Abstract
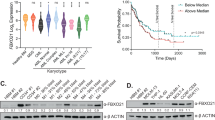
Chronic lymphocytic leukemia (CLL) mainly affects the health of older adults and is difficult to cure. Upstream stimulatory factor 2 (USF2) has been implicated in several diseases and conditions including cancers. However, the effect of USF2 on CLL has not been elucidated. To investigate the effect of USP2 on proliferation and autophagy of CLL, and to explore the underlying mechanism. The mRNA of USF2 and STIP1 homology and U-Box containing protein 1 (STUB1) was analyzed using qRT-PCR. Western blots were used to evaluate the expression level of USF2, LC3II, Beclin-1, P62, STUB1, and NFAT5. The cell proliferation was evaluated using CCK-8 and EdU assays. The cell apoptosis was evaluated using flow cytometry. Indirect fluorescent assay (IFA) was performed to analyze LC3 signal. Nuclear factor of activated T-cells 5 (NFAT5) ubiquitination was detected using immunoprecipitation (IP) assay. The CLL progression was evaluated in xenotransplantation model of nude mice. USF2 was highly expressed in CLL tissues and cell lines. USF2 knockdown suppressed the cell viability and EdU incorporation, while promoting cell apoptosis. Meanwhile, USF2 knockdown reduced the level of LC3II and Beclin-1, but increased P62, illustrating USF2 knockdown inhibiting autophagy. USF2 induced NFAT5 ubiquitination and promoted NFAT5 protein level via repressing STUB1. The downregulation of USF2 weakened CLL progression in xenotransplantation model of nude mice. CLL survival and autophagy was dependent on highly expressed USF2 which promoted the expression and ubiquitination of NFAT5 through inhibiting the transcription of STUB1, which makes USF2 a promising therapeutic candidate for CLL treatment.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
Hallek M, Shanafelt TD, Eichhorst B (2018) Chronic lymphocytic leukaemia. Lancet 391(10129):1524–1537. https://doi.org/10.1016/S0140-6736(18)30422-7
Bosch F, Dalla-Favera R (2019) Chronic lymphocytic leukaemia: from genetics to treatment. Nat Rev Clin Oncol 16(11):684–701. https://doi.org/10.1038/s41571-019-0239-8
Chen Y, Peng J, Cao S (2022) Bauerenol inhibits proliferation, migration and invasion of retinoblastoma cells via induction of apoptosis, autophagy and cell cycle arrest. Trop J Pharm Res 21(7):1377–1382. https://doi.org/10.4314/tjpr.v21i7.3
Kong Y-L, Huang Y, Wu J-Z, Cao X, Liang J-H, Xia Y, Wu W, Cao L, Zhu H-Y, Wang L (2018) Expression of autophagy related genes in chronic lymphocytic leukemia is associated with disease course. Leuk Res 66:8–14. https://doi.org/10.1016/j.leukres.2017.12.007
Kong Y-L, Pan B-H, Liang J-H, Zhu H-Y, Wang L, Xia Y, Wu J-Z, Fan L, Li J-Y, Xu W (2020) Chidamide, a histone deacetylase inhibitor, inhibits autophagy and exhibits therapeutic implication in chronic lymphocytic leukemia. Aging (Albany NY) 12(16):16083. https://doi.org/10.18632/aging.103536
Auberger P, Puissant A (2017) Autophagy, a key mechanism of oncogenesis and resistance in leukemia. Blood, J Am Soc Hematol 129(5):547–552. https://doi.org/10.1182/blood-2016-07-692707
Corre S, Galibert MD (2005) Upstream stimulating factors: highly versatile stress-responsive transcription factors. Pigment Cell Res 18(5):337–348. https://doi.org/10.1111/j.1600-0749.2005.00262.x
Wutthisathapornchai A, Vongpipatana T, Muangsawat S, Boonsaen T, MacDonald MJ, Jitrapakdee S (2014) Multiple E-boxes in the distal promoter of the rat pyruvate carboxylase gene function as a glucose-responsive element. PLoS ONE 9(7):e102730. https://doi.org/10.1371/journal.pone.0102730
Kumari D, Gabrielian A, Wheeler D, Usdin K (2005) The roles of Sp1, Sp3, USF1/USF2 and NRF-1 in the regulation and three-dimensional structure of the Fragile X mental retardation gene promoter. Biochemical J 386(2):297–303. https://doi.org/10.1042/BJ20041124
Tan Y, Chen Y, Du M, Peng Z, Xie P (2019) USF2 inhibits the transcriptional activity of Smurf1 and Smurf2 to promote breast cancer tumorigenesis. Cell Signal 53:49–58. https://doi.org/10.1016/j.cellsig.2018.09.013
Huang W-Y, Liao Z-B, Zhang J-C, Zhang X, Zhang H-W, Liang H-F, Zhang Z-Y, Yang T, Yu J, Ks Dong (2022) USF2-mediated upregulation of TXNRD1 contributes to hepatocellular carcinoma progression by activating Akt/mTOR signaling. Cell Death Disease 13(11):917. https://doi.org/10.1038/s41419-022-05363-x
Chen M, Li X, Du B, Chen S, Li Y (2022) Upstream stimulatory factor 2 inhibits erastin-induced ferroptosis in pancreatic cancer through transcriptional regulation of pyruvate kinase M2. Biochem Pharmacol 205:115255. https://doi.org/10.1016/j.bcp.2022.115255
Agirre X, Jiménez-Velasco A, San José-Enériz E, Garate L, Bandrés E, Cordeu L, Aparicio O, Saez B, Navarro G, Vilas-Zornoza A (2008) Down-regulation of hsa-miR-10a in chronic myeloid leukemia CD34+ cells increases USF2-mediated cell growth. Mol Cancer Res 6(12):1830–1840. https://doi.org/10.1158/1541-7786.MCR-08-0167
Tang DE, Dai Y, Lin LW, Xu Y, Liu DZ, Hong XP, Jiang HW, Xu SH (2019) STUB1 suppresseses tumorigenesis and chemoresistance through antagonizing YAP1 signaling. Cancer Sci 110(10):3145–3156. https://doi.org/10.1111/cas.14166
Chen BL, Li Y, Xu S, Nie Y, Zhang J (2021) NFAT5 Regulated by STUB1, facilitates malignant cell survival and p38 MAPK activation by upregulating AQP5 in chronic lymphocytic leukemia. Biochem Genet 59:870–883. https://doi.org/10.1007/s10528-021-10040-3
Huang J, Dai W, Xiao D, Xiong Q, Liu C, Hu J, Ge F, Yu X, Li S (2022) Acetylation-dependent SAGA complex dimerization promotes nucleosome acetylation and gene transcription. Nat Struct Mol Biol 29(3):261–273. https://doi.org/10.1038/s41594-022-00736-4
Yu J, Peng W, Xue Y, Li Y, Yang L, Geng Y (2022) FUBP1 promotes the proliferation of lung squamous carcinoma cells and regulates tumor immunity through PD-L1. Allergol et Immunopathol 50(5):68–74. https://doi.org/10.15586/aei.v50i5.659
Chi TF, Khoder-Agha F, Mennerich D, Kellokumpu S, Miinalainen I, Kietzmann T, Dimova EY (2020) Loss of USF2 promotes proliferation, migration and mitophagy in a redox-dependent manner. Redox Biol 37:101750. https://doi.org/10.1016/j.redox.2020.101750
Liu L, Ji X, Huang X (2022) Dexmedetomidine improves myocardial ischemia-reperfusion injury by increasing autophagy via PINK1/PRKN pathway. Signa Vitae 18:5. https://doi.org/10.22514/sv.2022.061
Cao X, Wan H, Wan H (2022) Urolithin A induces protective autophagy to alleviate inflammation, oxidative stress, and endoplasmic reticulum stress in pediatric pneumonia. Allergol et Immunopathol 50(6):147–153. https://doi.org/10.15586/aei.v50i6.743
Sato AYS, Antonioli E, Tambellini R, Campos AH (2011) ID1 inhibits USF2 and blocks TGF-β-induced apoptosis in mesangial cells. Am J Physiol-Renal Physiol 301(6):F1260–F1269. https://doi.org/10.1152/ajprenal.00128.2011
Kang K, Choi Y, Moon H, Seo M, Yoon J, Beck BR, Kang K (2020) RAD51 may regulate the expression of genes involved in autophagy through interaction with E-box binding proteins in cancer cell lines https://doi.org/10.20944/preprints202010.0452.v1
Zhou L, Wang Z, Chen X, Li X, Ge C, Min X, Zhao F, Chen T, Li J (2023) Syntaxin-6 promotes the progression of hepatocellular carcinoma and alters its sensitivity to chemotherapies by activating the USF2/LC3B axis. Int J Biol Sci 19(12):3892. https://doi.org/10.7150/ijbs.86636
Sun J, Ge X, Wang Y, Niu L, Tang L, Pan S (2022) USF2 knockdown downregulates THBS1 to inhibit the TGF-β signaling pathway and reduce pyroptosis in sepsis-induced acute kidney injury. Pharmacol Res 176:105962. https://doi.org/10.1016/j.phrs.2021.105962
Liu W, Xie A, Xiong J, Li S, Yang L, Liu W (2023) WDR3 promotes stem cell‐like properties in prostate cancer by inhibiting USF2‐mediated transcription of RASSF1A. J Gene Med e3498 https://doi.org/10.1002/jgm.3498
Author information
Authors and Affiliations
Contributions
Beili Chen and Yanyi Zhao designed the study, completed the experiment, and supervised the data collection; Shujuan Xu analyzed the data and interpreted the data; Fang Jiang, Yuwei Nie, Ailin Tang, and Qin Zhou prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Ethical approval was obtained from the Ethics Committee of Affiliated Hospital of Guilin Medical University.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, B., Zhao, Y., Xu, S. et al. USF2 promotes autophagy and proliferation in chronic lymphocytic leukemia by inhibiting STUB1-induced NFAT5 ubiquitination. Ann Hematol 103, 533–544 (2024). https://doi.org/10.1007/s00277-023-05522-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-023-05522-w