relapsed or Refractory Multiple Myeloma. N Engl J Med 373(7):621–631
Bigley AB, Spade S, Agha NH et al (2021) FcεRIγ-negative NK cells persist in vivo and enhance efficacy of therapeutic monoclonal antibodies in multiple myeloma. Blood Adv 5(15):3021–3031
Cho H, Kim KH, Lee H et al (2021) Adaptive Natural Killer Cells Facilitate Effector Functions of Daratumumab in Multiple Myeloma. Clin Cancer Res 27(10):2947–2958
Bruzzese A, Derudas D, Galli M et al (2022) Elotuzumab plus lenalidomide and dexamethasone in relapsed/refractory multiple myeloma: Extended 3-year follow-up of a multicenter, retrospective clinical experience with 319 cases outside of controlled clinical trials. Hematol Oncol. https://doi.org/10.1002/hon.3031
Kumar S, Paiva B, Anderson KC et al (2016) International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol 17(8):e328-346
Greipp PR, San Miguel J, Durie BG et al (2005) International staging system for multiple myeloma. J Clin Oncol 23(15):3412–3420
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 48(3):452–458
Palumbo A, Chanan-Khan A, Weisel K et al (2016) Daratumumab, Bortezomib, and Dexamethasone for Multiple Myeloma. N Engl J Med 375(8):754–766
Moreau P, Hulin C, Perrot A et al (2021) Maintenance with daratumumab or observation following treatment with bortezomib, thalidomide, and dexamethasone with or without daratumumab and autologous stem-cell transplant in patients with newly diagnosed multiple myeloma (CASSIOPEIA): an open-label, randomized, phase 3 trial. Lancet Oncol 22(10):1378–1390
Mateos MV, Dimopoulos MA, Cavo M et al (2018) Daratumumab plus Bortezomib, Melphalan, and Prednisone for Untreated Myeloma. N Engl J Med 378(6):518–528
Saltarella I, Desantis V, Melaccio A et al (2020) Mechanisms of Resistance to Anti-CD38 Daratumumab in Multiple Myeloma. Cells 9(1):167
van de Donk NW, Usmani SZ (2018) CD38 Antibodies in Multiple Myeloma: Mechanisms of Action and Modes of Resistance. Front Immunol 9:2134. https://doi.org/10.3389/fimmu.2018.02134
Sanchez L, Wang Y, Siegel DS et al (2016) Daratumumab: a first-in-class CD38 monoclonal antibody for the treatment of multiple myeloma. J Hematol Oncol 9(1):51
van der Veer MS, de Weers M, van Kessel B et al (2011) Towards effective immunotherapy of myeloma: enhanced elimination of myeloma cells by combination of lenalidomide with the human CD38 monoclonal antibody daratumumab. Haematologica 96(2):284–290
Martin TG, Corzo K, Chiron M et al (2019) Therapeutic Opportunities with Pharmacological Inhibition of CD38 with Isatuximab. Cells 8(12):1522
Dimopoulos M, Bringhen S, Anttila P et al (2021) Isatuximab as monotherapy and combined with dexamethasone in patients with relapsed/refractory multiple myeloma. Blood 137(9):1154–1165
Mikhael J, Belhadj-Merzoug K, Hulin C, Vincent L, Moreau P et al (2021) A phase 2 study of isatuximab monotherapy in patients with multiple myeloma who are refractory to daratumumab. Blood Cancer J 11(5):89
Lejeune M, Duray E, Peipp M et al (2021) Balancing the CD38 Expression on Effector and Target Cells in Daratumumab-Mediated NK Cell ADCC against Multiple Myeloma. Cancers 13(12):3072
Kararoudi MN, Nagai Y, Elmas E et al (2020) CD38 deletion of human primary NK cells eliminates daratumumab-induced fratricide and boosts their effector activity. Blood 136(21):2416–2427
Shimazu Y, Kanda J, Kosugi S et al (2023) Efficacy of elotuzumab for multiple myeloma in reference to lymphocyte counts and kappa/lambda ratio or B2 microglobulin. Sci Rep 13(1):5159
Garajales-Cruz AF, et al. (2019) Elotuzumab in Combination with an Immunomodulatory Agent (IMID) in Patients with Multiple Myeloma Refractory to Imids and/or Daratumumab: A Single-Institution Analysis. Blood 2019 Nov; 134(Supplement_1): 3183.
Robak P, Drozdz I, Szemraj J et al (2018) Drug resistance in multiple myeloma. Cancer Treat Rev 70:199–208
Nijhof IS, Casneuf T, van Velzen J et al (2016) CD38 expression and complement inhibitors affect response and resistance to daratumumab therapy in myeloma. Blood 128(7):959–970
Pick M, Vainstein V, Goldschmidt N et al (2018) Daratumumab resistance is frequent in advanced stage multiple myeloma patients irrespective of CD38 expression and is related to dismal prognosis. Eur J Haematol 100(5):494–501
Casneuf T, Steven X, Adams HC 3rd (2017) Effects of daratumumab on natural killer cells and impact on clinical outcomes in relapsed or refractory multiple myeloma. Blood Adv 1(23):2105–2114
Canioni D, Salles G, Mounier N et al (2008) High numbers of tumor-associated macrophages have an adverse prognostic value that can be circumvented by rituximab in patients with follicular lymphoma enrolled onto the GELA-GOELAMS FL-2000 trial. J Clin. Oncol 26:440–446
Pander J, Heusinkveld M, van der Straaten T et al (2011) Activation of tumor-promoting type 2 macrophages by EGFR-targeting antibody cetuximab. Clin Can Res 17:5668–5673
Biswas SK, Mantovani A. (2011) Macrophage plasticity and interaction with lymphocyte subsets: cancer as paradigm. Nat. Immunol. 889–896.
Herter S, Birk MC, Klein C et al (2014) Glycoengineering of Therapeutic Antibodies Enhances Monocyte/Macrophage-Mediated Phagocytosis and Cytotoxicity. J Immunol 192(5):2252–2260
Kirschfink M (2001) Targeting complement in therapy. Immunol Rev 180:177–189
Zipfel PF, Skerka C (2009) Complement regulators and inhibitory proteins. Nat Rev Immunol 9(10):729–740
Meyer S, Leusen JH, Boross P (2014) Regulation of complement and modulation of its activity in monoclonal antibody therapy of cancer. MAbs 6(5):1133–1144
Acknowledgements
We thank the staff of KMF for their dedicated support
Funding
This study was supported by Management Expenses Grants from the Shinko Hospital Hematologic Disease Center.
Author information
Authors and Affiliations
Consortia
Contributions
NN designed the research. NN organized the project. NN performed the statistical analysis. The remaining authors, NA, TT, SY, KI, KF, MH, SF, JK, NU, YS, HT, KO, SK, HY, SY, RY, YA, HH, HS, NH, TI, CS, ATK, JK, IM, and MH, interpreted the data. NN wrote the first draft. All other authors critically reviewed the draft and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics committee of the KMF and Shinko Hospital.
Competing interests
TT received honoraria from Sanofi K.K. and Janssen Pharmaceutical K.K. and grants from Bristol-Myers Squibb Co., Incyte Biosciences Japan G.K., GSK Corp. and Pfizer Japan Inc. SF received personal fees from Takeda Pharmaceutical Co., Ltd., Janssen Pharmaceutical K.K. and Sanofi K.K. JK received honoraria from Bristol-Myers Squibb Co., Janssen Pharmaceutical K.K., Takeda Pharmaceutical Co., Ltd., Sanofi K.K. and Ono Pharmaceutical Co., Ltd. and is in advisory roles at Janssen Pharmaceutical K.K, and Novartis Pharma K.K. NU received honoraria from Nippon Shinyaku Co., Ltd., Bristol-Myers Squibb Co. and Janssen Pharmaceutical K.K. KO received an honorarium from Takeda Pharmaceutical Co., Ltd. SY received honoraria from Janssen Pharmaceutical K.K. and Bristol-Myers Squibb Co. HH received honoraria from CLS Behring K.K. and Takeda Pharmaceutical Co., Ltd. HS received honoraria from Takeda Pharmaceutical Co., Ltd., Fujimoto Pharmaceutical Corp., Eisai Co., Ltd., Chugai Pharmaceutical Co., Ltd., AstraZeneca K.K., Ono Pharmaceutical Co., Ltd., Janssen Pharmaceutical K.K., Sanofi K.K., AbbVie G.K. and Novartis Pharma K.K., as well as grants from Bristol-Myers Squibb Co., Ono Pharmaceutical Co., Ltd. and AbbVie G.K. NH received personal fees from Otsuka Pharmaceutical K.K. and Chugai Pharmaceutical Co., Ltd. and grants from Chugai Pharmaceutical Co., Ltd., Teijin Pharma Corp., Kyowa Kirin Co., Eisai Co., Ltd., Ono Pharmaceutical Co., Ltd., Nippon Shinyaku Co., Ltd., AbbVie G.K., Shionogi Pharma and Asahi Kasei Pharma Corp. TI received honoraria from Bristol-Myers Squibb Co., CSL Behring, Janssen Pharmaceutical, Novartis Pharma, Takeda Pharmaceutical Co and AbbVie G.K. CS received honoraria from Janssen Pharmaceutical K.K., Bristol-Myers Squibb Co. and Sanofi K.K. AT-K received honoraria from Nippon Shinyaku Co., Ltd., Bristol-Myers Squibb Co., Janssen Pharmaceutical K.K., Otsuka Pharmaceutical K.K. and Megakaryon Corp., as well as personal fees from Ono Pharmaceutical Co., Ltd., COGNOA Corp., and PharmaEssentia Corp. JK is a consultant for Janssen Pharmaceutical, Pfizer, AbbVie and Bristol Myers Squibb; has received research funding from Kyowa Kirin, Chugai Pharmaceutical, Asahi Kasei, Sumitomo Pharma, Otsuka Pharmaceutical, Mochida Pharmaceutical and the Japan Blood Products Organization; and has received honoraria from Janssen Pharmaceutical, Kyowa Kirin, Chugai Pharmaceutical, Ono Pharmaceutical Co., Ltd. , Sanofi. K.K., Astra Zeneca. K.K., Astellas. K.K., Eisai Co. Ltd., AbbVie G.K., Novartis, Daiichi Sankyo, Amgen, Otsuka Pharmaceutical and BMS. IM received honoraria from AstraZeneca K.K., AbbVie G.K., Otsuka Pharmaceutical K.K., Novartis Pharma K.K., Pfizer Japan Inc., Janssen Pharmaceutical K.K., Ono Pharmaceutical Co., Ltd. and Bristol-Myers Squibb Co., as well as grants from Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Taiho Pharmaceutical, AbbVie GK., Otsuka Pharmaceutical K.K., Asahi Kasei Pharma Corporation, Sumitomo Pharma Co., Ltd. and Kyowa Kirin Co. MH received honoraria from Novartis Pharma K.K., Bristol-Myers Squibb Co., Otsuka Pharmaceutical K.K. and Kyowa Kirin Co.; personal fees from LabCorp Drug Development Japan Co., Ltd.; and grants from Otsuka Pharmaceutical K.K., Kyowa Kirin Co., Sekisui Medical Co., Ltd., Takeda Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co., Ltd. and Taiho Pharmaceutical. The other authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakamura, N., Arima, N., Takakuwa, T. et al. Efficacy of elotuzumab for multiple myeloma deteriorates after daratumumab: a multicenter retrospective study. Ann Hematol (2024). https://doi.org/10.1007/s00277-024-05705-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00277-024-05705-z