Abstract
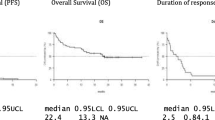
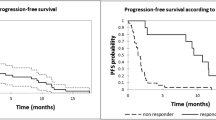
We report herein a multicentre retrospective analysis of 192 consecutive patients with symptomatic refractory/relapsed multiple myeloma (RRMM) treated with daratumumab in combination with bortezomib or lenalidomide as salvage therapy at 9 haematological centres in Puglia. Choice of both regimens was based on previous treatment and/or physicians’ preference. Considering the under-representation of older patients (very old patient ≥ 80 years) in clinical trials and the prognostic and predictive importance and value of frailty status, here, we further characterised the patient cohort by age. The overall response rate (ORR) was generally lower than what was previously reported in the CASTOR (ORR 72.6% vs 85%) and POLLUX (ORR 86.5% vs 93%) trials. The lower ORR in our analysis compared to the CASTOR and POLLUX trials could be related to a less selected population. Similarly, amongst very old patients, the ORR was encouraging: ORR to treatment with DVd (daratumumab + bortezomib + dexamethasone) was 66.7%, and ORR to treatment with DRd (daratumumab + lenalidomide + dexamethasone) was 92.3%. Median TTP (time to progression) was 10.8 months (1-year TTP: 44.7%; 2-year TTP: 25.3%) in the DVd group; median TTP was not reached in the DRd group (1-year TTP: 82.7%; 2-year TTP: 71.4%). Median OS (overall survival) was not reached either in the DRd group (1-year OS: 85.9%; 2-year OS: 73.7%) or the DVd group (1-year OS: 70.2%; 2-year OS: 58.9%).




Similar content being viewed by others
References
Usmani SZ, Weiss BM, Pleisner T et al (2016) Clinical efficacy of Daratumumab monotherapy in patients with heavily pretreated relapsed or refractory multiple myeloma. Blood 128(1):37–44
Palumbo A, Chanan-Khan A, Weisel K et al (2016) Daratumumab, bortezomib and dexamethasone for multiple myeloma. N Engl J Med 375(8):754–766
Dimopoulos MA, Oriol A, Nahi H et al (2016) Daratumumab, lenalidomide and dexamethasone for multiple myeloma. N Engl J Med 375(14):1319–1331
Mateos MV, Sonneveld P, Hungria V et al (2020) Daratumumab, bortezomib and dexamethasone versus bortezomib and dexamethasone in patients with previously treated multiple myeloma: three-years follow-up of CASTOR. Clin Lymphoma Myeloma Leuk 20(8):509–518
Weisel KC, Sonneveld P, Mateos VM et al (2019) Efficacy and safety of daratumumab, bortezomib and dexamethasone (D-Vd) versus bortezomib and dexamethasone (Vd) in first relapse patients with multiple myeloma (MM): four-year update of Castor. Blood 134(s2):Abstr 3192
Kaufman JL, Usmani SZ, San-Miguel J et al (2019) Four-year follow-up of the phase 3 Pollux study of daratumumab plus lenalidomide and dexamethasone (D-Rd) versus lenalidomide and dexamethasone (Rd) alone in relapsed or refractory multiple myeloma (RRMM). Blood 134(s2):Abstr 1866
Mateos MV, Dimopoulos MA, Cavo M et al (2018) Daratumumab plus bortezomib, melphalan and prednisone for untreated myeloma. N Engl J Med 378(6):518–528
Facon T, Kumar S, Plesner T et al (2019) Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N Eng J Med 380(22):2104–2115
Palumbo A, Rajkumar SV, San Miguel JF et al (2014) International Myeloma Working Group consensus statement for the management, treatment and supportive care of patients with myeloma not eligible for standard autologous stem-cell transplantation. J Clin Oncol 32(6):587–600
Offidani M, Boccadoro M, Di Raimondo F et al (2019) Expert panel consensus statement for proper evaluation of first relapse in multiple myeloma. Curr Hematol Malig Rep 14:187–196
Gavriatopoulou M, Kastritis E, Ntanasis-Stathopoulos I et al (2018) The addition of IMiDs for patients with daratumumab-refractory multiple myeloma can overcome refractoriness to both agents. Blood 131(4):464–467
Lovas S, Varga G, Farkas P et al (2019) Real-word data on efficacy and safety of daratumumab treatment in Hungarian relapsed/refractory multiple myeloma patients. Int J Hematol 110(5):559–565
Antonioli E, Staderini M, Pilerci S et al (2020) Daratumumab, lenalidomide and dexamethasone combination in relapsed/refractory myeloma patients: a real-life single-center experience. Leuk Lymphoma 61(13):3255–3258
Davies F, Rifkin R, Costello C et al (2021) Real-world comparative effectiveness of triplets containing bortezomib (B), carfilzomib (C), daratumumab (D), or ixazomib (I) in relapsed/refractory multiple myeloma (RRMM) in the US. Ann Hematol 100:2325–2327
Fazio F, Franceschini L, Tomarchio V et al (2021) Daratumumab combined with dexamethasone and lenalidomide or bortezomib in relapsed/refractory multiple myeloma (RRMM) patients: report from the multiple myeloma GIMEMA Lazio group. eJHaem: 1–8. https://doi.org/10.1080/10428194.2020.1802452
Kumar S, Paiva B, Anderson KC et al (2016) International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol 17(8):e328–e346
Palumbo A, Bringhen S, Mateos MV et al (2015) Geriatric assessment predicts survival and toxicities in elderly myeloma patients: an International Myeloma Working Group report. Blood 125(13):2068–2074
Kaplan EL, Meier P (1958) Non-parametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Wildes TM, Campagnaro E (2017) Management of multiple myeloma in older adults: gaining ground with geriatric assessment. J Geriatr Oncol 8(1):1–7
Panitsas F, Kothari J, Vallance G et al (2018) Treat o palliate: outcome of very elderly myeloma patients. Haematologica 103:e32
de Weers M, Tai YT, van der Veer MS et al (2011) Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other haematological tumors. J Immunol 186:1840–1848
Krejcik J, Casneuf T, Nijhof IS et al (2016) Daratumumab depletes CD38+ immune regulatory cells, promotes T-cell expansion and skews T-cell repertoire in multiple myeloma. Blood 128:384–394
Overdijk MB, Verploegen S, Bogels M et al (2015) Antibody-mediated phagocytosis contributes to the anti-tumor activity of the therapeutic antibody daratumumab in lymphoma and multiple myeloma. mAbs 7:311–321
Fedele PL, Willis SN, Liao Y et al (2018) IMiDs prime myeloma cells for daratumumab-mediated cytotoxicity through loss of Ikaros and Aiolos. Blood 132(20):2166–2178
Author information
Authors and Affiliations
Contributions
All authors have contributed to the preparation of the manuscript. In detail, Mele G. has contributed to the acquisition, analysis and interpretation of data, and contributed to drafts of the manuscript. Curci P., Falcone M.P., Germano C., Mele A., Palazzo G., Palumbo G., Reddiconto G. and Rossini B. were involved in the collection of clinical and laboratory data. Cascavilla N., Di Renzo N., Guarini A., Melillo L., Pavone V., Tarantini G., Specchia G., Musto P. and Pastore D. were involved in the critical revision of the article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mele, G., Cascavilla, N., Di Renzo, N. et al. Daratumumab plus bortezomib or daratumumab plus lenalidomide as salvage therapy for patients with myeloma: initial follow-up of an Italian multicentre retrospective clinical experience by ‘Rete Ematologica Pugliese’. Ann Hematol 101, 1727–1739 (2022). https://doi.org/10.1007/s00277-022-04857-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-022-04857-0