Abstract
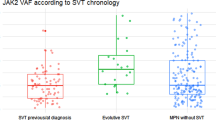
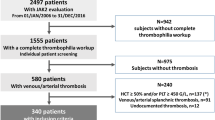
Here, we reviewed clinical-morphological data and investigated mutational profiles by NGS in a single-center series of 58 consecutive MPN-SVT patients admitted to our hospital between January 1979 and November 2021. We identified 15.5% of PV, 13.8% of ET, 34.5% of PMF, 8.6% of SMF and 27.6% of MPN-U. Most cases (84.5%) carried JAK2V617F mutation, while seven patients were characterized by other molecular markers, namely MPL in four and CALR mutations in three cases. NGS was performed in 54 (93.1%) cases: the most frequent additional mutations were found in TET2 (27.8%) and DNMT3A (16.7%) genes, whereas 25 (46.3%) patients had no additional mutation. Cases with JAK2V617F homozygosity had a higher median number of additional mutations than those with low allele burden. More importantly, all cases of leukemic evolution were characterized by a higher median number of co-mutations, and a co-mutational pattern of high-risk lesions, such as truncating mutations of ASXL1, bi-allelic TP53 loss, and CSMD1 mutations. Nevertheless, no difference was found between cases with and without additional somatic mutations regarding fibrotic progression, SVT recurrence, other thrombo-hemorrhagic complications, or death. After a median follow-up of 7.1 years, ten deaths were recorded; fibrotic progression/leukemic evolution was ascertained in one (1.7%) and six (10.3%) patients, respectively, while 22 (37.9%) patients suffered from recurrent thrombosis. In conclusion, our data underline the importance of using NGS analysis in the management of MPN-related SVT as it can support the MPN diagnosis, particularly in “triple-negative” cases, and provide additional information with potential consequences on prognosis and therapeutic strategies.




Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the senior authors upon reasonable request.
References
Barbui T, Carobbio A, Cervantes F et al (2010) Thrombosis in primary myelofibrosis: incidence and risk factors. Blood 115:778–782
Falanga A, Marchetti M (2012) Thrombotic disease in the myeloproliferative neoplasms. Hematology (Am Soc Hematol Educ Program) 2012:571–581
Harrison CN, Campbell PJ, Buck G et al (2005) Hydroxyurea compared with anagrelide in high-risk essential thrombocythemia. N Engl J Med 353:33–45
Gisslinger H, Gotic M, Holowiecki J et al (2013) Anagrelide compared to hydroxyurea in WHO-classified essential thrombocythemia: the ANAHYDRET Study, a randomized controlled trial. Blood 121:1720–1728
Carobbio A, Thiele J, Passamonti F et al (2011) Risk factors for arterial and venous thrombosis in WHO-defined essential thrombocythemia: an international study of 891 patients. Blood 117:5857–5859
Marchioli R, Finazzi G, Landolfi R et al (2005) Vascular and neoplastic risk in a large cohort of patients with polycythemia vera. J Clin Oncol 23:2224–2232
Dentali F, Ageno W, Rumi E et al (2014) Cerebral venous thrombosis and myeloproliferative neoplasms: results from two large databases. Thromb Res 134:41–43
Martinelli I, De Stefano V, Carobbio A et al (2014) Cerebral vein thrombosis in patients with Philadelphia-negative myeloproliferative neoplasms. An European Leukemia Net study. Am J Hematol 89:E200–E205
Thatipelli MR, McBane RD, Hodge DO, Wysokinski WE (2010) Survival and recurrence in patients with splanchnic vein thrombosis. Clin Gastroenterol Hepatol 8:200–205
Finazzi G, De Stefano V, Barbui T (2013) Are MPNs Vascular Diseases? Curr Hematol Malig Rep 8:307–316
Cortelezzi A, Moia M, Falanga A et al (2005) Incidence of thrombotic complications in patients with haematological malignancies with central venous catheters: a prospective multicentre study. Br J Haematol 129:811–817
Elkrief L, Payancé A, Plessier A et al (2023) Management of splanchnic vein thrombosis. JHEP Rep. https://doi.org/10.1016/j.jhepr.2022.100667
Smalberg JH, Arends LR, Valla DC, Kiladjian JJ, Janssen HL, Leebeek FW (2012) Myeloproliferative neoplasms in Budd-Chiari syndrome and portal vein thrombosis: a meta-analysis. Blood 120:4921–4928
Darwish Murad S, Plessier A, Hernandez-Guerra M et al (2009) Etiology, management, and outcome of the Budd-Chiari syndrome. Ann Intern Med 151:167–175
Bayraktar Y, Harmanci O, Buyukasik Y et al (2008) JAK2V617F mutation in patients with portal vein thrombosis. Dig Dis Sci 53:2778–2783
Valla D, Casadevall N, Huisse MG et al (1988) Etiology of portal vein thrombosis in adults: a prospective evaluation of primary myeloproliferative disorders. Gastroenterology 94:1063–1069
Donadini MP, Dentali F, Ageno W (2012) Splanchnic vein thrombosis: new risk factors and management. Thromb Res 129:S93–S96
Chagneau-Derrode C (2013) Impact of cytoreductive therapy on the outcome of patients with myeloproliferative neoplasms and hepato-splanchnic vein thrombosis. Hepatology 58:847A-898A
Primignani M, Barosi G, Bergamaschi G et al (2006) Role of the JAK2 mutation in the diagnosis of chronic myeloproliferative disorders in splanchnic vein thrombosis. Hepatology 44:1528–1534
Jones C, Levy Y, Tong AW (2014) Elevated serum erythropoietin in a patient with polycythaemia vera presenting with Budd-Chiari syndrome. BMJ Case Rep 2014:1–4
Sekhar M, McVinnie K, Burroughs AK (2013) Splanchnic vein thrombosis in myeloproliferative neoplasms. Br J Haematol 162:730–747
Bergamaschi GM, Primignani M, Barosi G et al (2008) MPL and JAK2 exon 12 mutations in patients with the Budd-Chiari syndrome or extrahepatic portal vein obstruction. Blood 111:4418
Kiladjian JJ, Cervantes F, Leebeek FW et al (2008) The impact of JAK2 and MPL mutations on diagnosis and prognosis of splanchnic vein thrombosis: a report on 241 cases. Blood 111:4922–4929
Fiorini A, Chiusolo P, Rossi E et al (2009) Absence of the JAK2 exon 12 mutations in patients with splanchnic venous thrombosis and without overt myeloproliferative neoplasms. Am J Hematol 84:126–127
Magaz M, Alvarez-Larrán A, Colomer D et al (2021) Next-generation sequencing in the diagnosis of non-cirrhotic splanchnic vein thrombosis. J Hepatol 74:89–95
Iurlo A, Cattaneo D, Gianelli U, Fermo E, Augello C, Cortelezzi A (2015) Molecular analyses in the diagnosis of myeloproliferative neoplasm-related splanchnic vein thrombosis. Ann Hematol 94:881–882
Klampfl T, Gisslinger H, Harutyunyan AS et al (2013) Somatic mutations of calreticulin in myeloproliferative neoplasms. N Eng J Med 369:2379–2390
Nangalia J, Massie CE, Baxter EJ et al (2013) Somatic CALR mutations in myeloproliferative neoplasms with nonmutated JAK2. N Eng J Med 369:2391–2405
Turon F, Cervantes F, Colomer D, Baiges A, Hernández-Gea V, Garcia-Pagán JC (2015) Role of calreticulin mutations in the etiological diagnosis of splanchnic vein thrombosis. J Hepatol 62:72–74
Haslam K, Langabeer SE (2015) Incidence of CALR mutations in patients with splanchnic vein thrombosis. Br J Haematol 168:459–460
Gianelli U, Iurlo A, Cattaneo D et al (2015) Discrepancies between bone marrow histopathology and clinical phenotype in BCR-ABL1-negative myeloproliferative neoplasms associated with splanchnic vein thrombosis. Leuk Res 39:525–529
Grinfeld J, Nangalia J, Baxter EJ et al (2018) Classification and personalized prognosis in myeloproliferative neoplasms. N Engl J Med 379:1416–1430
Ortmann CA, Kent DG, Nangalia J et al (2015) Effect of mutation order on myeloproliferative neoplasms. N Engl J Med 372:601–612
Lavu S, Szuber N, Mudireddy M et al (2018) Splanchnic vein thrombosis in patients with myeloproliferative neoplasms: the Mayo clinic experience with 84 consecutive cases. Am J Hematol 93:E61–E64
Cattaneo D, Gianelli U, Bianchi P, Cortelezzi A, Iurlo A (2018) Heterogeneity among splanchnic vein thrombosis associated with myeloproliferative neoplasms. Eur J Intern Med 52:e25–e26
Hoekstra J, Bresser EL, Smalberg JH, Spaander MC, Leebeek FW, Janssen HL (2011) Long-term follow-up of patients with portal vein thrombosis and myeloproliferative neoplasms. J Thromb Haemost 9:2208–2214
Gangat N, Wolanskyj AP, Tefferi A (2006) Abdominal vein thrombosis in essential thrombocythemia: prevalence, clinical correlates, and prognostic implications. Eur J Haematol 77:327–333
De Stefano V, Vannucchi AM, Ruggeri M et al (2016) Splanchnic vein thrombosis in myeloproliferative neoplasms: risk factors for recurrences in a cohort of 181 patients. Blood Cancer J 6:e493
Sant’Antonio E, Guglielmelli P, Pieri L et al (2020) Splanchnic vein thromboses associated with myeloproliferative neoplasms: An international, retrospective study on 518 cases. Am J Hematol 95:156–166
Barosi G, Vannucchi AM, De Stefano V et al (2014) Identifying and addressing unmet clinical needs in Ph-neg classical myeloproliferative neoplasms: a consensus-based SIE, SIES, GITMO position paper. Leuk Res 38:155–160
Marchioli R, Finazzi G, Specchia G et al (2013) Cardiovascular events and intensity of treatment in polycythemia vera. N Engl J Med 368:22–33
Barosi G, Mesa RA, Thiele J et al (2008) Proposed criteria for the diagnosis of post-polycythemia vera and post-essential thrombocythemia myelofibrosis: a consensus statement from the International Working Group for Myelofibrosis Research and Treatment. Leukemia 22:437–438
Swerdlow SH, Campo E, Harris L et al. (2008) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, 4th ed. IARC, Lyon, 127–129
Baxter EJ, Scott LM, Campbell PJ et al (2005) Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 365:1054–1061
Bolli N, Manes N, McKerrell T et al (2015) Characterization of gene mutations and copy number changes in acute myeloid leukemia using a rapid target enrichment protocol. Haematologica 100:214–222
McKerrell T, Moreno T, Ponstingl H et al (2016) Development and validation of a comprehensive genomic diagnostic tool for myeloid malignancies. Blood 128:e1–e9
Nik-Zainal S, Van Loo P, Wedge DC et al (2012) The life history of 21 breast cancers. Cell 149:994–1007
Bolli N, Biancon G, Moarii M et al (2018) Analysis of the genomic landscape of multiple myeloma highlights novel prognostic markers and disease subgroups. Leukemia 32:2604–2616
Ziccheddu B, Biancon G, Bagnoli F et al (2020) Integrative analysis of the genomic and transcriptomic landscape of double-refractory multiple myeloma. Blood Adv 4:830–844
Rack KA, van den Berg E, Haferlach C et al (2019) European recommendations and quality assurance for cytogenomic analysis of haematological neoplasms. Leukemia 33:1851–1867
Gianelli U, Iurlo A, Vener C et al (2008) The significance of bone marrow biopsy and JAK2V617F mutation in the differential diagnosis between the early pre-polycythemic phase of polycythemia vera and essential thrombocythemia. Am J Clin Pathol 130:336–342
Spivak JL, Silver RT (2011) The treatment of essential thrombocytosis revisited. Blood 28:1179–1180
Silver RT, Chow W, Orazi A, Arles SP, Goldsmith SJ (2013) Evaluation of WHO criteria for diagnosis of polycythemia vera: a prospective analysis. Blood 122:1881–1886
Rosti V, Villani L, Riboni R et al (2013) Spleen endothelial cells from patients with myelofibrosis harbor the JAK2V617F mutation. Blood 121:360–368
Giordano G, Napolitano M, Cellurale M et al (2022) Circulating Endothelial Cell Levels Correlate with Treatment Outcomes of Splanchnic Vein Thrombosis in Patients with Chronic Myeloproliferative Neoplasms. J Pers Med 12:364
Pieri L, Paoli C, Arena U et al (2017) Safety and efficacy of ruxolitinib in splanchnic vein thrombosis associated with myeloproliferative neoplasms. Am J Hematol 92:187–195
Barosi G, Buratti A, Costa A et al (1991) An atypical myeloproliferative disorder with high thrombotic risk and slow disease progression. Cancer 68:2310–2318
Segura-Díaz A, Stuckey R, Florido Y et al (2020) Thrombotic risk detection in patients with polycythemia vera: the predictive role of DNMT3A/TET2/ASXL1 mutations. Cancers (Basel) 12:934
Solary E, Bernard OA, Tefferi A, Fuks F, Vainchenker W (2014) The Ten-Eleven Translocation-2 (TET2) gene in hematopoiesis and hematopoietic diseases. Leukemia 28:485–496
Delhommeau F, Dupont S, Della Valle V et al (2009) Mutation in TET2 in myeloid cancers. N Engl J Med 360:2289–2301
Colaizzo D, Tiscia GL, Pisanelli D et al (2010) New TET2 gene mutations in patients with myeloproliferative neoplasms and splanchnic vein thrombosis. J Thromb Haemost 8:1142–1144
Sousos N, Ní Leathlobhair M, Simoglou Karali C et al (2022) In utero origin of myelofibrosis presenting in adult monozygotic twins. Nat Med 28:1207–1211
Fuster JJ, MacLauchlan S, Zuriaga MA et al (2017) Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 355:842–847
Laurie CC, Laurie CA, Rice K et al (2012) Detectable clonal mosaicism from birth to old age and its relationship to cancer. Nat Genet 44:642–650
Jaiswal S, Fontanillas P, Flannick J et al (2014) Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med 371:2488–2498
Genovese G, Kähler AK, Handsaker RE et al (2014) Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med 371:2477–2487
Steensma DP, Bejar R, Jaiswal S et al (2015) Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood 126:9–16
Jaiswal S, Natarajan P, Silver AJ et al (2017) Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N Engl J Med 377:111–121
Westbrook R, Lea N, Mohamedali A et al (2012) Prevalence and clinical outcomes of the 46/1 haplotype, Janus kinase 2 mutations, and ten-eleven translocation 2 mutations in Budd-Chiari syndrome and their impact on thrombotic complications post liver transplantation. Liver Transpl 18:819–827
How J, Zhou A, Oh ST (2017) Splanchnic vein thrombosis in myeloproliferative neoplasms: pathophysiology and molecular mechanisms of disease. Ther Adv Hematol 8:107–118
Iurlo A, Cattaneo D, Gianelli U (2019) Blast Transformation in Myeloproliferative Neoplasms: Risk Factors, Biological Findings, and Targeted Therapeutic Options. Int J Mol Sci 20:1839
Debureaux PE, Cassinat B, Soret-Dulphy J et al (2020) Molecular profiling and risk classification of patients with myeloproliferative neoplasms and splanchnic vein thromboses. Blood Adv 4:3708–3715
Cattaneo D, Croci GA, Bucelli C et al (2021) Triple-Negative Essential Thrombocythemia: Clinical-Pathological and Molecular Features. A Single-Center Cohort Study. Front Oncol 11:637116
Maddali M, Venkatraman A, Kulkarni UP et al (2022) Molecular characterization of triple-negative myeloproliferative neoplasms by next-generation sequencing. Ann Hematol 101:1987–2000
Acknowledgements
This study was partially funded by Italian Ministry of Health—Current research IRCCS to NB. NB is recipient of an Investigator grant from the Associazione Italiana Ricerca sul Cancro (AIRC IG n. 25739).
Author information
Authors and Affiliations
Contributions
D. Cattaneo, N. Bolli, and A. Iurlo were responsible for study concept and design and wrote the paper. D. Consonni was responsible for statistical analyses. A. Marchetti, M. Lionetti, E. Fermo, A. Maeda, A. Marella, A. Neri, and N. Bolli made and interpreted mutational analyses. U. Gianelli reviewed bone marrow biopsies. D. Cattaneo, C. Bucelli, V. Bellani, C. De Magistris, M. Primignani, L. Baldini, and A. Iurlo followed the patients and collected data. All authors critically reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest disclosures
All the authors declare they have no potential conflicts of interest.
Ethical statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Niccolò Bolli and Alessandra Iurlo co-contributing senior authors.
Supplementary Information
Below is the link to the electronic supplementary material.

277_2023_5217_Fig5_ESM.png
Supplementary Fig. S4 VAF mutation study with flanking CIs in our patient cohort. VAF was modeled according to a binomial distribution. (PNG 234 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cattaneo, D., Bucelli, C., Marchetti, A. et al. Pathological and genomic features of myeloproliferative neoplasms associated with splanchnic vein thrombosis in a single-center cohort. Ann Hematol 102, 1409–1420 (2023). https://doi.org/10.1007/s00277-023-05217-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-023-05217-2